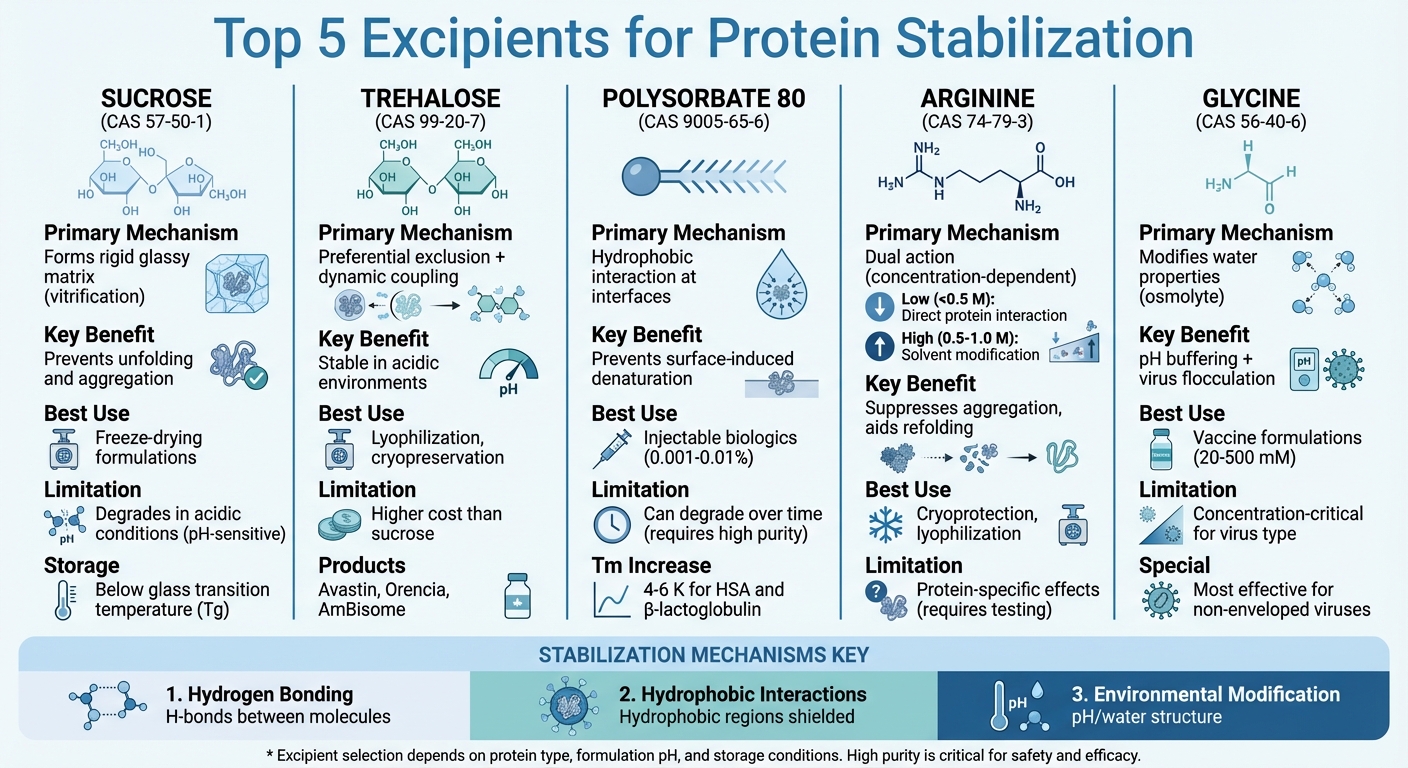
Protein-based drugs like vaccines and monoclonal antibodies are sensitive to instability, which can lead to aggregation, denaturation, and loss of potency. To combat these issues, excipients – substances added to formulations – play a key role in stabilizing proteins during production, storage, and transport. Below are five excipients and how they work:
- Sucrose (CAS No. 57-50-1): Stabilizes proteins by forming a rigid, glassy matrix that prevents unfolding and aggregation. Common in freeze-drying formulations, it extends shelf life but may degrade in acidic conditions.
- Trehalose (CAS No. 99-20-7): A non-reducing sugar that protects proteins through hydration and forms a stable glassy matrix. It’s effective in acidic environments and widely used in lyophilization.
