Enzyme-responsive materials (ERMs) are specialized systems that release drugs in response to specific enzymes present in the body. These materials improve precision in drug delivery, targeting diseased tissues while reducing side effects. Here are the five key types of ERMs and their uses:
- Polymer-Based Redox Systems: Utilize redox reactions triggered by enzymes like Glutathione Reductase or Glucose Oxidase to release drugs, such as insulin for diabetes management.
- Protease-Cleavable Peptides: Use enzymes like MMPs or Cathepsins to release drugs in cancer or cardiovascular treatments.
- Enzyme-Responsive Hydrogels: Hydrogels react to enzymes such as MMPs or glycosidases for tissue engineering and localized therapies.
- Hyaluronidase & Cathepsin-Responsive Materials: Target cancer or bone disorders by degrading specific biological scaffolds.
- Carboxylesterase & P450 Systems: Trigger drug release in inflammatory diseases or liver-related conditions.
These materials rely on enzymatic activity for controlled drug release, offering targeted solutions for complex medical conditions.
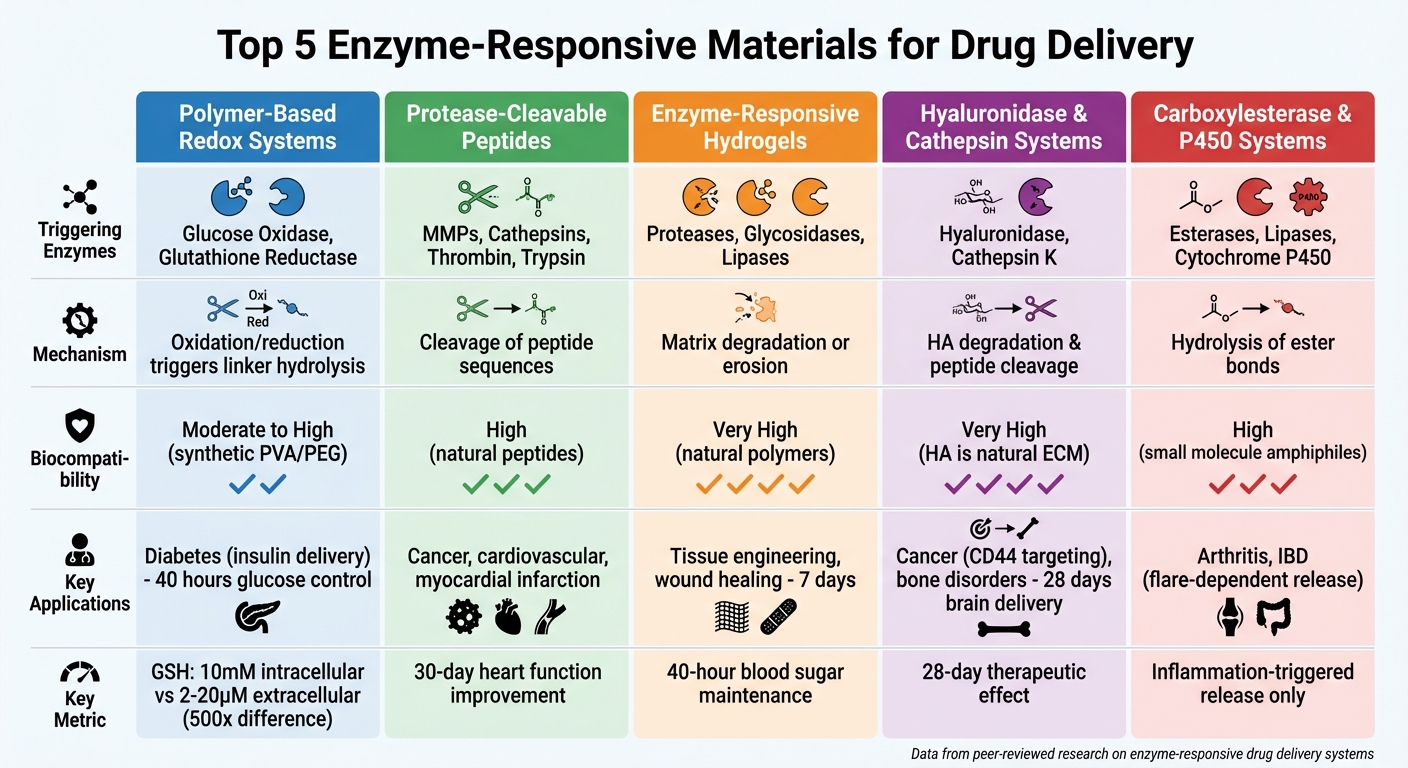
Comparison of 5 Enzyme-Responsive Drug Delivery Systems
How Smart Microcapsules Release Molecules on Command
sbb-itb-aa4586a
1. Polymer-Based Redox-Responsive Systems
Polymer-based redox-responsive systems use changes in oxidation and reduction states to control drug release. These systems rely on chemical linkages like disulfide bonds (-S-S-) or diselenide bonds (-Se-Se-), which break when exposed to specific reducing or oxidizing agents in the body. This mechanism ensures that drug release is activated only under the right biochemical conditions.
