Ultraviolet (UV) light is a non-thermal method for preserving food, targeting harmful microorganisms while keeping flavor and nutritional quality intact. In the U.S., the FDA regulates UV treatment for food safety under 21 CFR 179.39, which primarily applies to reducing pathogens in juice, water, and food surfaces. UV-C light, particularly at 253.7 nm, is effective in disrupting microbial DNA. Facilities must comply with HACCP and FSMA standards, ensuring proper validation, monitoring, and record-keeping for UV processes.
In the EU, UV-treated foods are classified as "novel foods" under Regulation (EC) No 258/97, requiring a broader safety assessment, including nutritional impacts. Manufacturers exporting to the EU must meet stricter compositional requirements compared to U.S. standards.
UV-C technology offers precise control over microbial contamination, but compliance with FDA, USDA, and FSMA guidelines is mandatory to ensure food safety and market access. Regular validation and adherence to these regulations are critical for maintaining product integrity and public trust.
Ultraviolet light could extend shelf life of food and reduce waste
sbb-itb-aa4586a
21 CFR 179.39: FDA Standards for UV Radiation in Food Treatment
21 CFR 179.39, titled "Ultraviolet radiation for the processing and treatment of food," is the key FDA regulation overseeing UV light use as a food additive [1][2]. Found under Subpart B (Radiation and Radiation Sources), it authorizes UV irradiation for three specific purposes: reducing surface microorganisms on food and food products, sterilizing potable water used in food production, and minimizing human pathogens in juice products [1][4]. These guidelines establish the safety protocols required for UV systems in food processing.
The regulation mandates the use of low-pressure mercury lamps that emit at least 90% of their radiation at a wavelength of 253.7 nanometers (nm) [3][4]. This UV-C wavelength is particularly effective in killing germs, making it essential for achieving the desired germicidal effect during food treatment.
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
USDA and HACCP Guidelines for UV-C in Food Safety
In addition to the FDA’s 21 CFR 179.39 guidelines, the USDA and HACCP standards also highlight the importance of UV-C in maintaining food safety. The USDA Food Safety and Inspection Service (FSIS) recognizes UV-C as a safe antimicrobial method for treating the surfaces of meat and poultry. This physical treatment is often used alongside natural antimicrobials for food shelf-life to provide a multi-hurdle approach to preservation.
UV-C operates at 254 nm, which is its most effective wavelength for disrupting microbial DNA and ensuring safety.
Within HACCP (Hazard Analysis and Critical Control Points) systems, UV-C is identified as a Critical Control Point (CCP), making it an integral part of food safety management plans.
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
FDA Food Safety Modernization Act (FSMA) and UV-C Applications in Produce
The FDA Food Safety Modernization Act (FSMA) builds on USDA and HACCP guidelines by emphasizing prevention over response when it comes to food safety. For facilities using UV-C treatments in produce processing, FSMA’s Preventive Controls Rule requires a comprehensive food safety plan. This plan must identify key biological hazards and outline specific preventive controls. Since UV-C treatments are considered process controls, facilities need to define critical parameters like exposure time and lamp intensity to ensure the process is effective [5].
FSMA also mandates scientific validation. According to the FDA:
Verification activities include scientifically validating process preventive controls to ensure that the control measure is capable of effectively controlling an identified hazard [5].
This means facilities must support their UV-C sanitation processes with peer-reviewed studies or third-party validation data. These studies must demonstrate that the specific UV-C dosage effectively reduces pathogens on the produce being treated [6].
In addition to validation, ongoing monitoring and detailed record-keeping are essential to maintain UV-C treatment effectiveness. Facilities are required to continuously track parameters like exposure time, lamp intensity, and application methods. Supervisors must review and log these readings for at least two years. If any parameters fall outside the established limits, corrective actions must be taken immediately, and the safety of the produce must be reassessed before it is released [5]. Regular calibration of UV-C sensors and lamps is also necessary to ensure accurate data, especially during FSMA inspections [5].
