Silane coupling agents (SCAs) are chemical compounds with a dual-function structure, typically represented as R-Si-(X)₃. These agents act as molecular bridges, binding inorganic fillers like silica or alumina to organic elastomers. This bonding improves the mechanical properties, dispersion, and durability of hybrid filler systems. Key applications include tire manufacturing, where SCAs enhance rolling resistance, wet grip, and abrasion resistance, as well as automotive seals and industrial hoses.
SCAs work through a two-step process: hydrolysis and surface bonding with fillers, followed by linking to elastomer matrices. Common types include epoxy silanes (e.g., Glycidoxypropyltrimethoxysilane), aminosilanes (e.g., Aminopropyltriethoxysilane), and methacryloxy silanes (e.g., Methacryloxypropyltrimethoxysilane). Each type is tailored for specific polymer systems, such as epoxies, polyurethanes, or polyesters.
Proper application methods, such as filler pretreatment or direct blending, and precise control of reaction conditions (e.g., pH 4–5, moisture levels) are critical for achieving optimal performance. SCAs also reduce moisture absorption, improve tensile strength, and ensure uniform filler dispersion, making them essential for advanced elastomer applications.
HOLD TIGHT! Coupling Agents for Polymer-Matrix Composites (Basics)
sbb-itb-aa4586a
How Silane Coupling Agents Work in Hybrid Filler Systems
How Silane Coupling Agents Bond Fillers to Elastomers: Two-Step Chemical Process
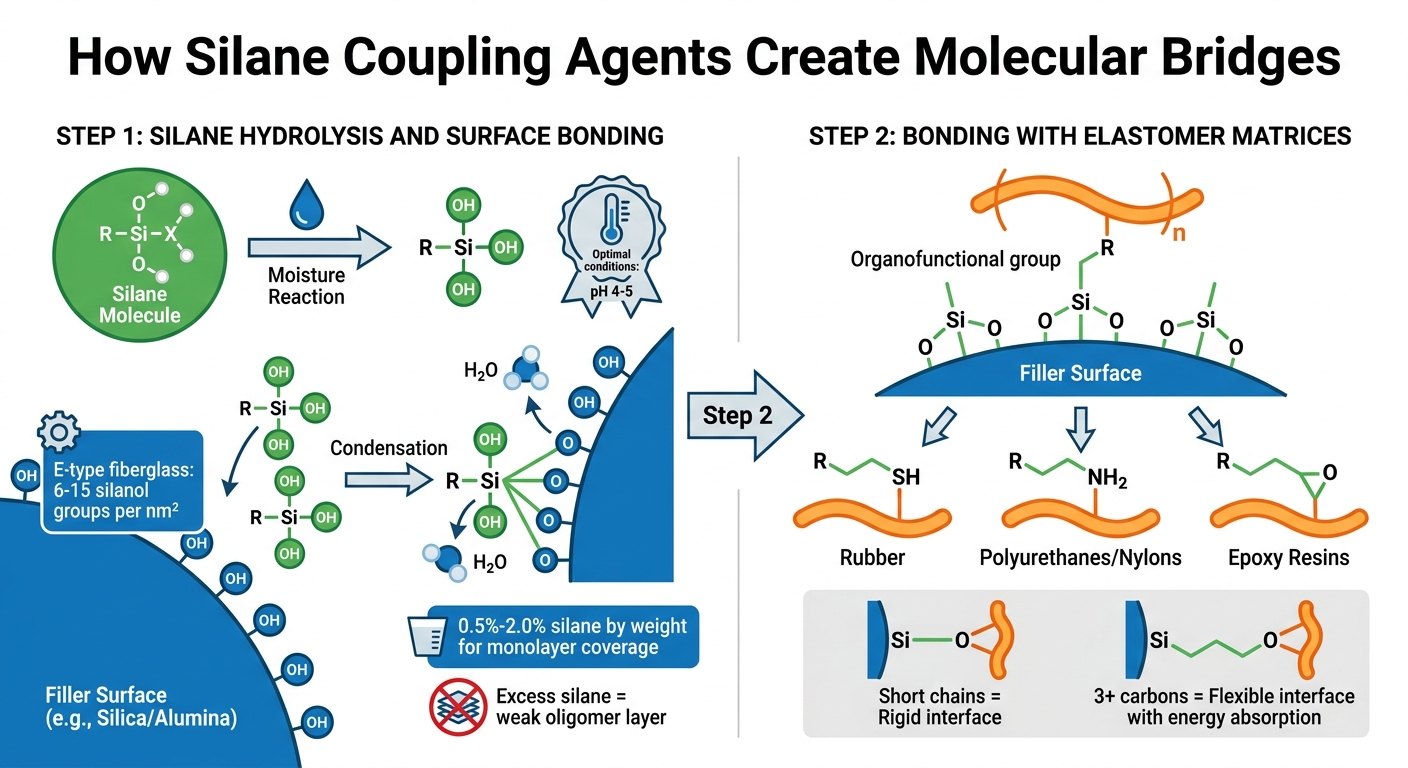
Silane coupling agents create a bridge between fillers and elastomers through a two-step bonding process: attaching to the filler surface and linking to the elastomer. Each step involves specific chemical reactions that convert weak physical interactions into durable covalent bonds.