Triggering Enzyme(s)
Glutathione reductase plays a key role by maintaining high intracellular levels of GSH (glutathione). Using NADPH as a cofactor, it converts glutathione disulfide (GSSG) back into GSH. Inside cells, GSH levels reach about 10 mM – approximately 500 times higher than the 2–20 μM typically found in extracellular fluid [5]. Tumor tissues, in particular, show at least a fourfold increase in GSH concentration compared to normal tissues [5]. For oxidation-responsive systems, glucose oxidase (GOx) catalyzes the reaction of glucose into hydrogen peroxide (H₂O₂), which acts as the oxidative trigger. Catalase is often included alongside GOx to break down excess H₂O₂, fine-tuning the release process [2][3]. These enzymatic reactions drive the structural changes in the polymers.
Mechanism of Action
Reduction-responsive polymers degrade when GSH cleaves disulfide or diselenide linkages in the polymer’s backbone or cross-links. Diselenide bonds, which have a bond energy of about 172 kJ/mol, are highly sensitive to redox changes. For example, micelles with diselenide bonds can release their cargo within five hours at a GSH concentration of 0.01 mg/mL [5]. On the other hand, oxidation-responsive systems rely on GOx to produce H₂O₂, which either oxidizes thioether groups or cleaves aryl boronic ester linkages. This process triggers polymer degradation or transforms the material from hydrophobic to hydrophilic, enabling drug release [2][3].
Applications in Drug Delivery
One standout example is the poly(vinyl alcohol) (PVA) microneedle patch designed for insulin delivery. In this system, GOx oxidizes blood glucose to generate H₂O₂, which then triggers insulin release through H₂O₂-sensitive linkers. In diabetic mouse models, this innovative patch maintained normal blood sugar levels for 40 hours [3]. This demonstrates how redox-triggered systems can align precisely with therapeutic needs.
Biocompatibility and Specificity
These systems are highly compatible with biological environments, often using hydrogel networks that resemble natural tissues. By targeting specific redox conditions – such as the elevated GSH levels in tumor cells or increased H₂O₂ in inflamed tissues – they ensure that drug release happens exactly where it is needed. This approach reflects the broader strategy of enzyme-responsive drug delivery. Jinming Hu from the University of Science and Technology of China highlights the potential of these systems:
"The integration of enzyme-catalyzed reactions with responsive polymers can further broaden the design flexibility and scope of applications by endowing the latter with enhanced triggering specificity and selectivity." [6]
2. Protease-Cleavable Peptide Conjugates
Protease-cleavable peptide conjugates link drugs to peptide-based connectors that specific enzymes can break down. This design ensures that the drug stays inactive until the right protease triggers the release, avoiding the uncontrolled release often seen with simpler drug delivery systems.
By taking advantage of enzymes that are overproduced in certain diseases, these conjugates improve the precision of treatments for conditions like cancer, inflammation, and myocardial infarction.
Triggering Enzyme(s)
Certain enzymes are commonly used to activate these systems:
- Matrix metalloproteinases (MMPs): MMP-2 and MMP-9 are frequently used because they are highly active during disease states. They recognize sequences like PLGLAG and PVGLIG [2][3].
- Cathepsins: Cathepsin K and Cathepsin B are effective for bone-related treatments and intracellular delivery due to their activity in lysosomes [1][2].
- Thrombin: This enzyme plays a role in cardiovascular therapies, thanks to its function in the coagulation process [3].
- Trypsin: Found in the small intestine, this enzyme works well for gastrointestinal drug delivery when paired with arginine- and lysine-rich linkers [3].
These enzymes not only indicate the presence of disease but also serve as triggers to release the drug at the right time and location.
Mechanism of Action
Protease-cleavable peptides work through several mechanisms:
- Matrix degradation: Enzyme activity breaks down cross-linkers, disassembling the matrix.
