Trehalose (CAS No. 99-20-7) and Glycerol (CAS No. 56-81-5) are two widely used excipients in biopharmaceutical formulations. Both are essential for stabilizing proteins under various conditions, but they work differently and suit specific applications. Trehalose, a non-reducing sugar, forms a rigid glassy matrix, making it ideal for freeze-drying and long-term storage. Glycerol, a liquid polyol, excels in liquid formulations and cryopreservation by preventing protein aggregation and ice damage.
Key Highlights:
- Trehalose: Best for solid-state uses like lyophilization; stabilizes proteins through hydration and vitrification.
- Glycerol: Effective in liquid systems; prevents ice formation and stabilizes hydrophobic protein regions.
- Synergy: Combining small amounts of Glycerol (e.g., 5%) with Trehalose enhances protein stability across temperature ranges.
Understanding their mechanisms and temperature-dependent performance helps formulators choose the right excipient or combination for specific protein-based drugs.
1. Trehalose
Trehalose (CAS No. 99-20-7) is a non-reducing disaccharide made up of two D-glucopyranose units connected by an α,α(1→1) bond. This unique structure gives trehalose exceptional chemical stability, preventing reactions like the Maillard reaction. Thanks to this stability, trehalose is widely used in biopharmaceutical formulations to protect complex protein-based drugs during processes such as freeze-drying, cryopreservation, and long-term storage. These properties make it a key ingredient for maintaining protein integrity.
Protein Stabilization Mechanisms
Trehalose stabilizes proteins through two main mechanisms. First, it employs preferential exclusion, which involves keeping trehalose molecules away from the protein surface. This ensures a protective hydration layer remains intact. By making protein unfolding energetically unfavorable, this mechanism helps maintain the protein’s structure, as unfolding would expose more surface area to the solvent, leading to less favorable interactions.
The second mechanism is vitrification, where trehalose forms a rigid, glassy matrix with a high glass transition temperature (Tg). This matrix restricts protein movement, reducing the risk of unfolding and aggregation [2]. Additionally, trehalose slows the movement of hydration water around proteins, which is critical since protein stability is closely tied to the dynamics of its surrounding hydration layer. By dampening these motions, trehalose further enhances protein stability [4].
Professor Jan Swenson from Chalmers University of Technology notes: "The hydration layer around protein molecules acts as a bridge between the sugar and the protein."
Temperature-Dependent Performance
Trehalose’s stabilization effects adjust based on temperature. Below its glass transition temperature, it slows local water relaxation. Above Tg, it hinders the cooperative solvent movements that drive protein motions [3]. In trehalose-lysozyme solutions, the α-relaxation of hydration water undergoes a fragile-to-strong transition at a temperature roughly 20° higher than that of bulk water [4]. This adaptability makes trehalose effective across various storage and processing conditions.
Applications in Biopharmaceuticals
Trehalose is widely used in biopharmaceuticals, particularly for freeze-drying (lyophilization), cryopreservation, and stabilizing protein-based drugs for long-term storage at room temperature. Its ability to form a protective glassy matrix is especially valuable for preserving monoclonal antibodies, vaccines, and other biologics. When combined with small amounts of glycerol (around 5% by weight), trehalose enhances protein stability even further, raising the protein unfolding temperature to its peak [1]. These synergistic effects highlight trehalose’s versatility and set the stage for comparisons with glycerol in later sections.
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
sbb-itb-aa4586a
2. Glycerol
Glycerol (CAS No. 56-81-5) is a liquid polyol, or sugar alcohol, widely recognized for its ability to stabilize proteins. Unlike trehalose, which is more suited for solid-state uses like freeze-drying, glycerol works best in liquid formulations. Its liquid state makes it a go-to choice for aqueous solutions, refolding buffers, and cryopreservation, where maintaining proteins in their native form is critical.
Protein Stabilization Mechanisms
Glycerol operates differently from trehalose, thriving in liquid environments to stabilize proteins. It achieves this through preferential hydration, where glycerol molecules are excluded from the protein surface due to electrostatic forces. This exclusion raises the protein’s chemical potential, encouraging it to remain in its compact native state. Additionally, glycerol acts as an amphiphilic stabilizer, binding to large hydrophobic patches on proteins, which helps stabilize intermediates prone to aggregation [5][6].
Vincent Vagenende from the Massachusetts Institute of Technology notes: "Glycerol prevents protein aggregation by inhibiting protein unfolding and by stabilizing aggregation-prone intermediates through preferential interactions with hydrophobic surface regions" [5].
Temperature-Dependent Performance
Glycerol generally raises the temperature at which proteins unfold, though its effects can vary depending on the protein [1][5]. For instance, while glycerol stabilizes yeast enzymes, higher concentrations (7.5–25% v/v) at 122°F (50°C) can reduce the activity of rabbit muscle phosphofructokinase by 2–4 times compared to untreated samples [7].