Silane Hydrolysis and Surface Bonding
The process starts when the silane’s hydrolyzable groups, like methoxy or ethoxy groups, react with moisture to form reactive silanol groups (Si-OH). This hydrolysis occurs optimally at a pH of 4–5, ensuring full conversion without triggering premature silane oligomerization [1]. Once formed, the silanol groups bond with hydroxyl groups on fillers such as silica or alumina. Through a condensation reaction, these groups create strong siloxane bonds (Si-O-Substrate), which are far stronger than hydrogen or van der Waals bonds [1].
The success of this bonding depends on the availability of surface hydroxyl groups. For example, E-type fiberglass offers 6–15 silanol groups per square nanometer, providing plenty of bonding sites [2]. Most mineral fillers need 0.5%–2.0% silane by weight to achieve full monolayer coverage [1]. However, exceeding this range can lead to a weak layer of unreacted silane oligomers, which may reduce mechanical strength rather than enhance it [1].
This initial bonding step prepares the silane for its next role – linking to elastomer matrices.
Bonding with Elastomer Matrices
After bonding to the filler, the silane’s organofunctional group connects with the elastomer matrix, completing the hybrid system. One end is anchored to the filler, while the organofunctional group bonds to the polymer. The choice of silane depends on the polymer type: mercapto silanes work well with rubber, while amino silanes are suited for polyurethanes or nylons [1][2]. This compatibility ensures the organofunctional group can bond with the polymer during mixing or curing, forming a molecular bridge between the filler and the matrix.
The length of the silane’s alkyl chain also plays a critical role. Shorter chains create a rigid interface, while chains with three or more carbons provide flexibility and energy absorption [1][2]. This adaptability allows formulators to adjust the mechanical properties of elastomer compounds by selecting silanes with the right chain length, improving dispersion, strength, and durability.
Disclaimer: This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Types of Silane Coupling Agents for Hybrid Filler Systems
When tailoring filler–elastomer interfaces to fit specific polymer chemistries, three primary types of silane coupling agents stand out. The choice of silane depends on aligning its functional group with the chemistry of the elastomer, ensuring optimal bonding and performance.
Epoxy Silanes
Epoxy silanes are equipped with an epoxy-functional group, making them a strong fit for epoxy, phenolic, and polyimide polymer systems. A commonly used example, Glycidoxypropyltrimethoxysilane (GPTMS), forms covalent bonds during curing. This occurs when reactive groups in the polymer interact with the epoxy ring, resulting in improved tensile strength and impact resistance.
Aminosilanes
Aminosilanes feature an amino-functional group, allowing them to bond effectively with systems like polyurethane, nylon, acrylic, and phenolic. Aminopropyltriethoxysilane (APTES) is a standout in this category. It reacts with isocyanates in polyurethanes and carbonyl groups in nylons, providing strong adhesion and thermal stability. This makes it particularly useful for high-temperature thermoplastics. Additionally, the amino group can form hydrogen bonds, offering flexibility while maintaining covalent bonding during processing.