- Polysorbate 80 (CAS No. 9005-65-6): A surfactant that prevents protein aggregation by binding to hydrophobic regions and protecting against surface-induced denaturation. Its purity is critical to avoid degradation over time.
- Arginine (CAS No. 74-79-3): Suppresses aggregation by interacting with hydrophobic regions and modifying solvent properties. Its stabilizing effects vary with concentration and protein type.
- Glycine (CAS No. 56-40-6): Modifies water properties to enhance protein stability and acts as a buffering agent. It’s particularly useful in freeze-drying and vaccine formulations.
These excipients help maintain protein integrity, extend product shelf life, and improve formulation stability. Proper sourcing of high-purity materials is essential to ensure safety and effectiveness.
Top 5 Excipients for Protein Stabilization: Mechanisms and Applications
Mechanisms of aggregation and stabilisation
sbb-itb-aa4586a
1. Sucrose
Sucrose (CAS No. 57-50-1) is commonly used as an excipient in protein therapeutics. This disaccharide plays a key role in stabilizing proteins during manufacturing, storage, and distribution through several molecular mechanisms.
Molecular Interaction with Proteins
Sucrose interacts with proteins primarily through preferential exclusion. This means sucrose molecules are largely kept away from the protein surface, which helps preserve the protein’s native hydration layer [1]. By doing so, sucrose thermodynamically favors the protein’s compact, folded state over its unfolded form. In freeze-drying or low-moisture conditions, sucrose takes the place of water molecules by forming direct hydrogen bonds. This substitution helps maintain the protein’s native structure and reduces its mobility during storage [2].
"Sugars should be able to adequately form interactions with the protein during drying, thereby maintaining it in its native conformation and reducing both local and global mobility during storage."
- Maarten A. Mensink, University of Groningen [2].
Stabilization Mechanism
Sucrose stabilizes proteins by forming a rigid, glassy matrix – a process known as vitrification. This glassy state immobilizes proteins and prevents them from unfolding or aggregating. With its high viscosity, the matrix effectively restricts molecular-level protein movement. According to Christoffer Olsson from Chalmers University of Technology:
"Storing proteins below $T_g$ stabilizes the protein by preventing these conformational changes to occur" [1].
Additionally, sucrose slows down solvent relaxation, further suppressing protein motion [1]. These combined effects make sucrose highly effective at stabilizing proteins under various storage conditions.
Compatibility with Biopharmaceutical Formulations
Sucrose works well with most protein formulations and is included in compendial monographs for pharmaceutical use. Satish K. Singh from Lonza AG explains:
"Sucrose can generally be considered as suitable in most cases, unless constrained by a low-pH formulation" [3].
However, caution is needed with acidic formulations. At low pH, sucrose can undergo acid-catalyzed hydrolysis, breaking down into glucose and fructose. These reducing sugars can react with proteins via glycation or the Maillard reaction, potentially affecting shelf life [3]. Despite this limitation, sucrose remains a critical tool for creating stable biopharmaceutical formulations.
Impact on Protein Shelf Life
Sucrose significantly enhances protein shelf life by slowing degradation at the molecular level. In sucrose–protein systems, the water β-relaxation process has an activation energy of approximately 0.55 eV, and protein stability in the glassy state is closely tied to these water dynamics [1]. To maximize shelf life, formulations should be stored below the glass transition temperature ($T_g$) of the sucrose–protein matrix. Additionally, sucrose’s smaller molecular size, compared to polysaccharides, allows it to pack efficiently around proteins. This dense packing reduces local mobility more effectively, further stabilizing the protein [2].
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
2. Trehalose
Trehalose (CAS No. 99-20-7) is a non-reducing disaccharide widely recognized for its role in stabilizing protein therapeutics. It is a key ingredient in several biopharmaceutical products, such as Avastin (bevacizumab), Orencia (abatacept), and AmBisome (amphotericin B) [3]. This sugar is particularly valued for its ability to improve formulation stability.
Molecular Interaction with Proteins
Trehalose stabilizes proteins through a process known as preferential exclusion. This means proteins are surrounded by a layer of water rather than directly interacting with the sugar. In trehalose solutions, the hydration shell around the protein is significantly stronger, with protein–water interactions (451) far outnumbering protein–sugar interactions (9.63) [4]. This "water bridge" effect helps maintain the protein’s natural structure.
"The hydration layer around protein molecules maintains a hydration bridge between the sugar and the protein."