Interestingly, glycerol can work in tandem with trehalose, providing even greater protein stability [1].
Applications in Biopharmaceuticals
Thanks to its protein-stabilizing properties, glycerol plays a vital role in biopharmaceutical applications. It complements trehalose by stabilizing proteins in liquid formulations, where solid-state methods are less effective. In recombinant protein production, glycerol is used in refolding buffers to prevent aggregation [5]. It also serves as a cosolvent in liquid formulations, extending shelf life and preserving protein stability during storage. As a cryoprotectant, glycerol prevents ice crystal formation, which could otherwise damage proteins in frozen or supercooled environments [6]. Beyond these uses, glycerol is employed in protein crystallization to improve crystal quality and in biocatalyst formulations to maintain enzyme activity in microtiter plates [6].
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Performance at Different Temperatures
Temperature significantly influences the effectiveness of osmolytes like trehalose and glycerol in stabilizing proteins. Each performs differently across temperature ranges, directly affecting their ability to combat protein degradation. Understanding these mechanisms highlights their unique roles under varying conditions.
At low temperatures (below 32°F or 0°C), glycerol proves more effective than trehalose. Thanks to its high mobility in the liquid state, glycerol prevents ice crystal formation – a key factor in cryopreservation. However, choosing the right agent is critical, as there are various cryoprotectant use limitations to consider regarding toxicity and formulation. Trehalose, meanwhile, forms a protective glassy state upon freezing, offering long-term stabilization.
The dynamic shifts at high temperatures. A January 2005 study in Molecular and Cellular Biochemistry examined rabbit muscle 6-phosphofructo-1-kinase (PFK) stability at 122°F (50°C). Researchers Joana Faber-Barata and Mauro Sola-Penna found that 1 M trehalose completely protected the enzyme from inactivation during a 120-minute incubation, while the control enzyme’s half-life was only 56 minutes. Conversely, glycerol concentrations between 7.5% and 25% (v/v) resulted in enzyme activity reductions of 2–4 times compared to the control[7].
"Trehalose was able to protect phosphofructokinase against thermal inactivation as well as to promote an activation of its catalytic activity… On the other hand, enzyme incubated in the presence of 37.5% (v/v) glycerol was not protected against incubation at 50 °C."
– Mauro Sola-Penna, Professor, Universidade Federal do Rio de Janeiro[7]
Combining these osmolytes can provide benefits across a broader temperature spectrum. For instance, adding about 5% glycerol to a trehalose matrix creates an anti-plasticizing effect that stiffens the matrix, enhancing protein stability beyond what trehalose alone achieves[1].
Here’s a summary of their temperature-dependent performance:
| Temperature Range | Trehalose Efficiency | Glycerol Efficiency | Primary Mechanism |
|---|---|---|---|
| Low (Below 32°F/0°C) | Moderate; effective for long-term vitrification | High; prevents ice crystal formation during freezing | Glycerol for immediate cryoprotection; Trehalose for glass formation |
| Moderate (Room Temperature) | Excellent; high glass transition temperature ensures a stable, rigid environment | Moderate; acts as a liquid stabilizer yet may increase reactivity | Trehalose preferred for lyophilized products |
| High (Above 122°F/50°C) | Superior; prevents unfolding and maintains catalytic activity | Low to Negative; may accelerate inactivation of certain enzymes | Trehalose is the primary choice for heat-stable formulations |
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Use in Biopharmaceutical Formulations
Protein-based drug formulations rely heavily on excipients to preserve protein integrity during manufacturing, storage, and administration. These excipients are critical to ensuring the stability of protein-based biopharmaceuticals. Among the most effective stabilizers in this area are Trehalose and Glycerol, each offering unique benefits that can directly impact product shelf life, performance, and compliance with regulatory standards.
Trehalose plays a key role in stabilizing lyophilized (freeze-dried) formulations by creating a protective glassy matrix around the proteins. On the other hand, Glycerol is essential for stabilizing liquid formulations and is particularly effective in cryopreservation, where it prevents damage caused by freezing. Together, these excipients address different stability challenges, making them indispensable in formulation development.
For example, a study on stem cell preservation demonstrated the benefits of combining these two excipients. A formulation with 1.0 M Trehalose and 20% Glycerol achieved 77% cell viability, outperforming Trehalose alone (65% viability) and Glycerol alone (63% viability) [8]. This synergy highlights their potential as an effective alternative to traditional cryoprotectants in clinical applications.
