Cosmetic pigments are the color additives that give cosmetics their visual appeal and help identify products. In the U.S., the FDA regulates these pigments under the Federal Food, Drug, and Cosmetic (FD&C) Act to ensure safety and compliance. FDA-approved pigments undergo rigorous testing for purity, safety, and specific uses, while non-approved pigments do not meet these standards and are considered illegal for use in cosmetics.
Key points to know:
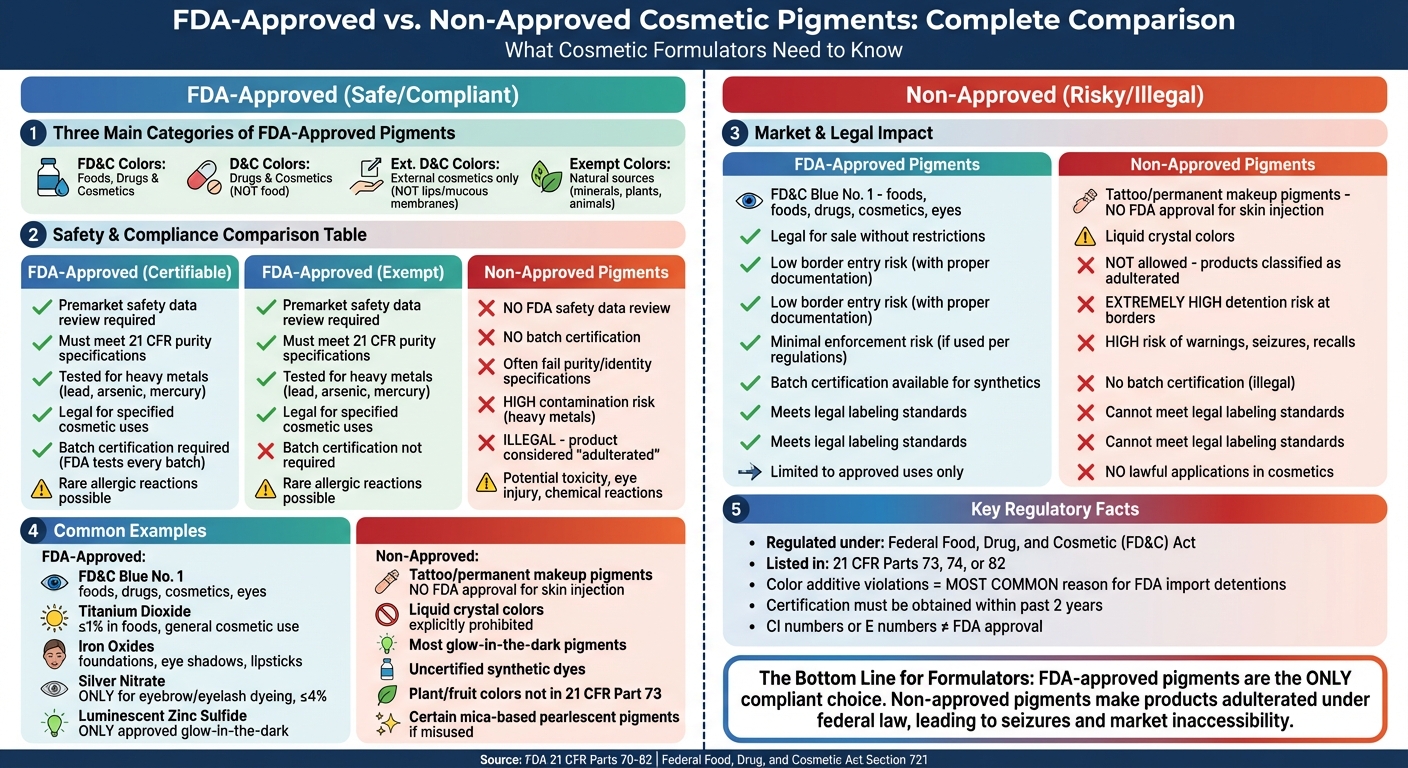
- FDA-approved pigments are listed in 21 CFR Parts 73, 74, or 82 and include categories like FD&C, D&C, Ext. D&C colors, and exempt natural colors (e.g., iron oxides, titanium dioxide). Some require batch certification for purity.
- Non-approved pigments lack FDA review, pose safety risks, and are illegal for cosmetic use. This includes pigments used in tattoos, non-certified synthetic dyes, and certain plant-derived colors.
