Navigating cosmetic regulations for antioxidants is crucial to ensuring product safety and market approval. Regulatory frameworks vary by region, with the U.S., EU, and Asia-Pacific enforcing distinct rules on ingredient usage, labeling, and safety documentation. Key updates include the EU’s expanded allergen list (effective July 2026) and stricter U.S. FDA reporting requirements under MoCRA.
Key Takeaways:
- Ingredient Restrictions: Antioxidants like Vitamin A derivatives have concentration limits (e.g., EU Annex III).
- Labeling Rules: Use INCI names and disclose allergens per regional laws.
- Documentation: Maintain safety records (e.g., CPSR in the EU, 6-year retention under MoCRA).
- Testing: Validate claims with robust safety and efficacy tests.
Staying compliant means working closely with suppliers, keeping up with regulatory changes, and ensuring proper safety testing. Always consult official regulations and experts before product formulation or sourcing decisions.
FDA Grand Rounds – Postmarket Regulation of Cosmetic Products: The Who, What, When, Why, and How
sbb-itb-aa4586a
Major Regulatory Frameworks for Cosmetic Antioxidants
Asia-Pacific Cosmetic Antioxidant Regulations: China, South Korea, and Japan Comparison
Regulations governing cosmetic antioxidants differ across regions, with each market setting its own standards for safety, ingredient approval, and labeling. Here’s an overview of the key frameworks in the United States, the European Union, and the Asia-Pacific.
United States: FDA Requirements
The Modernization of Cosmetics Regulation Act of 2022 (MoCRA) significantly expanded the FDA’s oversight of cosmetics, including products with antioxidants. Manufacturers are now required to maintain MoCRA safety substantiation records for their products and ensure compliance with MoCRA’s provisions. This includes registering facilities, listing marketed products and ingredients, and labeling antioxidants with their common or usual names as per 21 CFR Part 701.
If marketing claims for antioxidants imply they can "affect the structure or any function of the body" – such as claims about "revitalizing cells" or "treating" conditions – the product risks being classified as a drug. Such reclassification could lead to regulatory action. To avoid this, brands must carefully document product safety, review claims, and ensure compliance with FDA guidelines, including sustainable ingredient sourcing where applicable [3][5].
European Union: Cosmetics Regulation 1223/2009
The EU’s Cosmetics Regulation (EC) No 1223/2009 mandates that all cosmetic products, including those with antioxidants, must be safe for consumers under normal use. The Responsible Person for each product must compile a Cosmetic Product Safety Report (CPSR) and maintain it in a Product Information File (PIF) for at least 10 years after the last batch is sold. This report should include data supporting any claimed benefits, such as antioxidant effectiveness.
Specific antioxidants are regulated under the Regulation’s Annexes. For example, Butylated Hydroxytoluene (BHT) is listed under Annex III (Entry 324), while Vitamin A derivatives like Retinol and Retinyl Palmitate are also limited (Annex III, Entry 375). Brands should consult the European Commission‘s CosIng database to confirm ingredient restrictions, allowable concentrations, and required INCI names.
Labeling requirements dictate that antioxidants appear in descending order by weight, though ingredients under 1% can be listed in any order after higher-concentration components. If an antioxidant is a nanomaterial, it must be labeled with "nano" in brackets following the ingredient name [6][7][8].
