Wafer cleaning requires precision to remove microscopic contaminants that can compromise performance. Solvents like d-Limonene, Acetone, and Isopropyl Alcohol (IPA) are commonly used for tasks such as removing hydrocarbon waxes, cleaning organic residues, and preparing wafers for subsequent manufacturing steps. These solvents must meet high-purity standards (e.g., metallic impurities below 1 ppm) to avoid defects during processes like photoresist adhesion or metallization.
Key takeaways for effective solvent use include:
- Solvent Selection: Match solvents to contaminants (e.g., d-Limonene for waxes, IPA for final rinsing).
- Cleaning Procedures: Control temperature and timing; use multi-step cleaning methods like RCA cleaning.
- Solvent Quality: Monitor for degradation (e.g., particle buildup, metal contamination) and replace solutions as needed.
- Safety Protocols: Use appropriate PPE, follow handling guidelines, and ensure compliance with regulatory standards.
Proper solvent management not only ensures wafer integrity but also prevents costly production losses, which can exceed $50,000 per batch.
Selecting the Right Solvent for Wafer Cleaning
Common Solvent Types and Their Uses
Acetone and isopropyl alcohol (IPA) are popular choices for removing organic residues, such as grease and oils, during wafer processing. Acetone’s low flash point (–4°F) allows it to evaporate quickly, but it can leave behind carbon residues if not properly rinsed. IPA, which has a flash point of about 53°F, is often used as a final rinse to displace any lingering higher-boiling solvents before drying the wafer.
For removing temporary bonding wax, d‑Limonene is highly effective. It dissolves paraffin-based waxes without damaging materials like silicon, aluminum, copper, gold, or nickel. Since d‑Limonene is a high–boiling point solvent and water-insoluble, an IPA rinse is typically necessary to eliminate any residual film. Ethanol is another option, valued for its evaporation rate and material compatibility in certain cleaning steps.
Always verify each solvent’s compatibility with the materials in the wafer to avoid potential defects during processing.
Checking Material Compatibility
Material compatibility plays a crucial role in solvent selection. For instance, wafers with a surface oxide layer generally require less effort to remove particles compared to bare silicon. The wafer’s crystalline structure also impacts cleaning. Alkaline mixtures, such as potassium hydroxide combined with IPA, are often used with monocrystalline wafers due to their directional etching properties. On the other hand, hydrofluoric/nitric acid systems are preferred for multicrystalline silicon to achieve isotropic texturing.
Before introducing a new solvent, ensure it works well with all materials involved, including silicon substrates, metal layers, polymer passivation, and dielectric films. Different chemistries can result in dramatically varied etch rates, so careful testing is essential.
Matching Solvents to Contamination Types
To maintain wafer integrity, it’s important to match solvents to the specific type of contamination. Polar solvents, such as water or a 50/50 ethanol-acetone mixture, are more effective at removing particulate matter than non-polar options like Freons. For ionic contaminants, such as salts or sweat residues, deionized (DI) water with contamination levels below 1–2 parts per million is essential, as organic solvents cannot dissolve these compounds. Metallic impurities, on the other hand, are best addressed with acidic mixtures – typically hydrochloric acid combined with hydrogen peroxide – that form soluble metal complexes.
When dealing with molecular contaminants, such as photoresist or pump oil trapped in high–aspect-ratio features, solvent penetration becomes critical. Acetone evaporates too quickly for deep microstructures, so heating d‑Limonene to between 104°F and 158°F and using ultrasonic assistance can significantly improve penetration and residue removal.
Disclaimer: This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
sbb-itb-aa4586a
How to clean a wafer with a quick solvent clean?