- Using non-approved pigments can lead to product seizures, import detentions, or recalls due to legal non-compliance.
For formulators, sourcing pigments with proper FDA certification ensures product safety, legal compliance, and market access. Avoiding non-approved pigments protects consumers and prevents enforcement actions.
FDA-Approved vs Non-Approved Cosmetic Pigments Safety and Compliance Comparison
What the FDA Says About Cosmetic Neons and Fluorescents | Day 336/365
sbb-itb-aa4586a
What Are FDA-Approved Cosmetic Pigments?
An FDA-approved cosmetic pigment is defined in the Code of Federal Regulations (CFR) by its identity, chemical specifications, and approved uses. To gain approval, manufacturers must submit a Color Additive Petition, including scientific evidence that demonstrates the pigment’s safety [1]. Once approved, the pigment is officially listed in 21 CFR Parts 73, 74, or 82, depending on its source and category.
These pigments fall into three main categories:
- FD&C colors: Approved for use in foods, drugs, and cosmetics.
- D&C colors: Approved for use in drugs and cosmetics, but not in food.
- Ext. D&C colors: Approved only for externally applied cosmetics, excluding use on lips or mucous membranes.
A fourth group, exempt colors, includes pigments derived from natural sources like minerals, plants, or animals, such as iron oxides and titanium dioxide. While exempt colors are not subject to batch-by-batch FDA testing, they must still meet strict purity and identity standards outlined in 21 CFR Part 73. These classifications highlight the rigorous safety measures surrounding cosmetic color additives.
Approval is use-specific, meaning a pigment cleared for one purpose might not be allowed for another. For instance, a pigment approved for general use may still be prohibited around the eyes unless explicitly authorized. Using a pigment outside its approved application renders the product legally "adulterated", which may lead to detentions at U.S. borders or enforcement actions.
Characteristics of FDA-Approved Pigments
FDA-approved pigments undergo stringent safety testing and must meet strict purity standards. For synthetic-organic colors, often derived from petroleum, the FDA requires batch certification. This involves manufacturers submitting pigment samples for FDA testing before they can sell them. The certification process includes detailed analyses of color content, moisture levels, salts, and heavy metals [2].
Certified pigments must display an FDA lot certification number on their labels, along with their legal name (e.g., "FD&C Blue No. 1"). This certification number confirms the batch has passed FDA testing and complies with the purity standards outlined in 21 CFR Part 74 or Part 82. It’s important to note that a Colour Index (CI) number or European "E number" does not indicate FDA approval [4].
Below are some examples of pigments that meet these rigorous standards.
Common FDA-Approved Pigments
Several pigments have gained widespread use in the cosmetics industry due to their FDA approval and versatility. For instance:
- FD&C Blue No. 1: A synthetic certifiable color approved for use in foods, drugs, and cosmetics, including around the eyes [2].
- Titanium dioxide (CAS No. 13463-67-7): An exempt mineral pigment approved for general cosmetic use and in foods, provided its concentration does not exceed 1% by weight [2].
- Iron oxides: Exempt from certification, these are approved for general cosmetic use and are commonly found in products like foundations, eye shadows, and lipsticks.
Some pigments come with specific restrictions. For example, silver nitrate is the only color additive approved for dyeing eyebrows or eyelashes, with its use limited to professional applications at a maximum concentration of 4% [4]. Luminescent zinc sulfide is the only approved "glow-in-the-dark" pigment for cosmetic use [4]. However, it’s worth noting that no FDA-approved color additives are permitted for skin injection, including tattooing and permanent makeup [4].
Disclaimer: This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
Non-Approved Cosmetic Pigments: What You Need to Know
A cosmetic pigment is classified as non-approved if it: (1) is not listed in the FDA’s official regulations (21 CFR Parts 73, 74, or 82), (2) is used in ways outside its approved purpose, or (3) fails to meet the purity and identity standards outlined in its regulatory listing. While non-approved pigments are also subject to regulatory oversight, failing to meet these standards can compromise both product safety and marketability.
Types of Non-Approved Pigments
Pigments used in tattoos and permanent makeup are non-approved because no additives have been FDA-approved for injection into the skin. Some manufacturers falsely advertise their tattoo inks as "FDA-approved", a claim that is legally invalid.