When selecting excipients for biopharmaceutical formulations, quality is non-negotiable. Using compendial-grade materials – those that meet USP or NF standards – is essential to maintain consistent purity, identity, and quality. Lower-grade materials often contain impurities that can accelerate protein aggregation, undermining both safety and efficacy. In addition to stabilizers, surfactants prevent protein aggregation by protecting against surface-induced stress. For regulated pharmaceutical manufacturing, choosing high-quality excipients is a critical step. Allan Chemical Corporation provides USP-grade Trehalose and Glycerol, complete with full documentation, including Certificates of Analysis and Safety Data Sheets, ensuring compliance with stringent regulatory requirements.
Achieving the right balance between Glycerol and Trehalose is key to optimizing thermal protection while avoiding issues like increased viscosity or osmotic stress. This balance builds on earlier insights into temperature-dependent performance, offering a reliable approach to effective formulation strategies.
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Advantages and Disadvantages
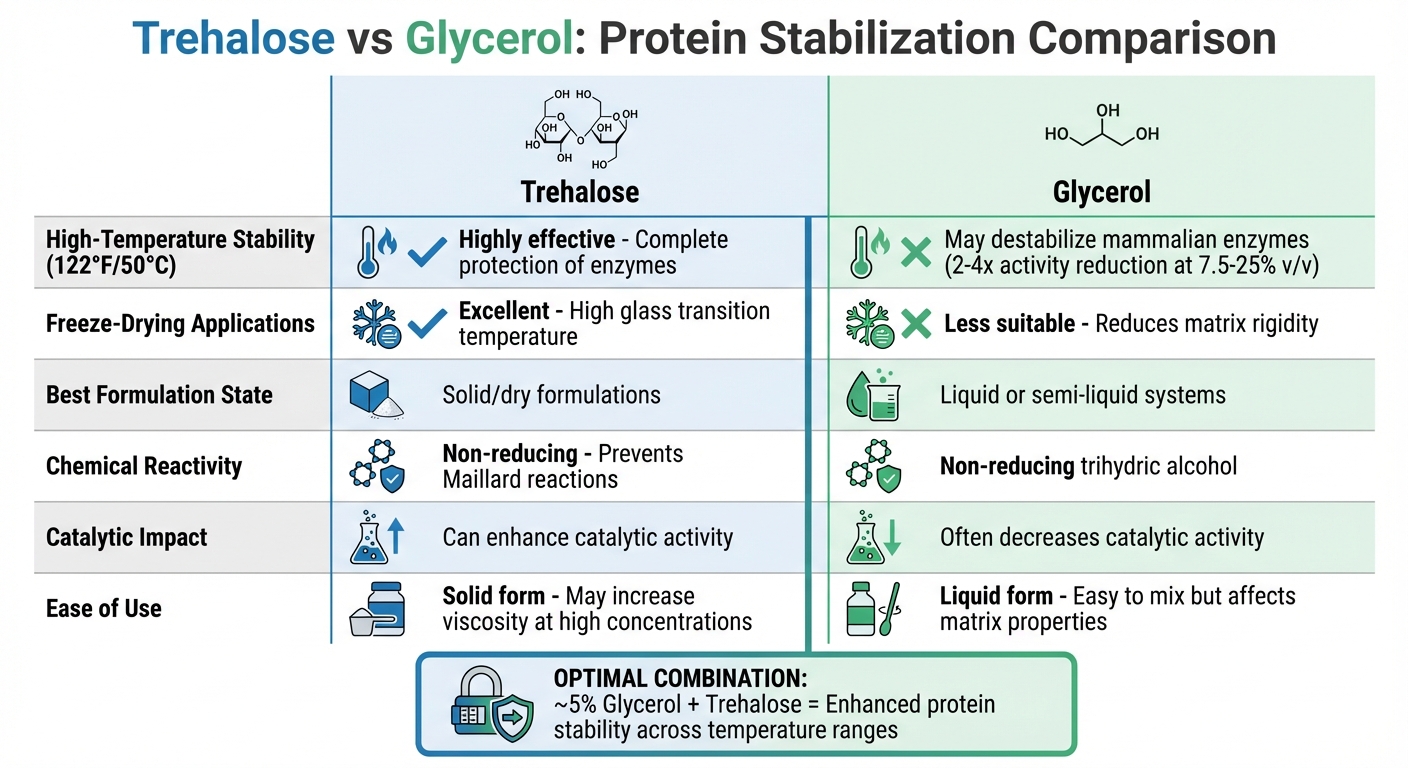
Trehalose vs Glycerol Protein Stabilization Comparison Chart
Looking at the benefits and challenges of trehalose and glycerol, it’s clear that these excipients bring unique strengths to protein stabilization, depending on the specific conditions and formulation needs.