Effective Cleaning Procedures for Wafer Solvents
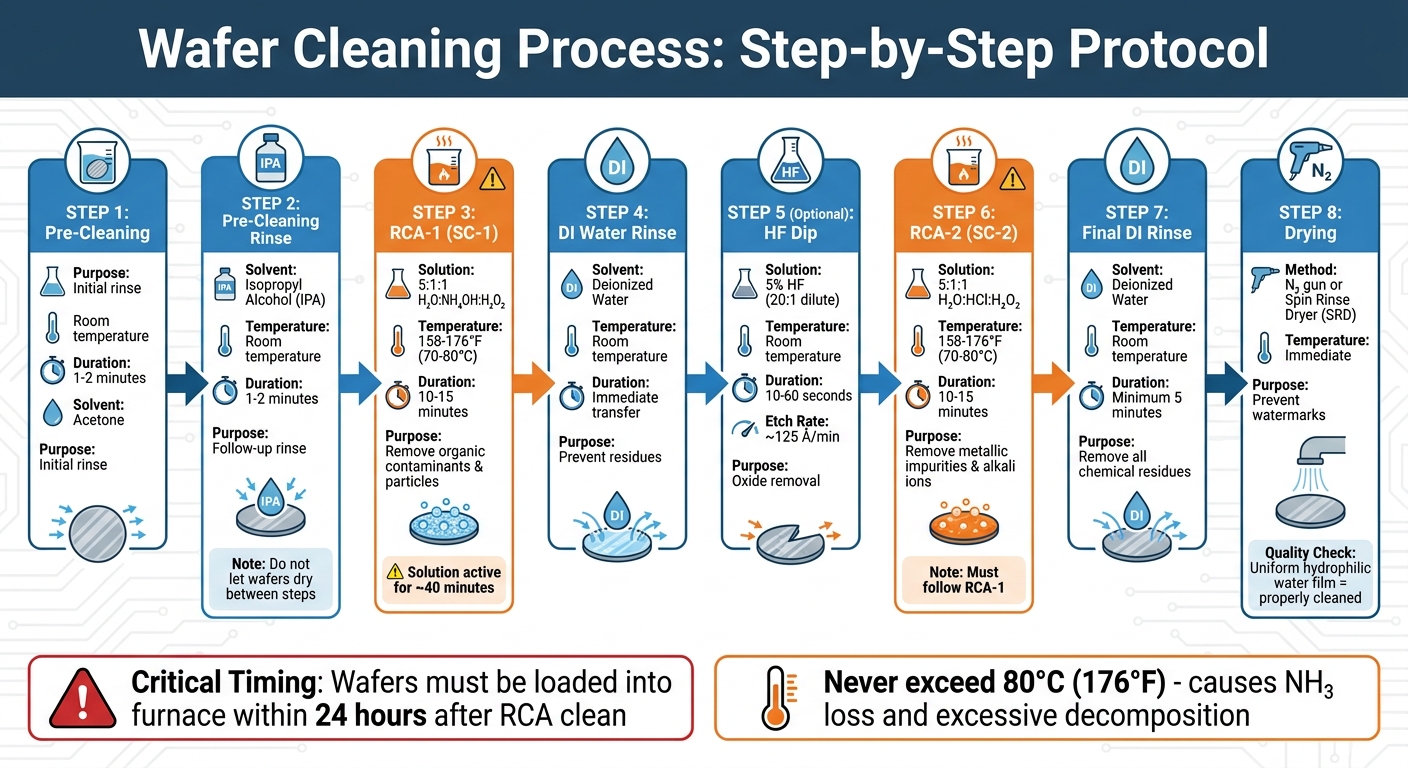
Multi-Step Wafer Cleaning Process with RCA Method
Once you’ve chosen the right solvents, following proper cleaning procedures is key to achieving the best results.
Temperature and Time Settings
Controlling temperature is essential to ensure effective cleaning while protecting wafer surfaces and maintaining the stability of chemical solutions. For pre-cleaning, start with a 1–2 minute acetone rinse at room temperature, immediately followed by a 1–2 minute rinse with IPA. For Piranha cleaning – a mix of sulfuric acid and hydrogen peroxide – temperatures should range from 176–248°F (80–120°C) for 10–20 minutes. RCA-1 (SC-1), which targets organic contaminants and particles, is performed at 158–176°F (70–80°C) for 10–15 minutes. Similarly, RCA-2 (SC-2), designed to remove metallic impurities and alkali ions, uses the same temperature and duration. According to DTU Nanolab:
"A temperature of 70°C is optimal [for RCA cleaning],"
and they caution that:
"RCA solutions should never be heated above 80°C since this will lead to excessive decomposition and loss of NH₃" [4].
For oxide removal, an HF dip is carried out at room temperature for 10–60 seconds, with dilute HF (20:1) etching at a rate of approximately 125 angstroms per minute [3].
By carefully managing temperature and timing, you can ensure effective cleaning while minimizing the risk of damage.