- Targeted linker cleavage: Drugs are released in a controlled manner, maintaining their intended structure.
- Morphological transformation: Enzyme cleavage can change cyclic peptides into linear forms, which then self-assemble into hydrogels [2][3].
Applications in Drug Delivery
A notable example comes from 2019, when Fan et al. created a dual-function hydrogel sensitive to MMP-2/9. This hydrogel delivered basic fibroblast growth factor (bFGF) to treat myocardial infarction. The system used a collagen-based hydrogel modified with glutathione (GSH) and a recombinant protein containing the MMP-sensitive sequence PLGLAG. In a rat model, the hydrogel improved heart function and reduced ventricular remodeling over 30 days, outperforming simpler encapsulation, which led to a rapid five-day release [2][3].
Another example is the MMP-2-responsive hydrogel developed by Cai et al. for delivering small interfering RNAs (siRNA) to injury sites. After 21 days, this system significantly reduced peritendinous adhesions compared to controls [2].
These examples highlight how protease-cleavable systems can provide localized, timed drug delivery for better therapeutic outcomes.
Biocompatibility and Specificity
Peptide-based systems are especially appealing because they are made from natural amino acids, which makes them biodegradable and generally safe for surrounding tissues [7][2]. Their high specificity ensures that treatments are delivered precisely where and when needed. For instance, MMP-2 and MMP-9 are upregulated during the inflammatory phase of a heart attack, allowing targeted therapies. Additionally, the use of cleavable linkers ensures the released drug maintains a consistent molecular structure, reducing the risk of unwanted side effects or interference with drug activity [3].
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
3. Enzyme-Responsive Hydrogels
Hydrogels provide a versatile and body-friendly platform for enzyme-triggered drug delivery, standing apart from polymer and peptide conjugates.
Enzyme-responsive hydrogels are three-dimensional polymer networks capable of absorbing water and reacting to specific biological enzymes. These materials mimic many features of living tissues, making them a natural fit for controlled drug delivery. What sets them apart is their ability to release therapeutics on demand, triggered by elevated enzyme levels, offering a level of precision not seen in traditional systems.
Triggering Enzyme(s)
Several enzyme classes can activate hydrogel-based drug delivery systems. Proteases are the most commonly used triggers, including enzymes like MMP‑1, MMP‑2, MMP‑7, MMP‑8, MMP‑9, and MMP‑13, which are associated with conditions such as inflammation, cancer, and cardiovascular diseases. Other proteases like trypsin, chymotrypsin, elastase, thrombin, and Cathepsin K also play a role. Hydrolases, such as lipases and glycosidases (e.g., hyaluronidase, β‑galactosidase, and β‑glucuronidase), break down specific bonds within the hydrogel matrix. Oxidoreductases, including glucose oxidase (GOx), horseradish peroxidase (HRP), and catalase, are particularly useful in glucose-responsive insulin delivery systems.
Mechanism of Action
Hydrogels release drugs through three main mechanisms:
- Matrix degradation: Enzymes break down the hydrogel’s polymer network, freeing the drugs.
- Covalent linker cleavage: Enzymes sever specific chemical bonds, ensuring precise drug release.
- Physical state transformation: Enzyme activity alters the hydrogel’s structure, making it easier for drugs to diffuse.
These mechanisms allow hydrogels to adapt to a wide range of therapeutic needs.
"Enzyme-responsive materials are highly advantageous as endogenous enzyme expression can be used in controlling the release rate of various therapeutics for a wide range of applications."
- Rachel L. Minehan and Mark P. Del Borgo, Monash University [3]
Applications in Drug Delivery
In 2018, Wang J. and colleagues developed a glucose-responsive insulin delivery system using a poly(vinyl alcohol) (PVA) hydrogel. Glucose oxidase (GOx) converted glucose into hydrogen peroxide (H₂O₂), which degraded H₂O₂-sensitive linkers, releasing insulin. In diabetic mice, this system maintained normal blood sugar levels for 40 hours and caused fewer hypoglycemic episodes compared to standard injections [3].
For wound healing, Gao et al. created a self-assembling peptide hydrogel (Nap‑FFGGG) linked to a nitric oxide (NO) donor. The release, triggered by β‑galactosidase, led to increased blood vessel formation and faster healing in mouse skin excision models over a seven-day period [3].