Liquid crystal colors, known for their distinctive light-diffraction effects, are explicitly prohibited for cosmetic use. Similarly, most glow-in-the-dark pigments are not FDA-approved, with luminescent zinc sulfide being the only approved option for creating a glowing effect. Using uncertified synthetic dyes also violates regulations – for instance, substituting plain tartrazine for the batch-certified FD&C Yellow No. 5 is illegal since it bypasses required FDA laboratory testing for purity [4][5].
Colors derived from plants or fruits that are not listed in 21 CFR Part 73 are also considered non-approved. Certain mica-based pearlescent pigments fall into the same category if used outside their approved applications or concentration limits. Another common misconception is that a Colour Index (CI) number or a European "E number" equates to FDA certification. However, these identifiers have no legal standing in the United States [4][5].
Using these unapproved pigments not only violates regulations but also raises serious safety concerns.
Safety Concerns with Non-Approved Pigments
Non-approved pigments bypass FDA safety testing, leaving significant health risks unaddressed. These pigments have not submitted a Color Additive Petition, which is required to demonstrate safety through scientific evidence. Without mandatory batch certification, synthetic pigments may contain harmful levels of heavy metals like lead, arsenic, or mercury, as well as carcinogenic contaminants [2].
The lack of oversight for non-approved pigments exposes both manufacturers and consumers to potential dangers, including severe allergic reactions and long-term health issues. Historical examples highlight these risks: in the 1930s, an unapproved eyelash dye caused blindness in several women, prompting the creation of stricter regulations under the Federal Food, Drug, and Cosmetic Act [2].
Violations involving color additives are among the most common reasons the FDA detains imported cosmetics at U.S. borders [4]. Using pigments outside their approved applications can also lead to additional safety issues. For instance, a pigment approved for external use might not be safe for application near the eyes or lips unless specifically authorized for those areas. The FDA defines the eye area as the skin within the supra-orbital and infra-orbital ridges, encompassing the eyebrows and eyelashes. Most color additives are prohibited in this sensitive region unless explicitly approved [4].
Disclaimer: This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
Safety Comparison: FDA-Approved vs. Non-Approved Pigments
When it comes to pigments in cosmetics, the difference between FDA-approved and non-approved options is all about safety and compliance. FDA-approved pigments go through a detailed safety evaluation process, while non-approved pigments skip this entirely, leaving potential risks unchecked. The FDA requires approved pigments to pass the Color Additive Petition process, where manufacturers submit scientific data proving the pigment is safe for its intended use. Only after this review does the FDA confirm there is a "reasonable certainty of no harm" under the proposed conditions [3]. Non-approved pigments, on the other hand, avoid this scrutiny, which can lead to unaddressed health concerns.
"Color additives are safe when used properly. There is no such thing as absolute safety of any substance. In the case of a new color additive, the FDA determines if there is ‘a reasonable certainty of no harm’ under the color additive’s proposed conditions of use." – Linda Katz, M.D., M.P.H., Director of the FDA’s Office of Cosmetics and Colors [3]
Another key safety measure for FDA-approved pigments is the batch certification process, required for certain synthetic dyes. The FDA tests samples from every batch to ensure they meet strict purity standards before issuing a certification lot number [3]. Non-approved pigments lack this rigorous testing, which increases the likelihood of contamination with harmful substances like lead, arsenic, or mercury [6].
Using non-approved pigments not only poses safety risks but also violates the Federal Food, Drug, and Cosmetic (FD&C) Act. Products containing such pigments are classified as "adulterated", making them subject to enforcement actions such as seizures, warning letters, or import alerts [3]. This legal status can severely impact both consumer safety and a company’s ability to sell its products.
Safety Comparison Table
| Feature | FDA-Approved (Certifiable) | FDA-Approved (Exempt) | Non-Approved Pigments |
|---|---|---|---|
| Safety Testing | Premarket safety data review required | Premarket safety data review required | No FDA safety data review for intended use |
| Batch Certification | Required; FDA tests every batch for purity | Not required | None |
| Purity Standards | Must meet 21 CFR specifications | Must meet 21 CFR specifications | Often fail purity/identity specifications |
| Contamination Risk | Tested to confirm acceptable heavy metal levels | Tested to confirm acceptable heavy metal levels | High risk of contamination with lead, arsenic, or mercury |
| Legal Status | Legal for specified cosmetic uses | Legal for specified cosmetic uses | Illegal; product considered "adulterated" |
| Health Concerns | Rare allergic reactions (e.g., hives from Yellow 5) | Rare allergic reactions | Potential toxicity, eye injury, or chemical reactions |
Disclaimer: This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
Regulatory Requirements and Certification
The Federal Food, Drug, and Cosmetic (FD&C) Act, through Section 721, grants the FDA authority to regulate cosmetic pigments. According to this law, any cosmetic product containing an unapproved or improperly used color additive is considered "adulterated" and cannot be legally sold in the U.S. Manufacturers must submit a Color Additive Petition that includes scientific evidence proving the pigment’s safety for its intended use. Only after this review can the pigment be added to the Code of Federal Regulations (CFR).