Compliance deadlines for the Preventive Controls Rule varied by business size: large businesses had to comply by September 2016, small businesses by September 2017, and very small businesses by September 2018 [5]. Together, these FSMA requirements and existing FDA and USDA guidelines create a cohesive framework for ensuring the safe use of UV-C in food preservation.
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
EFSA Novel Food Approvals for UV-Treated Foods
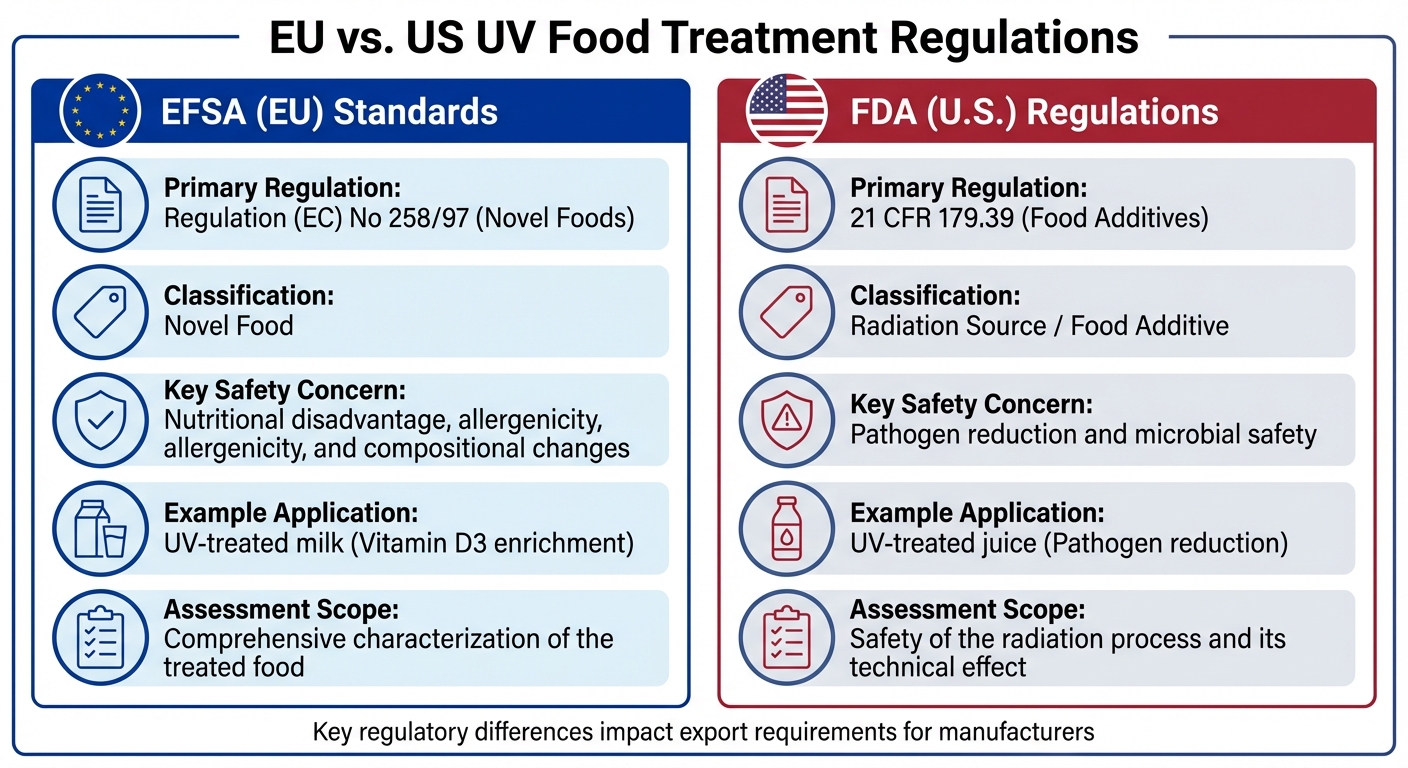
EU vs US UV Food Treatment Regulations Comparison
In the European Union, the focus on UV-treated foods goes beyond microbial safety, emphasizing the nutritional and compositional changes that may occur during processing. While the U.S. Food and Drug Administration (FDA) regulates UV treatment under 21 CFR 179.39 as a food additive primarily for pathogen reduction, the EU classifies these products as "novel foods" under Regulation (EC) No 258/97. This classification requires a detailed safety assessment to ensure consumer safety, nutritional adequacy, and minimal allergenicity risks [8]. The EU’s approach adds an additional layer of scrutiny compared to the U.S., prioritizing the broader impact of UV treatment on food composition.