Multi-Step Cleaning Methods
A step-by-step cleaning approach is critical to thoroughly removing all contaminants while preserving wafer integrity. Each step is designed to tackle specific residue types in the correct sequence. For example, RCA-1 must always come before RCA-2 because organic contaminants can interfere with metal removal. RCA-1 uses a 5:1:1 mixture of H₂O, NH₄OH, and H₂O₂ to oxidize and remove light organics, particles, and trace metals. After rinsing with deionized water, RCA-2 follows with a 5:1:1 mixture of H₂O, HCl, and H₂O₂ to dissolve alkali metal ions and metal hydroxides.
If oxide removal is necessary, include a 5% HF dip for 30 seconds between RCA-1 and RCA-2. This step eliminates the oxide layer formed during RCA-1, which can trap contaminants. Keep in mind that RCA solutions degrade quickly once heated to 70°C, with a functional lifespan of about 1 hour due to hydrogen peroxide decomposition. During RCA-1, H₂O₂ remains active for roughly 40 minutes; once it depletes, the solution may begin to etch the silicon [3]. To maintain cleaning efficiency, prepare fresh solutions shortly before use and agitate the wafer carrier 2–3 times to ensure consistent chemical activity.
Rinsing and Drying Methods
Proper rinsing and drying after chemical treatments are crucial to preserve the cleaning results and protect the wafer surface. After each chemical step, immediately transfer wafers to deionized water to prevent permanent residues or watermarks. Avoid letting the wafers dry between acetone, IPA, and DI rinses, as this can cause streaks and residues. For Piranha cleaning, a minimum 5-minute DI water rinse is required [5].
Drying should happen immediately after rinsing. Use a nitrogen (N₂) gun for individual wafers or a Spin Rinse Dryer (SRD) for larger batches. When drying multiple wafers manually with an N₂ gun, keep the remaining wafers submerged in DI water until ready to dry to avoid exposure to air [3]. A properly cleaned wafer will show a uniform, hydrophilic DI water film across its surface rather than water beading [5]. As James Lu from RPI advises:
"Wafers must be loaded into a furnace within 24 hours [after an RCA clean]… If more than 24 hours pass, the wafers must be considered contaminated" [3].
Disclaimer: This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Maintaining Solvent Quality and Solution Life
Ensuring the quality of solvents is just as important as implementing the appropriate cleaning methods. Even the purest solvents degrade over time due to the buildup of contaminants, and using depleted solutions can lead to poor cleaning results or even damage to wafers. To keep cleaning solutions effective, regular monitoring and proper handling are essential. Here’s a closer look at key indicators of solvent degradation and methods to maintain their effectiveness.
Tracking Solvent Degradation
One of the most critical indicators of solvent degradation is the concentration of transition metals, which must remain below parts per billion (ppb) levels [6]. Over time, metals like iron, copper, and nickel can accumulate, potentially recontaminating wafer surfaces. Another key factor is particle loading – solvents gradually become saturated as they remove particles from wafers, reducing their cleaning efficiency [6]. In specialized applications, such as solar cell production, chemical byproducts like phosphosilicate glass residues can alter the solution’s chemistry and diminish its etching capabilities [6].
To assess solvent effectiveness, you can rely on several techniques:
- Contact angle measurements to evaluate surface cleanliness.
- FTIR spectroscopy to detect organic residues.
- Particle counting for gauging contamination levels [1].
Additionally, optical microscopy offers a quick and simple way to check for degradation. If particles, streaks, or residues are visible on cleaned wafers, it’s a sign that the solution is no longer effective [5].
Storage and Handling Guidelines
Proper storage and handling can significantly extend the life of solvents while minimizing contamination risks. Always use dedicated glass or Teflon containers to avoid cross-contamination [5]. Clearly label each container with the chemical name, preparation date, and user initials [5]. Maintaining a Certificate of Analysis (COA) for each batch helps document purity levels and the presence of metallic impurities [1].
A two-stage bath system is an effective way to prolong solvent life. This setup involves:
- A primary bath for removing bulk contaminants.
- A secondary bath with fresh solvent for final cleaning.
When the primary bath becomes saturated, replace it with the secondary bath and refill the secondary with fresh solvent [1]. Additionally, point-of-use filtration can help maintain solvent purity during use [7]. Always handle solvents in a certified chemical fume hood or wet bench to prevent environmental particles from compromising solution quality [5].