Methacryloxy Silanes
Methacryloxy silanes, characterized by their methacrylate group, are ideal for unsaturated polyester and vinyl ester resins. 3-Methacryloxypropyltrimethoxysilane (MPTMS) is a standard choice here. Its methacrylate group reacts more readily than vinyl silanes in thermoset polyester systems, delivering enhanced UV resistance and durability. MPTMS is especially beneficial in outdoor applications, as it helps maintain mechanical properties even under UV exposure and repeated moisture cycles.
| Silane Type | Example Compound | Recommended Polymer Matrix |
|---|---|---|
| Epoxy Silane | Glycidoxypropyltrimethoxysilane (GPTMS) | Epoxy, Phenolic, Polyimide |
| Aminosilane | Aminopropyltriethoxysilane (APTES) | Polyurethane, Nylon, Acrylic, Phenolic |
| Methacryloxy Silane | Methacryloxypropyltrimethoxysilane (MPTMS) | Unsaturated Polyester, Vinyl Ester |
Disclaimer: This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Benefits of Silane Coupling Agents in Elastomeric Hybrid Fillers
Silane coupling agents play a crucial role in enhancing filler-matrix bonding, leading to better mechanical performance, improved dispersion, and increased resistance to heat and moisture in elastomer hybrids.
Improved Mechanical Performance
Silanes create covalent links between inorganic fillers and elastomer chains during vulcanization. This bonding significantly boosts properties like tensile strength, modulus (up to 300% reinforcement), and abrasion resistance. For instance, silica-filled natural rubber shows a 40% increase in tensile strength. In applications like green tire technology, bifunctional organosilanes such as TESPT (Bis[3-(triethoxysilyl)propyl]tetrasulfide) can lower rolling resistance by 20% to 30%. This reduction in rolling resistance also helps decrease the operating temperature of the tire carcass, making it more efficient and durable.
These mechanical advantages are further supported by better filler dispersion techniques.
Enhanced Filler Dispersion
Silanes reduce the surface energy of fillers, preventing agglomeration and minimizing the Payne Effect. This results in lower compound viscosity and more uniform mixing. Uniform particle distribution not only improves processing but also contributes to longer shelf life and better resistance to environmental wear over time.
Heat and Moisture Resistance
Silane treatment also provides hydrophobic properties, cutting water absorption by over 50%. Additionally, durable siloxane bonds formed through this process enhance thermal stability and reduce heat build-up in dynamic applications, making the material more reliable under stress.
| Property | Untreated Hybrid Filler | Silane-Treated Hybrid Filler |
|---|---|---|
| Moisture Absorption | High (Hydrophilic) | Low (Hydrophobic) |
| Tensile Strength | Lower (Poor stress transfer) | Higher (Covalent coupling) |
| Dispersion Quality | Poor (Agglomerates) | Excellent (Uniform) |
| Heat Build-up | High (High internal friction) | Low (Reduced Payne Effect) |
| Processing Viscosity | High (Filler-filler interaction) | Low (Improved flow) |
Disclaimer: This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
How to Apply Silane Coupling Agents in Hybrid Filler Systems
When integrating silane coupling agents into elastomer formulations, there are two main approaches: pretreating the filler particles or adding the silane during compounding. The choice between these methods depends on the specific application and the reaction conditions required.
Filler Pretreatment
This method involves treating the filler particles – such as silica, calcium carbonate, clays, or alumina – with a silane solution before mixing them with the elastomer. The goal is to chemically modify the filler surface, creating bonds that wouldn’t naturally form between the filler and elastomer molecules. The hydrolysis-condensation process forms stable siloxane linkages, enhancing compatibility.
Filler pretreatment is particularly effective for glass fiber products, as it can significantly improve mechanical and electrical properties. Selecting the right silane for the resin system is critical. For instance:
- Use vinyl- or methacryloxy-containing silanes with unsaturated polyesters.
- Opt for epoxy- or amino-containing silanes with epoxy resins.
Ensure adequate moisture is present during application to facilitate the necessary chemical reactions.