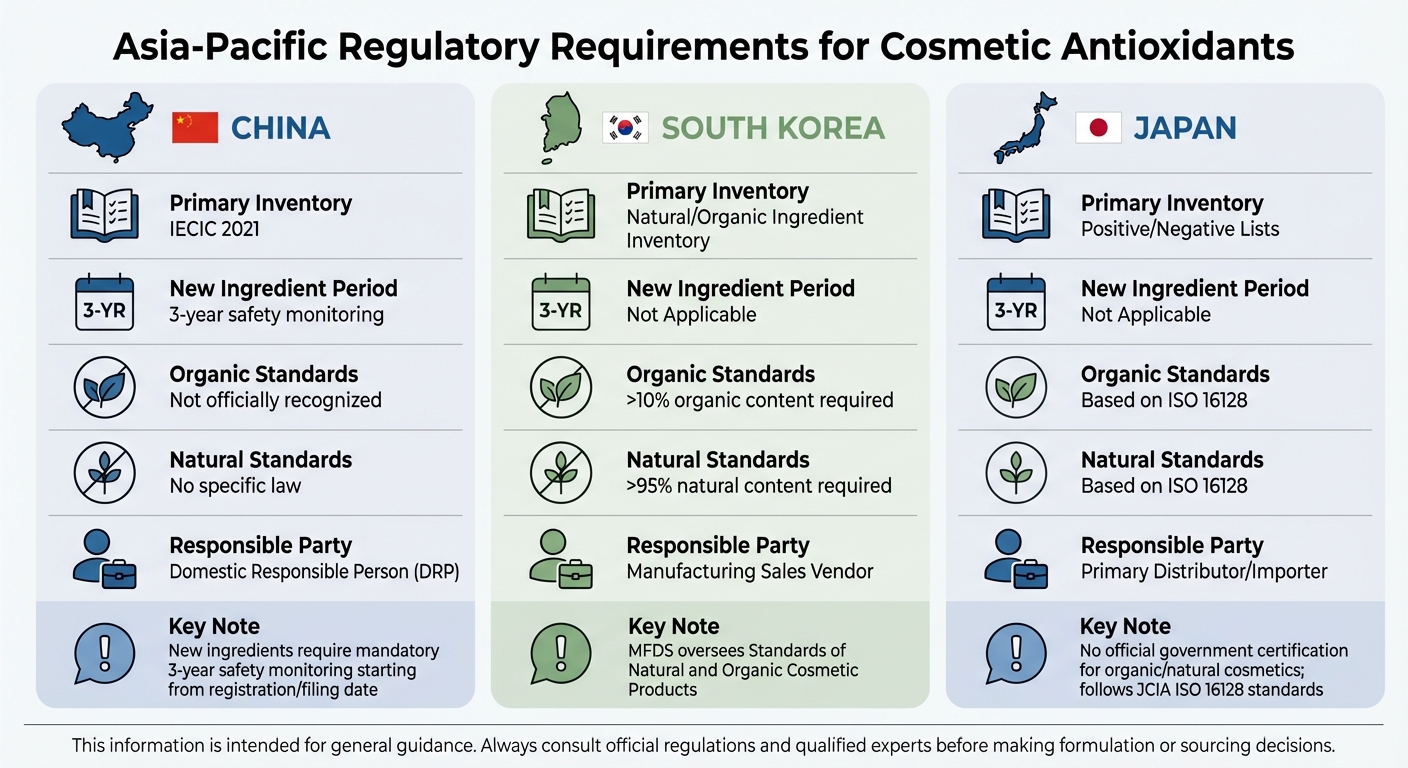
Asia-Pacific and Other Markets
Regulations in Asia-Pacific markets bring additional complexities to antioxidant compliance.
In China, the National Medical Products Administration (NMPA) requires all cosmetic ingredients to be listed in the Inventory of Existing Cosmetic Ingredients (IECIC 2021). Antioxidants not included in this inventory are treated as new ingredients and must undergo a mandatory three-year safety monitoring period after registration or filing. Article 19 of the Provisions for Registration and Filing of Cosmetics states:
The period of safety monitoring is 3 years, starting from the date when cosmetics using new cosmetic ingredients for the first time are registered or filed [11].
New ingredients are classified as high-risk (requiring registration) or non–high-risk (requiring filing), with most antioxidants falling into the latter category unless they have whitening properties. High-risk ingredient reviews typically take 90 working days. Foreign companies must appoint a Domestic Responsible Person (DRP) to handle registration and safety reporting in China.