Biocompatibility and Specificity
These hydrogels closely resemble natural tissues, reducing inflammation while ensuring precise drug delivery. Their water-rich, flexible structures mimic soft tissues and the extracellular matrix (ECM), which contributes to their high biocompatibility. This design minimizes inflammation when the hydrogels are injected or implanted. Over 30 injectable hydrogel products have already been approved by both the European Medicines Agency and the US FDA [3].
The specificity of enzyme activity ensures that drug release happens mainly at target sites, reducing the risk of side effects. Since enzymatic reactions occur under mild physiological conditions – neutral pH and body temperature – the bioactivity of sensitive drugs like proteins and growth factors remains intact. Natural polymers such as hyaluronic acid and collagen further enhance the system, offering biodegradability that aligns with tissue regeneration [8].
For those developing these advanced delivery systems, specialty providers like Allan Chemical Corporation offer reliable sources of high-quality polymers and cross-linking agents.
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
4. Hyaluronidase and Cathepsin-Responsive Materials
This section examines how materials responsive to hyaluronidase and cathepsins are utilized in targeted drug delivery systems. These enzymes are particularly effective in addressing conditions like cancer, inflammation, and bone resorption by leveraging their biological activity to target diseased tissues.
Triggering Enzyme(s)
Hyaluronidase (HAase) is a group of enzymes responsible for breaking down hyaluronic acid (HA), a key component of the extracellular matrix (ECM) [9]. The human genome encodes six hyaluronidase types – HYAL1, HYAL2, HYAL3, HYAL4, PH-20, and HYALP1 [9]. Their activity is influenced by pH: HYAL 1–4 function optimally in acidic environments (pH 3.0–4.0), while PH-20 remains active at neutral pH (pH 5.0–8.0) [9].
Cathepsins are proteases, including cysteine cathepsins, that are primarily found in lysosomes. These enzymes are often overexpressed in cancerous and inflamed tissues [1]. Cathepsin K, in particular, is involved in bone resorption and plays a critical role in bone remodeling [1].
Mechanism of Action
Hyaluronidase-responsive systems utilize HA as a scaffold or linker. The enzyme degrades the HA matrix by hydrolyzing glycosidic bonds, which facilitates drug release [9]. The resulting HA fragments can be absorbed by cells through CD44 receptors, which are commonly overexpressed on cancer cells [3].
Cathepsin-responsive materials are designed with peptide sequences that are specifically cleaved by these enzymes [1]. For example, Cathepsin K-sensitive linkers degrade during bone resorption, aligning material breakdown with the natural bone remodeling process [1].
"Hyaluronidase-mediated drug delivery systems represent a paradigm shift in overcoming biological barriers, enhancing bioavailability, and optimizing therapeutic outcomes through ECM remodeling."
- Molecular Biomedicine [9]
Applications in Drug Delivery
Research has demonstrated the potential of these enzyme-responsive systems in various therapeutic applications:
- Hyaluronic Acid and Methylcellulose Hydrogel: Tuladhar and colleagues at the University of Toronto developed a hydrogel responsive to hyaluronidase. It delivered cyclosporine and erythropoietin to the brain in a rat stroke model, reducing infarct size and aiding recovery [3].
- Cathepsin K-Sensitive Hydrogels: Researchers at Clemson University created polyethylene glycol (PEG) hydrogels that degrade specifically in response to bone resorption, offering targeted drug delivery for bone-related conditions [1].
- Dual-Phase Stroke Treatment: At Sun Yat-sen University, a hyaluronic acid hydrogel was designed to address both inflammatory and neuroregeneration phases of stroke. The scaffold degraded in response to hyaluronidase, releasing VEGF and BIO-loaded nanoparticles, improving outcomes in a mouse stroke model [3].
These examples highlight how enzyme-specific targeting can improve therapeutic precision and effectiveness.
Biocompatibility and Specificity
Materials based on hyaluronic acid are naturally biocompatible and biodegradable, as HA is a polysaccharide found in the ECM. Its soft tissue-like properties also minimize inflammation and promote healing [3]. Specificity is achieved by targeting enzymes that are upregulated in diseased states, such as hyaluronidase in inflamed tissues and Cathepsin K in bone remodeling sites [3][1].
Cathepsin-responsive systems rely on programmable peptide sequences that degrade only under specific proteolytic conditions, ensuring precise targeting [1]. For researchers developing these systems, specialty suppliers like Allan Chemical Corporation provide high-purity polymers and peptides essential for formulation.