- Kajsa Ahlgren et al., Division of Nano-Biophysics, Chalmers University of Technology [4]
Trehalose also supports protein stability through dynamic mechanisms, which complement its molecular interactions.
Stabilization Mechanism
Trehalose achieves protein stabilization by both preferential exclusion and dynamic coupling. It discourages denaturation and slows internal protein dynamics, favoring the protein’s native state [4]. This stabilization occurs because trehalose strongly couples with water, slowing the motion of the hydration layer. This reduced hydration layer motion, in turn, limits the internal movement of the protein.
"The better stabilizing properties of trehalose are then assumed to be due to a stronger dynamic coupling of trehalose to water, which, in turn, leads to a stronger reduction of the protein dynamics, since the protein dynamics is ‘slaved’ by the dynamics of the hydration layer."
- Kajsa Ahlgren et al., Physical Chemistry Chemical Physics [4]
In freeze-dried formulations, trehalose forms a glassy matrix with a high glass transition temperature (T₉). This matrix creates a rigid environment that restricts molecular movement, preventing protein unfolding or aggregation.
Compatibility with Biopharmaceutical Formulations
Trehalose’s compatibility with biopharmaceutical formulations further enhances its stabilizing properties. Unlike sucrose, which is prone to acid-catalyzed hydrolysis, trehalose remains stable in acidic environments, making it suitable for low-pH conditions [3]. Its high T₉ and stability under acidic conditions make it particularly effective in lyophilization and cryopreservation applications [3].
| Interaction Parameter | Trehalose System | Sucrose System |
|---|---|---|
| Protein–Water Coordination | 451 | 338 |
| Protein–Sugar Coordination | 9.63 | 15.5 |
| Sugar–Water Coordination | 19.3 | 23.9 |
Impact on Protein Shelf Life
The molecular and formulation benefits of trehalose directly contribute to extending the shelf life of protein therapeutics. By slowing protein dynamics and maintaining structural integrity, trehalose ensures long-term stability during storage. Additionally, the glassy matrix formed during lyophilization minimizes degradation and aggregation risks. To maximize stability, formulations should be stored below the glass transition temperature of the trehalose-protein matrix.
As Jan Swenson from Chalmers University of Technology explains:
"Trehalose is often considered as one of the most efficient and versatile stabilizing co-solutes, since it has shown a large ability to preserve the properties of proteins and many other biological molecules after different types of storages."
- Jan Swenson, Professor, Chalmers University of Technology [4]
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
3. Polysorbate 80
Polysorbate 80 (CAS No. 9005-65-6) is a non-ionic surfactant commonly used in protein formulations to prevent aggregation and surface denaturation. It plays a crucial role in stabilizing proteins by targeting hydrophobic patches, helping to maintain their structure. Typically, it is effective at concentrations ranging from 0.001% to 0.01% in biopharmaceutical formulations [5]. Its ability to combine interface protection with direct molecular interaction makes it especially useful for injectable biologics.
Molecular Interaction with Proteins
Polysorbate 80 interacts with proteins through hydrophobic interactions, hydrogen bonding, and van der Waals forces [6]. These interactions allow the surfactant to bind to hydrophobic regions on protein surfaces, reducing the likelihood of self-association. However, the strength and specificity of these interactions vary based on the protein. For instance, studies using isothermal titration calorimetry show that Polysorbate 80 binds to Human Serum Albumin (HSA) at a ratio of approximately 3:1 (surfactant to protein), with dissociation constants ranging from 429 to 653 μM [6]. On the other hand, it exhibits minimal interaction with Immunoglobulin G (IgG) due to the lack of exposed hydrophobic regions [6].
The table below highlights the protein-specific behavior of Polysorbate 80:
| Protein | Binding Stoichiometry (PS80:Protein) | Dissociation Constant ($K_d$) | $T_m$ Increase |
|---|---|---|---|
| Human Serum Albumin (HSA) | ~3:1 | 429–653 μM | 4–6 K |
| β-lactoglobulin | ~3:2 | 284–388 μM | 4–6 K |
| Immunoglobulin G (IgG) | Negligible | N/A | 0 K |
Stabilization Mechanism
Polysorbate 80 stabilizes proteins by adsorbing at interfaces such as air-water or solid-liquid boundaries, effectively outcompeting proteins for these surfaces. This prevents surface-induced denaturation [5][6][7]. Additionally, it enhances thermal stability by increasing the unfolding temperature ($T_m$) by 4–6 K, which may help proteins maintain their folded structure or refold when stressed [6].