In South Korea, the Ministry of Food and Drug Safety (MFDS) oversees the Standards of Natural and Organic Cosmetic Products. For a product to be labeled "natural", it must contain at least 95% natural raw materials. "Organic" products must include at least 10% organic ingredients in addition to meeting the natural content requirement. Brands must provide documentation verifying ingredient origins and manufacturing processes [9].
In Japan, there is no official government certification for "organic" or "natural" cosmetics. Instead, the Japan Cosmetic Industry Association (JCIA) follows ISO 16128 standards. These guidelines define natural and organic content and allow brands to calculate indices for marketing purposes. Only certain ingredients qualify as natural or organic under ISO 16128.
| Feature | China | South Korea | Japan |
|---|---|---|---|
| Primary Inventory | IECIC 2021 | Natural/Organic Ingredient Inventory | Positive/Negative Lists |
| New Ingredient Period | 3-year safety monitoring [10] | Not Applicable | Not Applicable |
| Organic Standards | Not officially recognized | >10% organic content [9] | Based on ISO 16128 |
| Natural Standards | No specific law | >95% natural content [9] | Based on ISO 16128 |
| Responsible Party | Domestic Responsible Person (DRP) | Manufacturing Sales Vendor | Primary Distributor/Importer |
This information is intended for general guidance. Always consult official regulations and qualified experts before making formulation or sourcing decisions.
Balancing Sustainability with Regulatory Requirements
Navigating the world of sustainable antioxidants means juggling a mix of consumer expectations and regulatory demands. Interest in sustainability within cosmetics has seen a noticeable rise, with consumer focus growing from 13.2% in 2018 to 18.9% in 2020 [14]. Yet, even as demand for "natural" or "organic" ingredients increases, these terms don’t always equate to lower regulatory concerns. For example, botanical antioxidants often include naturally occurring fragrance allergens, which trigger mandatory labeling. This is especially relevant in the European Union, where the list of declared allergens will expand from 26 to over 80 substances by 2026 [1]. Staying compliant requires constant vigilance, as restricted ingredients and transparency requirements continue to evolve. These complexities underline the need to examine certification standards and sourcing challenges more closely.
Certifications and Standards for Eco-Friendly Ingredients
To ensure accurate labeling, manufacturers must differentiate between terms like "natural" (unprocessed or mechanically processed), "naturally derived" (chemically processed plant-based materials), and "organic" (produced through biologically based farming methods). Standards like ISO 16128 and USDA NOP offer guidelines for these distinctions [14]. For products sold in Europe, ISO 16128 provides a method to calculate natural and organic content indices. Meanwhile, USDA NOP categorizes organic products into four levels based on their organic content percentage, each with specific labeling requirements. These standards help align sustainability claims with regulatory mandates [14].
In the European Union, compliance with Regulation (EC) No. 1223/2009 requires manufacturers to back eco-friendly antioxidant claims with validated evidence. Tests like DPPH, ORAC, or Cellular Antioxidant Activity (CAA) assays can provide this proof [12]. Combining chemical assays with cellular models can further support marketing claims by demonstrating how plant-derived antioxidants perform on living skin. As highlighted by Cosmetics Business:
As sustainability and natural formulations gain momentum, antioxidant testing helps demonstrate the real value of plant-derived actives and clean-label ingredients. [12]
Incorporating multifunctional ingredients with antimicrobial properties can also address "clean beauty" demands by reducing dependence on traditional high-dose preservatives [13]. Hurdle technology, which uses multiple preservation methods like pH control and antioxidant synergy, further minimizes risks while meeting regulatory requirements [13]. These frameworks help manufacturers tackle sourcing issues with greater confidence.
Common Sourcing Challenges
Even with clear certification guidelines, sourcing ingredients can be challenging. Transparency in the supply chain is critical, as regulations now require full disclosure of natural extract compositions to identify potential allergens. Laws like MoCRA in the United States and updated EU disclosure requirements mandate detailed composition data, making it essential for manufacturers to work closely with suppliers to verify ingredient information [1].