Trehalose stands out in freeze-dried formulations due to its high glass transition temperature and non-reducing nature, which help minimize unwanted chemical reactions during storage. For instance, in thermal stability studies, 1 M trehalose completely protected rabbit muscle phosphofructokinase (PFK) at 122°F (50°C) over two hours. In contrast, the control enzyme was fully inactivated with a half-life of just 56 ± 5 minutes [7]. Trehalose not only shields enzymes from heat damage but can also enhance catalytic activity. However, its performance can vary depending on the protein type and the overall formulation [7].
On the other hand, glycerol shines in different formulation states. When used at around 5% in a trehalose matrix, glycerol acts as an anti-plasticizer. This creates a more rigid trehalose/water/glycerol matrix, which can further stabilize proteins.
Giuseppe Bellavia from UMET, Université Lille 1, explained, "Glycerol anti-plasticizes the matrix with respect to the trehalose by enhancing the stability of the protein in a more rigid trehalose/water/glycerol matrix" [1].
However, glycerol has its downsides, particularly at higher concentrations. For example, during thermal stability tests at 122°F (50°C), glycerol (7.5–25% v/v) reduced the activity of mammalian enzymes like PFK by 2–4 times compared to control samples. Even at 37.5% (v/v), glycerol failed to provide protection [7].
Here’s a quick comparison of the two excipients:
| Factor | Trehalose | Glycerol |
|---|---|---|
| High-Temperature Stability | Highly effective; protects enzymes at 122°F (50°C) [7] | May destabilize mammalian enzymes [7] |
| Freeze-Drying Applications | Excellent due to high glass transition temperature [2] | Less suitable; reduces matrix rigidity [1] |
| Formulation State | Best for solid/dry formulations [2] | Ideal for liquid or semi-liquid systems [1] |
| Chemical Reactivity | Non-reducing; prevents Maillard reactions [2] | Non-reducing trihydric alcohol |
| Catalytic Impact | Can enhance catalytic activity [7] | Often decreases catalytic activity [7] |
| Ease of Use | Solid form; can increase viscosity at high concentrations | Liquid form; easy to mix but affects matrix properties |
Joana Faber-Barata and Mauro Sola-Penna noted, "The compatibility of effects previously shown for trehalose and glycerol with some yeast cytosolic enzymes can not be extended to all globular enzyme system" [7].
This variability highlights the importance of testing both excipients – individually and in combination – when developing formulations. Each protein system responds differently, making tailored testing essential.
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Conclusion
Different excipients shine under unique conditions, and understanding their roles is key to effective protein stabilization. Trehalose stands out in freeze-dried products and high-temperature environments, thanks to its high glass transition temperature and preferential hydration properties. On the other hand, Glycerol works well as a co-stabilizer in low-water systems, where it enhances the rigidity of the trehalose matrix and provides additional thermal protection.
A combined approach often yields the best results. For instance, incorporating about 5% glycerol by weight into a trehalose matrix creates a stable, rigid structure that helps maintain protein conformation and raises unfolding temperatures. This synergy amplifies the protective effects of each excipient, as outlined earlier.
It’s important to tailor formulations to the specific protein system. Trehalose is ideal for lyophilized formulations requiring extended stability, while glycerol can fine-tune matrix rigidity in liquid or semi-liquid systems. Testing both excipients individually and in combination will help identify the optimal ratio for your application.
For excipients that meet regulatory standards like USP, FCC, ACS, and NF, Allan Chemical Corporation provides trusted solutions backed by over four decades of experience in regulated industries. These insights empower formulators to harness the strengths of trehalose and glycerol for robust, customized protein stabilization.
This content is for informational purposes only. Always consult official regulations and qualified experts before making sourcing or formulation decisions.
FAQs
How do I choose trehalose vs glycerol for my protein?
When deciding between trehalose and glycerol, it’s all about the application and the stabilization effects you’re aiming for.
Trehalose works best for dehydration or freezing scenarios. It protects proteins by creating a glass-like matrix that helps maintain their structure. On the other hand, glycerol boosts stability by making proteins more rigid and limiting molecular motion, which is especially useful under stressful conditions.
To make the right choice, think about your process conditions, how well the stabilizer fits with other formulation components, and the specific stability requirements of your protein product.
When should trehalose and glycerol be used together?
Trehalose and glycerol are often paired in biopharmaceutical formulations to improve protein stability. While trehalose acts as a stabilizer, glycerol enhances its effectiveness by improving interactions between the protein and solvent. This pairing is particularly useful during stress conditions, such as freeze-drying, where glycerol extends trehalose’s protective effects. However, it’s essential to carefully control glycerol concentration, as too much can counteract the stabilization benefits. This combination is ideal when sensitive proteins require greater thermal and structural stability.
What glycerol level is “too much” for stability?
High glycerol levels – specifically those exceeding 50% – are often seen as excessive when it comes to maintaining protein stability. Such elevated concentrations can disrupt stability, which is why adhering to recommended limits is crucial during the formulation of biopharmaceuticals.





Comments are closed