When to Replace Cleaning Solutions
Even with proper storage and handling, timely replacement of cleaning solutions is essential. Replacement timing depends on visual inspections, performance tests, and facility-defined age limits. A surface wetting test provides a quick way to check solution effectiveness. A properly cleaned wafer should show a uniform hydrophilic water film; if water beads instead, it’s time to refresh solvents like acetone or IPA [5].
For high-precision cleaning, optical inspections can reveal residual particles. If particles are visible, replace the primary cleaning bath before proceeding to advanced cleaning methods like Piranha or RCA cleaning. Electronics-grade solvents, such as d-Limonene, should maintain metallic impurity levels below 1 ppm and non-volatile residue (NVR) below 5 ppm [1].
When a solution reaches the end of its useful life, follow your facility’s hazardous waste protocols. Be sure to separate organic solvent waste from acidic waste and label waste containers appropriately [5]. These practices not only protect wafer integrity but also ensure reliable cleaning processes.
Disclaimer: This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Safety and Compliance in Solvent Use
Handling solvents in wafer cleaning demands a strict focus on safety and adherence to regulatory guidelines. Mishandling these chemicals can compromise both worker safety and wafer quality. To avoid such risks, protective measures, precise documentation, and the use of high-purity solvents are critical. These practices align closely with established cleaning protocols, ensuring both safety and performance remain uncompromised.
Personal Protective Equipment and Safety Measures
Cleanroom personal protective equipment (PPE) is essential for safe solvent handling. Key items include a cleanroom suit (bunny suit), safety goggles or a face shield, and nitrile gloves [5]. For added protection when working with chemicals like acetone or isopropyl alcohol (IPA), double gloving is recommended [5]. Depending on the solvent in use, additional gear such as chemical-resistant aprons, coveralls, or waterproof boots may be necessary [8].
Before use, always inspect PPE for signs of damage or contamination [8]. Ensure that protective equipment is compatible with the chemicals being handled to avoid degradation or accidental exposure [8]. If the process generates hazardous fumes or dust, use certified respirators, and conduct all chemical cleaning in a chemical fume hood or wet bench to maintain proper ventilation [5]. Always review the Safety Data Sheet (SDS) and chemical labels for specific hazards and precautions. Collect solvent waste in clearly labeled containers, keeping organic solvents separate from acidic waste to meet safety compliance standards [5][8]. Reusable PPE should be cleaned and sanitized after each use, and any damaged equipment must be replaced immediately [8].
In addition to personal safety, maintaining thorough documentation and adhering to regulations ensures the integrity of the entire cleaning process.
Meeting Regulatory Standards and Documentation
Compliance with ISO cleanroom classifications, which regulate particle counts, is critical to maintaining wafer quality [2]. Detailed documentation supports this compliance. Manufacturers should maintain a comprehensive chain-of-custody and quality audit trail, including Certificates of Analysis (COA) and SDS for every solvent batch, to facilitate quality audits and validate processes [1]. Purity verification is just as important – electronic-grade solvents must be monitored for trace metals at parts-per-billion levels, and non-volatile residues (NVR) must stay within acceptable limits [1]. Cleanrooms should maintain overpressure and utilize laminar flow ventilation systems to prevent contamination and effectively manage solvent vapors [2]. Additionally, transitioning from harmful substances like trichloroethylene (TCE) to safer, biodegradable alternatives helps meet evolving safety and environmental standards [1].
Sourcing High-Purity Solvents
The effectiveness of wafer cleaning is directly tied to the quality of the solvents used. Electronics-grade solvents, tested for trace impurities, are essential. Avoid using industrial-grade or food-grade products that have been relabeled [1]. High-purity solvents should have metallic impurity levels below 1 ppm and NVR levels below 5 ppm [1]. Each order should include a COA and SDS for traceability and compliance [1]. Request lot-specific data, including actual test results for the batch, and verify purity through GC-MS data and trace metal analysis at parts-per-billion levels [1].
Partnering with experienced suppliers like Allan Chemical Corporation ensures access to high-purity solvents supported by proper documentation and expert technical assistance. With over 40 years of experience in regulated industries, Allan Chemical Corporation offers technical-grade and compendial-grade solutions (meeting standards such as USP, FCC, ACS, and NF) with just-in-time delivery and competitive pricing tailored for electronic applications. Their dedication to quality aligns seamlessly with the best practices outlined here.