Cost and verification hurdles are also common. One promising solution is plant cell culture technology, which produces antioxidant metabolites like phenolic acids and flavonoids in controlled bioreactors. This approach eliminates contamination risks and is unaffected by soil or climate conditions, ensuring a consistent and traceable ingredient supply. It supports both sustainability goals and regulatory compliance [14].
Proactive reformulation is another key strategy for managing high-risk ingredients that may face reclassification as carcinogenic, mutagenic, or toxic for reproduction (CMR). Identifying these ingredients early allows brands to develop mitigation plans before regulatory deadlines create urgent challenges [1]. Monitoring supply chain transparency and securing detailed composition data from suppliers can help manufacturers stay compliant and avoid legal or reputational risks.
Collaborating with experienced sourcing partners can simplify these processes. For instance, Allan Chemical Corporation provides technical support, detailed documentation, and antioxidant ingredients tailored to meet evolving regulatory requirements.
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
How to Ensure Compliance for Antioxidant Ingredients
Ensuring compliance for antioxidant ingredients involves a structured approach that addresses ingredient selection, safety testing, and accurate labeling. These steps work together to create a compliance framework that safeguards both your brand and your consumers. Start by choosing approved ingredients and maintaining thorough documentation of your processes.
Selecting Ingredients and Maintaining Documentation
To comply with regulations, verify that your ingredients meet the requirements of your target market. For instance, consult the EU Cosmetics Regulation Annexes for concentration limits, review the FDA’s restricted substance lists, and confirm that your ingredient adheres to all conditions of use. If you’re using green tea extract (Camellia Sinensis Leaf Extract), check for naturally occurring allergens that may require labeling, especially as the EU plans to expand mandatory allergen declarations from 26 to over 80 substances by 2026 [1].
Always use International Nomenclature of Cosmetic Ingredients (INCI) names in your documentation and on labels. For example, instead of "Vitamin E", use "Tocopherol" or "Tocopheryl Acetate" to meet compliance standards [2]. Obtain complete composition data from your suppliers, including Certificates of Analysis (COAs), Safety Data Sheets (SDS), and allergen statements. This is particularly important for botanical extracts, which may contain hidden allergens [1][4]. Companies like Allan Chemical Corporation provide detailed documentation and technical support to help manufacturers ensure transparency and maintain audit-ready records.
Keep centralized safety files for all antioxidants. Under MoCRA, safety substantiation records must be retained for at least three years, while serious adverse event reports require six years of retention [2][4]. Document every supplier change or adjustment to ingredient concentrations to maintain a clear compliance history [4]. As Annel highlights:
A product can become non-compliant without any change to its formulation [1].
Regularly audit your documentation to stay ahead of regulatory changes, such as the EU’s May 2026 deadline for restrictions on CMR (carcinogenic, mutagenic, or toxic for reproduction) substances [1]. Once documentation is in order, the next step is rigorous safety testing of antioxidant ingredients.
Testing and Safety Evaluations
Safety testing is essential to confirm that your antioxidant formulations are effective and safe throughout their shelf life. Stability testing ensures the antioxidant retains its protective properties over time, while microbiological testing verifies the absence of harmful organisms. Similar rigor is required when you formulate long-wear cosmetic colorants to ensure pigment stability. Preservative Efficacy Testing (PET) evaluates whether the formula remains safe after the package is opened [4]. These tests collectively support the safety substantiation required under MoCRA [2][4].
To back marketing claims like "proven antioxidant protection", adopt a multi-layered testing approach. This can include chemical assays such as DPPH and ORAC, as well as cellular assays like CAA, to validate antioxidant activity [15]. For even more robust evidence, conduct clinical studies using skin imaging and biophysical measurements to demonstrate benefits like reduced fine lines [15]. Under EU Regulation (EC) No. 1223/2009, all claims must be supported by adequate evidence [15].
In the UK and EU, a Cosmetic Product Safety Report (CPSR) is mandatory as part of the Product Information File (PIF). Keep the CPSR updated to reflect current restrictions and concentration limits, including those set for 2026 [1]. In the U.S., the FDA can review your safety substantiation records under MoCRA, and serious adverse events must be reported within 15 business days [2][4]. Beyond testing, accurate labeling is a critical component of compliance.