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
5. Carboxylesterase and Cytochrome P450-Responsive Systems
Enzyme-responsive systems using carboxylesterase and cytochrome P450 have added new dimensions to targeted drug delivery. Unlike approaches involving proteases or hydrogels, these systems rely on hydrolysis and redox reactions to trigger precise drug release. This makes them particularly useful for targeting tissues in conditions such as inflammation, gastrointestinal disorders, and liver-related diseases.
Triggering Enzyme(s)
Carboxylesterases are part of the hydrolase enzyme family, breaking down ester bonds through hydrolysis[3]. Found in most tissues, these enzymes are often upregulated in diseases like cancer and inflammation, making them valuable tools for targeted therapies.
Cytochrome P450 enzymes, on the other hand, belong to the oxidoreductase class. These enzymes facilitate oxidation-reduction reactions by transferring electrons between molecules. Their high concentration in the liver makes them ideal for drug delivery systems aimed at hepatic tissues[4].
Mechanism of Action
Carboxylesterase-responsive systems are designed to release drugs through the hydrolysis of ester bonds. These bonds may be part of the polymer backbone or link the drug to a delivery scaffold[3].
Cytochrome P450-responsive systems work by catalyzing redox reactions that break specific chemical bonds. This allows for controlled drug release, particularly in liver tissues[4][10]. These mechanisms offer precise control over drug delivery, enabling tailored therapeutic applications.
"Esterases can be advantageous in cleaving biomaterials as they are expressed in most tissues and can be upregulated in disease." – Frontiers in Biomaterials Science[3]
Applications in Drug Delivery
Carboxylesterase and cytochrome P450-responsive systems have been applied to a range of diseases:
- Inflammatory Bowel Disease (IBD): Zhang and colleagues developed a hydrogel using ascorbyl palmitate to trigger dexamethasone release. This system successfully reduced inflammation in a mouse model of colitis[3].
- Arthritis: Joshi’s team created a triglycerol monostearate-based hydrogel with ester bonds in its structure. This material degraded in response to synovial fluid from arthritic patients, releasing corticosteroids and reducing inflammation in a mouse model. The "flare-dependent" release mechanism ensured drug delivery occurred only during periods of heightened inflammation[3].
- Neurological Disorders: Kulkarni’s group used esterase-sensitive linkers to deliver brain-derived neurotrophic factor (BDNF) mimetics. This approach enhanced neuron survival and supported normal function in murine models. Therapeutic levels were maintained for up to 28 days with minimal systemic exposure[3].
These examples highlight how enzyme-specific systems improve precision and effectiveness in drug delivery.
Biocompatibility and Specificity
One of the strengths of these systems is their reliance on naturally occurring enzymes, eliminating the need for external triggers like light, ultrasound, or magnetic fields. They operate under mild physiological conditions, such as normal body temperature and pH, reducing the need for invasive activation methods[10].
Specificity comes from the natural upregulation of carboxylesterases in diseased tissues, which enhances targeting. Additionally, the ester bonds are designed to respond exclusively to specific enzymes, ensuring precise drug release. It’s also critical to ensure that any byproducts from the cleavage process are non-toxic and can be easily cleared from the body.
For researchers working on these systems, companies like Allan Chemical Corporation provide high-purity materials essential for creating enzyme-sensitive linkers and scaffolds.
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
Comparison Table
Five distinct material systems are frequently utilized for targeted drug delivery, each offering specific benefits based on their mechanisms and applications. The table below highlights essential parameters, including triggering enzymes, release mechanisms, biocompatibility, and relevant uses.
Polymer-based redox-responsive systems depend on oxidoreductases like glucose oxidase to trigger reactions, making them effective for insulin delivery in diabetes[3]. Protease-cleavable peptide conjugates leverage enzymes such as matrix metalloproteinases (MMPs) and thrombin for applications in cardiovascular health and cancer therapy[3][8]. Enzyme-responsive hydrogels utilize hydrolases to degrade their matrix, ideal for tissue engineering and localized drug delivery. Hyaluronidase and cathepsin-responsive materials target cancer cells through CD44 receptor binding and hyaluronic acid (HA) degradation, with some systems maintaining therapeutic effects in brain tissue for up to 28 days[3]. Lastly, carboxylesterase and cytochrome P450-responsive systems release drugs during inflammatory events, making them suitable for conditions like arthritis and inflammatory bowel disease (IBD). This table consolidates the material-specific insights discussed earlier.