Compatibility with Biopharmaceutical Formulations
The compatibility of Polysorbate 80 depends on its purity. Standard Polysorbate 80 NF contains at least 58% oleic acid, while high-purity grades exceed 98% oleic acid content [5]. Studies on Rituximab (10 mg/mL) and recombinant human Granulocyte-Colony Stimulating Factor (rhG-CSF, 0.6 mg/mL) have shown that high-purity Polysorbate 80 reduces particulate formation compared to standard grades [5].
Impact on Protein Shelf Life
Polysorbate 80 also plays a role in extending the shelf life of protein-based drugs. By improving thermal stability and preventing aggregation during storage, it helps maintain product integrity. However, the surfactant can degrade over time due to hydrolytic cleavage by esterases or autoxidation, releasing free fatty acids that may form visible or sub-visible particulates in injectable formulations [7][5]. High-purity grades, with lower peroxide values (maximum 2.0 meq O₂/kg) and fewer impurities, are more resistant to degradation and help mitigate these risks [6].
By targeting hydrophobic regions and protecting against surface-induced aggregation, Polysorbate 80 illustrates how carefully selected excipients can enhance protein stability at the molecular level.
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
4. Arginine
Arginine (CAS No. 74-79-3) is an amino acid excipient commonly used in biopharmaceutical formulations for its ability to suppress aggregation, aid in protein refolding, and act as a cryoprotectant during lyophilization. Unlike simple sugars or surfactants, arginine operates through a dual mechanism that changes with concentration, offering unique benefits for stabilizing therapeutic proteins and vaccines. While its molecular interactions are similar to those of sugars and surfactants, arginine’s dual approach provides additional flexibility for formulators.
Molecular Interaction with Proteins
Arginine stabilizes proteins through several molecular interactions, such as cation-π interactions, salt-bridge formation, and binding to hydrophobic regions [8][9]. Additionally, arginine molecules self-associate via head-to-tail hydrogen bonding, which reduces protein-protein interactions by forming stronger arginine-arginine bonds than arginine-water bonds [8].
The mechanism of action shifts with concentration. At low concentrations (below 0.5 M), arginine interacts directly with protein side chains. At higher concentrations (0.5–1.0 M), it influences the solvent structure instead, demonstrating the importance of tailoring arginine levels for specific proteins [11].
Stabilization Mechanism
Arginine primarily prevents protein aggregation by slowing down the association of protein molecules, rather than altering the folding pathway directly [10]. Acting as a neutral crowder, it increases the free energy required for aggregation and stabilizes intermediate protein states [8].
At elevated concentrations, arginine modifies the solvent environment to favor proper protein folding. Simulations have shown that its surfactant-like behavior, driven by preferential adsorption at the protein-solvent interface, distinguishes it from other amino acid excipients [11].
Compatibility with Biopharmaceutical Formulations
While arginine is often used as a general stabilizer, its effects can vary depending on the protein. For instance, it stabilizes Hen Egg White Lysozyme (HEWL) at concentrations between 0.1 M and 2.0 M. However, its interaction with other proteins depends on their surface chemistry [9][11]. For example, arginine has been observed to reduce the stability of Porcine Parvovirus (PPV) by altering charged interactions, leading to a lower viral titer during storage at 39°F or when exposed to heat stress at 140°F [11].
These context-dependent effects highlight the importance of compatibility testing for each protein. Formulators should evaluate a range of arginine concentrations, as its mechanism transitions from direct interaction to solvent modulation at higher levels. Combining arginine with other amino acids, such as glutamic acid, may further enhance solubility and stability [8].
Impact on Protein Shelf Life
Arginine helps extend the shelf life of proteins by preventing irreversible aggregation and maintaining structural integrity during lyophilization [10][11]. Studies have shown that arginine improves the thermal stability of HEWL by raising its hydrophobic exposure temperature (HET), a measure of protein stability under heat. Since arginine’s effectiveness depends on both the protein’s surface properties and the excipient concentration, it is critical to validate its use for each therapeutic protein.