To comply with FDA regulations, pigments must meet several requirements: approval through a CFR listing, batch certification for synthetic pigments, adherence to strict identity and purity standards outlined in 21 CFR, and use restrictions specifying allowable applications like the eye area, lips, or external use. These rules are detailed in 21 CFR Parts 70–82, with Parts 73, 74, and 82 focusing on approved additives and their chemical specifications. This regulatory framework underpins the rigorous certification process for FDA-approved pigments.
How FDA-Approved Pigments Get Certified
Pigments approved by the FDA fall into two categories: certified synthetic colors and exempt colors from natural sources. Certified colors are synthetic pigments, often derived from petroleum (historically referred to as "coal-tar dyes"), and require FDA testing for purity and composition before a lot certification number is issued. Examples include FD&C Yellow No. 5 and D&C Red No. 7.
Exempt colors, on the other hand, are derived from natural sources such as minerals, plants, or animals. These include materials like iron oxides and mica. While they do not require batch-by-batch testing, they must still meet strict identity and purity standards outlined in 21 CFR Part 73.
When sourcing certified colors, it’s crucial to ensure the label includes the legal name (e.g., "FD&C Yellow No. 5") and the FDA lot certification number. Additionally, suppliers should have obtained certification within the past two years, which can be verified using the FDA’s list of active companies.
Legal Risks of Using Non-Approved Pigments
The use of non-approved pigments poses serious legal risks. Violations related to color additives are among the most frequent reasons for the FDA to detain imported cosmetics. The agency can issue warning letters, impose import alerts, seize products, or even require voluntary recalls to remove non-compliant items from the market.
Even FDA-approved pigments can lead to legal issues if misused. Non-approved pigments, including those commonly found in tattoos or permanent makeup, violate regulatory standards. For example, silver nitrate is the only color additive permitted for professional eyebrow and eyelash dyeing, with a maximum concentration limit of 4% by weight [4]. While certification ensures safety for approved pigments, any deviation from these standards can result in severe consequences. Adhering to these regulations is essential to protect consumer safety and maintain product compliance.
Certification Process Comparison Table
| Feature | FDA-Certified Pigments | FDA-Exempt Pigments | Non-Approved Pigments |
|---|---|---|---|
| Source | Petroleum/synthetic | Mineral, plant, or animal | Various (unregulated) |
| FDA Batch Testing | Required for every batch | Not required | N/A (illegal for use) |
| Naming Convention | FD&C or D&C + Color + Number | Common or chemical name | N/A |
| CFR Reference | 21 CFR Parts 74 and 82 | 21 CFR Part 73 | None |
| Labeling Requirement | Must include legal name and lot number | Can often be listed as "artificial color" | N/A |
| Import Detention Risk | High if lot number is missing | Moderate if specifications aren’t met | Very high; detention |
| Legal Status | Approved for specified uses | Approved if meeting CFR specifications | Prohibited; considered adulterated |
Disclaimer: This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
Market and Legal Impact for Cosmetic Formulators
When it comes to cosmetics, the pigments you choose can make or break your product’s ability to reach the market. Beyond safety and certifications, these choices have far-reaching effects on legal compliance and market access.
Market Access and Legal Sales
In the U.S., the use of FDA-approved pigments is non-negotiable for legal sales. Under the Federal Food, Drug, and Cosmetic Act, any product containing non-approved pigments is labeled "adulterated" and cannot be sold across state lines [1]. The penalties for non-compliance aren’t just theoretical – they can include enforcement actions that block market entry entirely.
FDA approval is also highly specific, meaning pigments can only be used for their designated purpose. This makes it essential for formulators to confirm that every pigment aligns with its intended application. Failure to do so can lead to severe consequences, such as product seizures or import alerts.