Labeling Requirements and Marketing Claims
List ingredients in descending order using their INCI names, ensuring the physical label precisely matches the product listing submitted to the FDA. For packages larger than 5 square inches, ingredient declarations must use a minimum type size of 1/16 inch (1.6 mm) [2].
When making marketing claims, stay within the boundaries of cosmetic terminology. Use phrases like "improves appearance", "brightens", or "moisturizes", and avoid drug-like claims such as "prevents aging" or "treats skin damage" [2][4]. Review your website, social media, and influencer content to ensure that antioxidant claims remain compliant and do not unintentionally suggest therapeutic effects [4]. Both the EU and U.S. are intensifying their scrutiny of claim substantiation [1].
Fragrance transparency is also becoming more stringent. California and the FDA are moving toward requiring disclosure of specific hazardous components or allergens within fragrance blends, moving away from the blanket use of terms like "Parfum" or "Fragrance" [1][2]. If your antioxidant formulation includes fragrance, prepare for expanded allergen disclosure requirements anticipated between 2026 and 2027 [2].
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Conclusion
Ensuring regulatory compliance for antioxidant ingredients in cosmetics is an ongoing responsibility that requires constant attention. Regulatory frameworks evolve independently of your product formulations, making vigilance a necessity to keep your products compliant and your brand protected.
Recent developments in both the EU and U.S. highlight the importance of staying proactive. To address these changes effectively, precision and thoroughness should be at the heart of your compliance strategy. This not only safeguards consumer safety but also shields your brand from potential setbacks.
To stay market-ready, focus on three key practices: confirming that all ingredients meet the specific regulations of your target markets, maintaining detailed documentation (including INCI names and supplier information), and conducting comprehensive safety testing to validate your formulations. Whether dealing with the EU’s Annex restrictions, the FDA’s facility registration rules, or state-specific laws like California’s Proposition 65, a forward-thinking approach can help you avoid expensive reformulations or product recalls.
Collaborating with experienced suppliers, like Allan Chemical Corporation, can simplify compliance efforts. Reliable partners provide essential tools such as full composition data, Certificates of Analysis, and updated safety documentation, ensuring your records are always audit-ready. They also help identify potentially high-risk ingredients before regulatory deadlines become critical. With over 40 years of experience in regulated industries, Allan Chemical Corporation supports cosmetic manufacturers with technical expertise and comprehensive documentation to navigate complex global compliance demands.
As regulations around cosmetic antioxidants continue to tighten, treating compliance as a strategic advantage can position your brand for long-term success in a market that prioritizes safety and transparency.
This content is for informational purposes only. Always consult official regulations and qualified professionals when making sourcing or formulation decisions.
FAQs
When does an antioxidant claim make my product a drug in the U.S.?
In the United States, stating that a product has antioxidant properties can categorize it as a drug if the claim suggests it can prevent, treat, or cure a disease, or if it impacts the body’s structure or function. Such statements are regulated by the FDA and require the product to comply with drug-related standards.
What documents do I need to sell an antioxidant cosmetic in the EU?
To market an antioxidant cosmetic in the EU, you need to follow the EU Cosmetics Regulation (EC) No 1223/2009. This involves several key steps:
- Notification through the CPNP: Register your product using the Cosmetic Product Notification Portal (CPNP) before it hits the market.
- Creating a Product Information File (PIF): Compile detailed documentation about the product, including its composition, safety assessments, and manufacturing details.
- Ingredient Compliance: Verify that all ingredients adhere to safety standards and avoid substances that are banned or restricted under EU regulations.
These steps ensure your product meets legal and safety standards for the European market.
How do I know if my antioxidant is a ‘new ingredient’ in China?
In China, any antioxidant used for the first time in a cosmetic product is classified as a "new ingredient." This designation requires the ingredient to go through a formal registration or notification process to meet the country’s regulatory standards.





Comments are closed