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
5. Glycine
Glycine offers a distinct method for stabilizing proteins by influencing water properties instead of directly binding to the proteins themselves. This small, uncharged amino acid (CAS No. 56-40-6) is particularly useful for preserving protein stability during storage, transportation, and freeze-drying processes.
Molecular Interaction with Proteins
Rather than directly interacting with proteins, glycine enhances protein stability by modifying the behavior of water. It increases water’s surface tension, reinforcing the hydration shell around proteins and reducing the risk of aggregation. As a "compatible solute", glycine supports protein stability without significantly altering the formulation’s dielectric constant.
Stabilization Mechanism
Glycine stabilizes proteins through two main mechanisms. First, it acts as a buffering agent, helping to maintain a stable pH and prevent protein denaturation. Second, it serves as an osmolyte, offering protection against environmental stresses such as temperature changes, light exposure, and chemical reactions.
In vaccine development, glycine plays a unique role in purifying and flocculating organisms, particularly non-enveloped viruses. It is considered the most effective osmolyte for flocculating non-enveloped viruses, though care must be taken with concentration levels, as the same can inactivate enveloped viruses.
Compatibility with Biopharmaceutical Formulations
Glycine is typically used in concentrations ranging from 20 to 500 mM in protein solutions. For flocculation purposes, the concentration must be carefully adjusted based on whether the target is an enveloped or non-enveloped virus. Its effectiveness depends on the solution’s pH in relation to the virus’s isoelectric point.
A study conducted in January 2021 by Ghazal Karevan and colleagues evaluated a brucellosis vaccine candidate using glycine nanoparticles as both an adjuvant and a delivery system. The research involved a chimeric antigen (Omp31 and Bp26) loaded onto glycine nanoparticles, which successfully triggered cellular and humoral immune responses in a mouse model [12].
This water-based stabilization complements the action of other excipients that interact directly with proteins, offering a broader approach to maintaining protein integrity.
Impact on Protein Shelf Life
Glycine helps extend the shelf life of proteins by preserving their potency and functionality during storage and transportation. Derivatives like glycine betaine provide additional benefits, such as stabilizing protein quaternary structures under thermal and oxidative stress. Farwa Basit from the Department of Biology at Wenzhou-Kean University highlights this benefit:
"GB [glycine betaine] effectively stabilizes the quaternary structure of proteins and enzymes… helping to stabilize the cellular structures, particularly membranes and proteins, under conditions of high salinity and temperature stress" [13].
By protecting proteins from reactive oxygen species (ROS) and ensuring continued protein synthesis, glycine-based stabilizers maintain the functionality of protein complexes even under challenging conditions.
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
How Excipients Interact with Proteins at the Molecular Level
The ways excipients interact with proteins highlight the mechanisms behind protein stabilization. These interactions primarily involve hydrogen bonding, hydrophobic effects, and changes to the surrounding environment.
Hydrogen Bonding
Hydrogen bonding is critical for stabilizing proteins in dried formulations. During processes like lyophilization or spray-drying, water molecules that typically interact with protein surfaces are removed. Excipients such as trehalose step in to fill this gap by forming hydrogen bonds with the protein, preserving its structure. Research in Molecular Pharmaceutics illustrates this:
"Trehalose remained amorphous after spray-drying and was miscible with BSA, forming hydrogen bonds to maintain protein conformation" [14].
However, excipients prone to crystallization may not provide the same level of protection, as they fail to establish a robust hydrogen bonding network.
Hydrophobic Interactions
Hydrophobic interactions also play a significant role in protein stability. Proteins naturally fold in a way that buries their hydrophobic amino acid residues, shielding them from water. This phenomenon, often referred to as the "hydrophobic effect", is described in RSC Advances:
"In the folded protein, hydrophobic amino acid residues are buried in the protein avoiding contact with water" [15].
When proteins fold properly, hydrophobic regions collapse inward, releasing structured water into the surrounding solvent and increasing entropy. However, if a protein unfolds, these exposed hydrophobic regions can lead to self-association and irreversible aggregation.
Environmental Modification
Excipients can also stabilize proteins by altering their microenvironment. Osmolytes and chemical chaperones, such as glycine betaine, are often excluded from the protein’s hydration shell. This exclusion makes the unfolded protein state less favorable thermodynamically. Additionally, buffers help regulate pH, which directly affects the protonation of amino acid residues. This, in turn, impacts their ability to form hydrogen bonds and electrostatic interactions [15].