EFSA’s evaluations have highlighted specific findings related to UV-treated foods. For example, a 2015 EFSA opinion showed that UV treatment converts ergosterol in yeast to vitamin D2 in bread, resulting in 0.75–3 μg of vitamin D2 per 100 g. Importantly, the reaction products formed during UV treatment were found to be at lower levels than those produced during standard baking processes. As noted by the EFSA NDA Panel:
Under certain conditions, UV treatment may result in reactions of biomolecules. However, the levels of potential reaction products that may be formed under the employed conditions are low compared with the reactions induced by the baking process [7].
EFSA also places limits on vitamin D levels to ensure they remain within safe thresholds. For instance, in its 2016 opinion on UV-treated milk, EFSA assessed a process applied after pasteurization that both extended shelf life and increased vitamin D3 concentrations. The panel concluded that the product was safe for the general population but excluded infants under one year due to the risk of excessive vitamin D intake [8]. Additionally, EFSA mandates that UV-treated foods must not pose nutritional disadvantages or increased allergenicity compared to conventional counterparts.
| Feature | EFSA (EU) Standards | FDA (U.S.) Regulations |
|---|---|---|
| Primary Regulation | Regulation (EC) No 258/97 (Novel Foods) | 21 CFR 179.39 (Food Additives) |
| Classification | Novel Food | Radiation Source / Food Additive |
| Key Safety Concern | Nutritional disadvantage, allergenicity, and compositional changes | Pathogen reduction and microbial safety |
| Example Application | UV-treated milk (Vitamin D3 enrichment) | UV-treated juice (Pathogen reduction) |
| Assessment Scope | Comprehensive characterization of the treated food | Safety of the radiation process and its technical effect |
These regulatory differences create unique challenges for manufacturers exporting UV-treated foods to the EU. Producers must provide detailed compositional data to demonstrate that nutrient increases and any byproducts remain within safe limits. While U.S. regulations focus heavily on microbial safety, EU standards demand a broader analysis, including the nutritional and compositional impacts of UV treatment. For manufacturers, adapting to these distinct regulatory frameworks is essential for successful market entry.
This content is for informational purposes only. Consult official regulations and qualified professionals before making formulation decisions.
UV Dosage Efficacy on Foodborne Pathogens
UV-C Dosage and Pathogen Control Studies
Recent studies have analyzed the effectiveness of UV-C light in reducing microbial contamination on food surfaces, adhering to the requirements of 21 CFR 179.39. For instance, experiments on strawberries demonstrated that mercury lamps emitting UV-C at 254 nm achieved a 2.75 log reduction for E. coli and a 2.63 log reduction for L. monocytogenes at a dose of 477 mJ/cm². Comparatively, UV-C LED arrays operating between 265–280 nm resulted in 2.39 and 2.15 log reductions for the same pathogens at a higher dose of 589.5 mJ/cm²[9]. The uneven surface of strawberry skin often leads to shadowing, allowing bacteria to persist in protected areas. To mitigate this, processors are advised to rotate the produce during treatment to ensure uniform exposure[9].