Disclaimer: This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Conclusion
Summary of Best Practices
Using wafer solvents effectively requires attention to detail at every step of the cleaning process. Choosing the right solvent for the specific contaminant is critical. For instance, high-purity d‑Limonene is highly effective at removing hydrocarbon waxes, achieving over 99.9% removal from surfaces with features as small as 500 nm [1]. Process temperatures should be carefully managed to maximize solvent performance while protecting the integrity of materials during cleaning cycles.
A multi-stage cleaning approach enhances results. Implementing chemical dispensing systems for wafer cleaning can further optimize these multi-stage processes through automated delivery. For example, a two-bath system – where the first bath removes bulk contaminants and the second addresses residual traces – extends the life of the cleaning solution and ensures a higher level of cleanliness [1]. Intermediate rinsing is essential to prevent redistribution of contaminants. Since d‑Limonene has a high boiling point of 176°C (349°F) and doesn’t evaporate easily, an intermediate rinse with Isopropyl Alcohol (IPA) is necessary. Water-only rinses can cause the non-polar solvent to bead up, potentially re-depositing contaminants [1]. Final drying should be done using high-purity nitrogen blow-off or vacuum drying at 40–50°C (104°F to 122°F) to eliminate residual films and prevent oxidation. Even a residue as small as 0.01% can compromise metallization and wire bonding, leading to significant losses [1]. This systematic approach ensures wafer integrity throughout manufacturing.
Strict purity standards are equally important. Solvents must meet rigorous purity requirements, with each batch accompanied by a Certificate of Analysis (COA) confirming trace metal content and particle counts. This ensures cleaning processes remain efficient and avoid costly production delays [1]. Following these best practices not only improves cleaning performance but also aids in selecting dependable chemical suppliers.
Working with Reliable Suppliers
Achieving precise cleaning outcomes consistently depends on sourcing high-quality solvents. Allan Chemical Corporation specializes in electronics-grade solvents, drawing on over 40 years of experience in regulated industries. They offer both technical-grade and compendial-grade solutions that meet USP, FCC, ACS, and NF standards. With just-in-time delivery, competitive pricing, and ISO-backed quality, they ensure reliable supply chains. Their technical support team assists with documentation and usage questions, helping ensure compliance and effectiveness in wafer cleaning processes. Partnering with a supplier who understands the precision required in semiconductor fabrication – where contaminants are measured at the nanoscale – helps safeguard product quality and operational efficiency.
Disclaimer: This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
FAQs
How do I choose the right solvent for my wafer contamination?
When selecting a solvent for cleaning, it’s crucial to match it to the specific contaminants you’re addressing and ensure it’s compatible with your cleaning process. For cleaning silicon wafers, acetone, methanol, and isopropyl alcohol are commonly used in sequence, as each is effective against different types of residues. If you’re dealing with organic residues, d-Limonene works well for tasks like dewaxing.
Always opt for high-purity solvents, such as semiconductor-grade, to maintain process integrity. For tailored solutions, consider reaching out to suppliers like Allan Chemical Corporation, who can offer guidance based on your specific needs.
How can I tell when a solvent bath is too contaminated to use?
When a solvent bath becomes excessively contaminated, it means the levels of impurities have surpassed acceptable limits. This can compromise both the integrity of materials and the effectiveness of cleaning processes. To determine if contamination has reached this critical point, techniques like SME-ICP-MS (Surface Metal Enrichment Inductively Coupled Plasma Mass Spectrometry) are used. These methods measure contamination concentrations to ensure they remain within safe thresholds.
What’s the safest way to rinse and dry wafers without leaving residue?
To ensure wafers are rinsed and dried without residue, follow these steps carefully. Begin by rinsing the wafers with deionized (DI) water for approximately 1 minute to eliminate any solvent residues. After rinsing, dry the wafers using a nitrogen gun to avoid water spots. Always perform these tasks in a cleanroom environment while wearing appropriate personal protective equipment (PPE) to reduce the risk of contamination. By adhering to these practices, you can achieve a clean, residue-free surface ready for further processing.





Comments are closed