Direct Blending with Compounds
Also called integral blending, this method involves adding the silane directly into the elastomer or resin during compounding. It’s especially suitable for white fillers that lack natural chemical affinity with elastomer molecules. In thermosetting systems, silanes may be mixed with the binder resin or curing catalyst to enhance the final composite’s strength.
For thermoplastic resins like polypropylene, specific combinations of silanes and additives can create effective coupling sites. For example:
- Use vinyl or methacrylic silanes with an organic peroxide like dicumyl peroxide (0.15%-0.25%).
- Consider 3-mercaptopropyl trimethoxysilane or a mix of aminoalkyl silane and chlorinated compounds (e.g., chlorinated xylene) for synergistic effects.
In phenolic or furan resin systems, aminosilanes can boost mold strength, allowing for a reduction in binder resin usage.
Processing Conditions for Best Results
To achieve maximum bonding efficiency, precise control of processing conditions is essential. Key factors include:
- Hydroxyl group concentration: A higher number of accessible hydroxyl groups on the filler surface improves surface modification.
- Water availability: Water is necessary for hydrolysis of alkoxysilyl groups into silanols, which then condense to form siloxane linkages.
- Surface tension alignment: The critical surface tension of the treated filler should match the polymer’s surface tension in its melt or uncured state.
Temperature management is equally crucial. For example, when using silylsulfonylazides in polyolefins, maintain temperatures above 150°C (302°F) to ensure the azide decomposes into reactive nitrene, enabling carbon-hydrogen bond insertion. The required silane amount depends on filler particle size:
- For particles smaller than 1 μm, use at least 1.5% silane.
- For particles larger than 100 μm, 0.1% or less is typically sufficient.
In moisture-cured systems, ensure the presence of a tin catalyst to facilitate crosslinking.
Disclaimer: This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
How to Select the Right Silane Coupling Agent
Selecting the right silane coupling agent involves aligning its chemistry with the filler and elastomer system you’re working with. The key lies in the organofunctional group – the "R" group attached to the silane – which must be compatible with your polymer matrix to ensure strong chemical bonds. For instance, epoxy-functional silanes pair well with epoxy resins, while amino-functional silanes are better suited for polyurethanes and nylons.
Matching Filler and Elastomer Chemistry
The filler’s surface chemistry plays a pivotal role in choosing the right silane. Silica, glass, and alumina, for example, have abundant hydroxyl groups on their surfaces, making them highly reactive to silane treatments. On the other hand, calcium carbonate and barytes are less reactive to standard silanes and require specialized options. Materials like carbon black and graphite, which lack surface hydroxyl groups, do not react with silanes at all [1].
Another important factor is the alkyl chain length between the silicon atom and the functional group. A three-carbon propyl linker is commonly used for its thermal stability. Longer chains, however, add flexibility, making them ideal for complex systems by allowing the functional group to better reach reactive sites. Shorter chains, while more rigid, are useful in applications where matching the modulus between the filler and matrix is critical [2].
For non-functional polymers like polyethylene, the focus shifts to surface tension matching. In these cases, aligning the critical surface tension of the treated filler with that of the polymer melt can optimize reinforcement, even when chemical bonding isn’t an option [2].
Testing Performance Metrics
When evaluating silane performance, focus on three main areas: mechanical strength, durability in harsh conditions, and filler dispersion quality. Mechanical tests like tensile strength, flexural strength, and impact resistance help determine if the silane is forming effective bonds. Scanning electron microscopy (SEM) can reveal how the material fractures – strong coupling typically results in the matrix or fibers breaking, rather than the interface separating [2].
Hydrolytic stability is another critical factor, especially for applications exposed to moisture or humidity. Standard silanes may degrade in such conditions, but dipodal silanes, which feature two silicon atoms, offer significantly greater resistance – up to 100,000 times more stable in hot, wet environments. This makes them ideal for marine or outdoor applications [2].
Filler dispersion can be assessed by measuring contact angles and comparing critical surface tension. When the treated filler’s critical surface tension matches the liquid resin, better dispersion and improved thixotropic properties result, enhancing performance in coatings and elastomers [2].