Proteins are inherently delicate, with what scientists describe as "marginal" stability [15]. This sensitivity means they are highly responsive to shifts in concentration, ionic strength, temperature, and pH. Understanding these molecular interactions is essential for designing effective formulations and aligns with the sourcing and formulation strategies discussed in the following sections.
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Sourcing High-Purity Excipients with Allan Chemical Corporation
When working with protein-based biopharmaceuticals, the quality of excipients plays a crucial role in ensuring effective protein stabilization. Impurities or inconsistencies in these substances can lead to protein aggregation, increasing the risk of immunogenicity and reducing therapeutic effectiveness. To maintain consistent and predictable molecular interactions, sourcing high-purity excipients is non-negotiable.
With over 40 years of experience, Allan Chemical Corporation supports regulated industries like pharmaceuticals by providing excipients that meet stringent compendial standards, including USP, FCC, ACS, and NF. These high-quality solutions are critical for maintaining the precise formulation parameters required by sensitive biologics. Their expertise ensures excipients are suitable for demanding applications where even minor deviations can have significant consequences.
In addition to prioritizing quality, Allan Chemical Corporation emphasizes reliability. They offer just-in-time delivery, flexible batch sizes, and strong manufacturer partnerships. Their responsive technical support and easy access to Certificates of Analysis and Safety Data Sheets simplify compliance and streamline production processes. Whether you need trehalose for lyophilization, polysorbate 80 for surfactant protection, or arginine to prevent aggregation, their sourcing-first approach ensures both quality and cost-efficiency.
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
Conclusion
Protein therapeutics require careful stabilization to maintain their effectiveness throughout manufacturing, storage, and delivery. Compounds like sucrose and trehalose play a key role by forming hydrogen bonds that help proteins retain their native structure. Arginine prevents aggregation by interacting with aromatic hydrophobic regions, while glycine offers broad stabilization benefits. Polysorbate 80 provides additional protection against surface-induced denaturation at air-water interfaces and container surfaces, addressing mechanical stresses that sugars alone cannot handle.
Molecular interactions are at the heart of these stabilization strategies. For instance, trehalose forms an amorphous, miscible matrix that reduces monomer loss in spray-dried proteins [14]. This miscibility is crucial because excipients that crystallize can cause phase separation, undermining their stabilizing function. Since proteins are highly sensitive to their formulation environment, even small amounts of impurities can lead to aggregation, increasing the risk of immunogenicity and the formation of anti-drug antibodies [15]. These factors highlight the importance of using excipients with high purity to ensure the stability and safety of protein formulations.
The quality of excipients directly affects whether a protein formulation meets strict regulatory standards. Contaminants can compromise both safety and efficacy, making it essential to source high-purity, compendial-grade materials. Reliable suppliers who provide proper documentation and adhere to regulatory requirements enable formulation scientists to create stable and effective therapeutic proteins. Allan Chemical Corporation, with over 40 years of experience, supports these needs by offering compendial-grade solutions (USP, FCC, ACS, NF) tailored for regulated pharmaceutical applications.
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
FAQs
How do I choose the right excipient for my protein?
To choose the best excipient, focus on your protein’s stabilization requirements and how it interacts at the molecular level. Popular options include polyols like glycerol, mannitol, and sorbitol, which enhance thermal stability and provide bulk. Sugars such as sucrose and trehalose are often used for cryoprotection, while amino acids like arginine and glycine can help control viscosity and reduce aggregation. Testing excipients within your specific formulation conditions is crucial to identifying the most suitable stabilizers.
When should I use trehalose instead of sucrose?
Trehalose is often chosen over sucrose when there’s a need for stronger protein stabilization, especially during stress conditions like dehydration or exposure to extreme temperatures. This is because trehalose forms more robust molecular interactions, such as hydrogen bonds, which help maintain the structure and function of proteins more effectively. While sucrose can handle general stabilization needs, trehalose proves to be the better option for ensuring stability over extended periods or in challenging environments.
What makes polysorbate 80 “high purity,” and why does it matter?
Polysorbate 80 achieves "high purity" status when it complies with pharmacopeial standards and has very low levels of impurities, such as peroxides and reactive degradation products. Maintaining this purity is essential to ensure the stability and safety of pharmaceutical formulations, as impurities can negatively impact product effectiveness and pose risks to patient health.





Comments are closed