Table: UV-C Dose Efficacy Comparison
| UV-C Source | Pathogen | Food Type | UV Dose (mJ/cm²) | Log Reduction (CFU/g) | Study Reference |
|---|---|---|---|---|---|
| Mercury Lamp (254 nm) | E. coli | Strawberry | 477.0 | 2.75 | [9] |
| Mercury Lamp (254 nm) | L. monocytogenes | Strawberry | 477.0 | 2.63 | [9] |
| UV-C LED (All ON) | E. coli | Strawberry | 589.5 | 2.39 | [9] |
| UV-C LED (All ON) | L. monocytogenes | Strawberry | 589.5 | 2.15 | [9] |
| UV-C LED (Diagonal) | E. coli | Strawberry | 427.5 | 1.54 | [9] |
| UV-C LED (Top) | L. monocytogenes | Strawberry | 162.5 | 1.70 | [9] |
Weibull modeling, a statistical approach for predicting pathogen survival, has proven effective for complex surfaces like strawberries by accounting for non-linear inactivation patterns. This supports compliance with FSMA and HACCP requirements[9]. However, it’s important to note that microorganisms can repair UV-C-induced damage through photoreactivation or dark repair if storage conditions are inadequate[10]. These findings emphasize the importance of precise UV-C dosing and proper storage protocols to maintain food safety.
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Conclusion
UV-C food preservation in the United States is governed by a detailed regulatory framework aimed at safeguarding public health while supporting advanced pathogen reduction methods. The cornerstone of this framework is 21 CFR 179.39, which regulates the use of ultraviolet radiation in food processing. This regulation treats UV-C as a food additive, requiring proper safety validation – a process formalized through a petition approved for California Day-Fresh Foods, Inc. (FAP 9M4676) [1].
Regulatory compliance is critical for food processors utilizing UV-C technology. They must adhere to the specifications outlined in 21 CFR 179.39, maintain accurate records, and validate their processes in line with USDA HACCP and FSMA standards. Non-compliance can lead to products being deemed adulterated (under 21 U.S.C. 342) or misbranded (under 21 U.S.C. 343) [2].
Beyond UV-C, 21 CFR Part 179 also covers other radiation-based treatments, such as pulsed light (21 CFR 179.41). To remain compliant, manufacturers should regularly review the FDA’s Food Ingredient and Packaging Inventories for updates on approved applications and regulatory changes affecting UV-C technologies.
Validated UV-C processes not only enhance pathogen reduction but also strengthen consumer confidence. By meeting regulatory standards, manufacturers secure legal market access and maintain trust in UV-treated food products. As the industry continues to advance, strict compliance with these guidelines is essential for both public safety and the sustained credibility of UV-C applications in food preservation.
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
FAQs
Does 21 CFR 179.39 apply to UV-C LEDs or only mercury lamps?
The regulation 21 CFR 179.39 outlines the use of UV light in food preservation. However, it does not clearly state whether it applies only to mercury lamps or also includes UV-C LEDs. The language in the regulation broadly covers UV irradiation technologies without explicitly mentioning or excluding UV-C LEDs.
What UV-C process data must be monitored and kept for FSMA compliance?
To meet FSMA requirements, it’s essential to track and document critical UV-C process data. This includes UV dose, exposure time, and equipment calibration status. Keeping accurate records of these parameters is crucial for ensuring proper microbial inactivation and compliance with regulations such as 21 CFR 179.39, as well as adhering to industry guidance on UV food processing.
What extra testing is needed to sell UV-treated foods in the EU?
To sell UV-treated foods within the European Union, businesses must navigate additional testing requirements, including detailed safety assessments and strict compliance with EU novel food regulations. This process involves following specific legislation and undergoing rigorous safety evaluations, such as those carried out by the European Food Safety Authority (EFSA). These measures ensure that the food complies with all necessary safety and regulatory standards.





Comments are closed