Comparing Common Silane Types
Different silane types excel in specific applications. Here’s a breakdown of common options:
| Silane Type | Best For | Key Selection Criteria | Application Constraints |
|---|---|---|---|
| APTES (Aminopropyltriethoxysilane) | Polyurethanes, nylons, acrylics, epoxy hardeners | Affordable and compatible with many polymer types | Requires blocking agents in single-component systems to avoid premature crosslinking |
| GPTMS (Glycidoxypropyltrimethoxysilane) | Epoxy resins, phenolics, polyimides | High thermal stability, ideal for high-temperature uses | Limited to epoxy-compatible systems |
| MPS/MPTMS (Methacryloxypropyltrimethoxysilane) | Unsaturated polyesters, vinyl esters, dental composites | Rapid addition in free-radical systems; better performance than vinyl silanes | Requires peroxide initiators for best results |
Particle size also affects silane application. Fillers smaller than 1 μm typically need at least 1.5% silane, while larger particles (over 100 μm) require as little as 0.1%. Using too little silane weakens bonding, while overusing it can create a layer of unreacted oligomers, reducing mechanical strength [1].
Disclaimer: This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Conclusion
Key Takeaways
Silane coupling agents create durable covalent bonds between inorganic fillers and organic elastomers, improving tensile and flexural strength, enhancing filler dispersion, and increasing resistance to moisture and heat [1]. The siloxane bonds (Si-O-Substrate) act as a barrier against water infiltration at the filler-matrix interface, preserving material integrity even in humid conditions [1].
Selecting the right silane depends on the chemistry of your elastomer. For example, amino-functional silanes pair well with polyurethanes and nylons, vinyl or mercapto silanes suit polyolefins and EPDM, while epoxy silanes are ideal for epoxy resins [1][2]. Dosage is also critical – smaller fillers typically require more silane [1][2]. Using too little can weaken bonds, while overuse may lead to unreacted oligomers that compromise mechanical strength [1].
Maintaining optimal reaction conditions is equally important. Factors like pH (4–5), moisture levels, and proper storage significantly influence bonding performance [1]. When integrated correctly, silane coupling agents improve elastomer durability and performance, making them indispensable in demanding industrial applications.
Sourcing Quality Silane Coupling Agents
The performance of silane coupling agents hinges on their chemical purity and structural integrity. Poor-quality silanes can self-condense, weakening the bond and reducing effectiveness [1]. Partnering with reliable suppliers ensures access to the precise chemical structures needed for specific filler-elastomer combinations, minimizing risks like premature polymerization or inconsistent results.
Allan Chemical Corporation supplies technical-grade and compendial-grade silane coupling agents with over 40 years of expertise in specialty chemicals. They offer just-in-time delivery, flexible batch sizes, and technical support for documentation and application guidance. Their strong relationships with vetted manufacturers ensure consistent quality, meeting the stringent demands of regulated industries that require precise chemical specifications.
Disclaimer: This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
FAQs
How do I choose the right silane for my polymer and filler?
To choose the right silane for your needs, consider these key factors:
- Substrate Surface Chemistry: Evaluate the presence of hydroxyl groups on your substrate. Trialkoxysilanes are often a good choice for achieving optimal surface coverage.
- Polymer or Filler Type: Align the silane’s functionality with your material. For example, acrylate silanes work well with acrylics, while vinyl silanes are suited for olefins.
- Environmental Stability: For conditions involving moisture or instability, adipodal silanes can provide better performance.
- Application Goals: Select silanes specifically designed to enhance adhesion, improve dispersion, or boost mechanical properties, depending on your desired outcome.
What causes silanes to underperform during mixing or curing?
Silanes may not perform as expected when hydrolysis and condensation reactions are incomplete. This typically happens when there isn’t enough moisture or when reaction conditions are not ideal. These shortcomings can prevent proper bonding and dispersion within the elastomer matrix. To ensure effective performance, it’s crucial to maintain the right reaction conditions.
How can I tell if my silane dose is too high or too low?
To determine if your silane dose is appropriate, pay close attention to the mechanical and processing properties of your composite. Using too much silane can negatively impact curing efficiency and filler dispersion. On the other hand, insufficient silane may result in weak interfacial bonding. These issues typically show up in how the filler interacts within the composite and in the material’s overall performance.





Comments are closed