"The FDA can issue warning letters, detentions, and import alerts for products that are found to be unsafe or contain color additives that are unauthorized, misused, or not properly identified as ingredients. The FDA can also seize such products." – U.S. Food and Drug Administration [3]
These legal requirements significantly influence both formulation strategies and overall costs.
Compliance Costs and Sourcing
While using FDA-approved pigments may involve upfront costs for verification, these costs are small compared to the financial and reputational damage caused by enforcement actions. To ensure compliance, formulators need to source pigments with an FDA-issued certification lot number for synthetic colors [4]. Checking the FDA’s list of certified suppliers is a critical step in this process.
Working with knowledgeable chemical suppliers can also ease the burden of compliance. For example, Allan Chemical Corporation, a specialty chemical provider with over 40 years of experience in regulated industries, offers robust supplier networks and documentation support. They help ensure pigments meet FDA standards for identity and purity. AllanChem is known for its just-in-time delivery and competitive pricing, as well as providing technical-grade and compendial-grade products (USP, FCC, ACS, NF). This level of support helps formulators maintain market access while managing compliance costs effectively.
For formulators, the upfront investment in certified pigments is a safeguard against the far more significant risks of product recalls, detentions, or legal issues.
Market Impact Comparison Table
To better understand the stakes, here’s a side-by-side look at the market and legal implications of using FDA-approved versus non-approved pigments:
| Feature | FDA-Approved Pigments | Non-Approved Pigments |
|---|---|---|
| Market Eligibility | Legal for sale without restrictions | Not allowed; products are classified as adulterated [1] |
| Border Entry Risk | Low with proper documentation | Extremely high risk of detention [3] |
| Enforcement Risk | Minimal if used according to regulations | High risk of warnings, seizures, or recalls [3] |
| Batch Certification | Mandatory for synthetic pigments [4] | Not available (illegal) [4] |
| Labeling Requirements | Must include legal name and certification lot number [4] | Cannot meet legal labeling standards |
| Application Flexibility | Limited to approved uses only [3] | No lawful applications in cosmetics |
| Supplier Verification | Requires checking FDA certification list [4] | Not applicable (prohibited) |
Disclaimer: This information is intended for general guidance only. Always refer to official regulations and consult qualified professionals for specific decisions.
Conclusion
When it comes to cosmetic pigments, the decision between FDA-approved and non-approved options is straightforward: FDA-approved pigments are the only compliant choice. These pigments meet strict premarket approval standards, adhere to FDA regulations, and, when necessary, undergo batch-by-batch certification. Non-approved pigments, on the other hand, lack this safety validation and render products adulterated under federal law – leading to potential product seizures and complete market inaccessibility [1].
For cosmetic formulators operating in regulated markets, working with a trusted supplier is essential. Allan Chemical Corporation, with over 40 years of experience in regulated industries, specializes in providing FDA-compliant pigments. They offer verified certification lot numbers, Certificates of Analysis, and expertise in just-in-time delivery, all while maintaining competitive pricing. This combination of reliability and expertise helps ensure compliance without compromising on quality or efficiency.
By choosing FDA-approved pigments, formulators not only meet regulatory requirements but also protect their customers, safeguard their brand reputation, and maintain market competitiveness. Partnering with experienced suppliers who understand 21 CFR standards ensures a smoother path to product launches and builds consumer trust while offering legal peace of mind.
Disclaimer: This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
FAQs
How can I verify if a pigment is FDA-approved for my specific use?
To ensure a pigment is approved by the FDA for your specific use, verify that it has passed the required safety testing and batch certification mandated by the FDA. Always obtain pigments from reputable suppliers who can provide full documentation, including detailed ingredient information and certification confirming its approval for your intended application. FDA-approved pigments adhere to stringent purity standards and undergo rigorous safety testing to meet regulatory compliance.
What proof should suppliers provide for FDA-certified colors?
Suppliers are required to submit documentation verifying FDA approval for the color additive. This usually involves batch certification and proof that the product adheres to the necessary safety and purity standards. All provided materials must clearly show compliance with FDA regulations.
What happens if my product uses a non-approved pigment?
Using a pigment that hasn’t been approved can cause your product to be labeled as adulterated under U.S. law. This can trigger serious consequences, like health hazards, product recalls, or even border detentions. Non-approved colorants haven’t undergone the FDA’s safety testing or received clearance, making compliance with FDA regulations critical for ensuring both product safety and legal integrity.





Comments are closed