| Material System | Triggering Enzymes | Primary Mechanism | Biocompatibility | Key Applications |
|---|---|---|---|---|
| Polymer-Based Redox-Responsive | Oxidoreductases (Glucose Oxidase, Catalase) | Enzymatic oxidation/reduction triggers linker hydrolysis or matrix swelling[3][8] | Moderate to High (synthetic PVA or PEG) | Diabetes (insulin delivery), biosensing[3][8] |
| Protease-Cleavable Peptide Conjugates | Proteases (MMPs, Thrombin, Trypsin) | Cleavage of specific peptide sequences tethering the drug or cross-linking the matrix[3][8] | High (natural peptide sequences) | Myocardial infarction, clot prevention, cancer[3][8] |
| Enzyme-Responsive Hydrogels | Various (Proteases, Glycosidases, Lipases) | Bulk or surface erosion of the hydrogel matrix to release encapsulated cargo[3] | Very High (natural polymers like alginate, collagen) | Tissue engineering, localized drug delivery[1][8] |
| Hyaluronidase & Cathepsin-Responsive | Hyaluronidase, Cathepsin K | Degradation of hyaluronic acid backbone or cleavage of cathepsin-sensitive peptide linkers[1][3] | Very High (HA is a natural ECM component) | Cancer (CD44 targeting), bone resorption[3] |
| Carboxylesterase & Cytochrome P450-Responsive | Esterases, Lipases, Cytochrome P450 | Hydrolysis of ester bonds in the material backbone or specific drug-scaffold linkers[3] | High (small molecule amphiphiles) | Arthritis, Inflammatory Bowel Disease[3] |
Natural polymer-based systems, such as those utilizing hyaluronic acid and collagen, exhibit the highest biocompatibility as they closely resemble the extracellular matrix found in living tissues[8]. For sourcing high-purity chemical components, Allan Chemical Corporation offers technical-grade and compendial-grade solutions tailored for enzyme-sensitive linkers and scaffolds. This summary underscores the strategic selection of enzyme-responsive systems for specific drug delivery needs.
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
Conclusion
Enzyme-responsive materials are transforming targeted drug delivery by offering precision tailored to specific therapeutic needs. The five material types covered – polymer-based redox-responsive systems, protease-cleavable peptide conjugates, enzyme-responsive hydrogels, hyaluronidase-responsive materials, and esterase-responsive systems – each bring unique solutions by responding to distinct enzymatic environments.
The success of these systems relies on carefully matching the material’s enzyme trigger to the enzymatic profile of the target tissue. For example, conditions like arthritic flare-ups, characterized by increased lipase activity, are well-suited for esterase-responsive systems that release corticosteroids only during active disease phases. Similarly, cancer treatments benefit from hyaluronidase-responsive materials that degrade within tumor microenvironments, ensuring the drug is delivered exactly where it is needed.
Drug release mechanisms also play a critical role in material selection. Systems that use covalent enzyme-sensitive linkers tend to provide more predictable release patterns while preserving drug potency. This level of control is particularly important in challenging applications like inflammatory bowel disease. In such cases, trypsin-degradable nanogels must survive the acidic environment of the stomach to release their therapeutic payload precisely in the intestines.
To support the development of these advanced systems, researchers need access to high-purity chemical components. Allan Chemical Corporation offers technical- and compendial-grade materials (USP, FCC, ACS, NF) ideal for creating enzyme-sensitive linkers and scaffolds. With more than 40 years of experience, their reliable delivery and strong supplier relationships are valuable assets for innovators working on next-generation drug delivery platforms.
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
FAQs
How do I choose the right enzyme trigger for a specific disease target?
When choosing an enzyme trigger, focus on the enzyme’s activity profile and its specific connection to the disease. Enzyme-responsive biomaterials are designed to interact with enzymes that are overproduced in certain medical conditions, allowing for targeted drug release. For instance, hydrogels can be tailored to react to enzymes associated with cardiovascular or musculoskeletal disorders. By identifying the active enzymes present in the disease environment and pairing them with biomaterials engineered to respond selectively, you can achieve precise and controlled drug delivery.
What safety risks are associated with enzyme-triggered byproducts like hydrogen peroxide?
Enzyme-triggered byproducts, such as Hydrogen Peroxide, can introduce safety concerns, including tissue damage and oxidative stress. Without effective management, these issues may escalate, potentially causing cellular injury. Implementing robust control measures is crucial to minimize these risks and ensure the safe use of such systems in drug delivery applications.
How do these materials prevent premature drug release in the body?
These materials are crafted to interact specifically with enzymes present at targeted locations within the body. This precise targeting allows for controlled drug release, reducing the chances of premature activation and enhancing the accuracy and efficiency of the delivery system.





Comments are closed