Excipients are no longer just fillers in pharmaceuticals – they now help solve major challenges like drug solubility, safety, and stability. These substances reduce reliance on toxic solvents, which have long been a concern due to health risks, regulatory restrictions, and waste generation. For example, Cyclodextrins (CAS No. 7585-39-9) create water-soluble complexes with poorly soluble drugs, while Polymers like Hydroxypropyl Methylcellulose prevent crystallization, ensuring consistent drug performance.
Here’s why excipients matter:
- Safer Alternatives: They eliminate residual solvent toxicity, reducing risks for patients and manufacturers.
- Improved Drug Solubility: Many drugs (90% of new APIs) struggle with poor water solubility; excipients tackle this directly.
- Lower Environmental Impact: Solvent use generates up to 90% of pharmaceutical waste, while excipients provide cleaner options.
- Regulatory Compliance: Excipients meet strict standards, simplifying drug approvals.
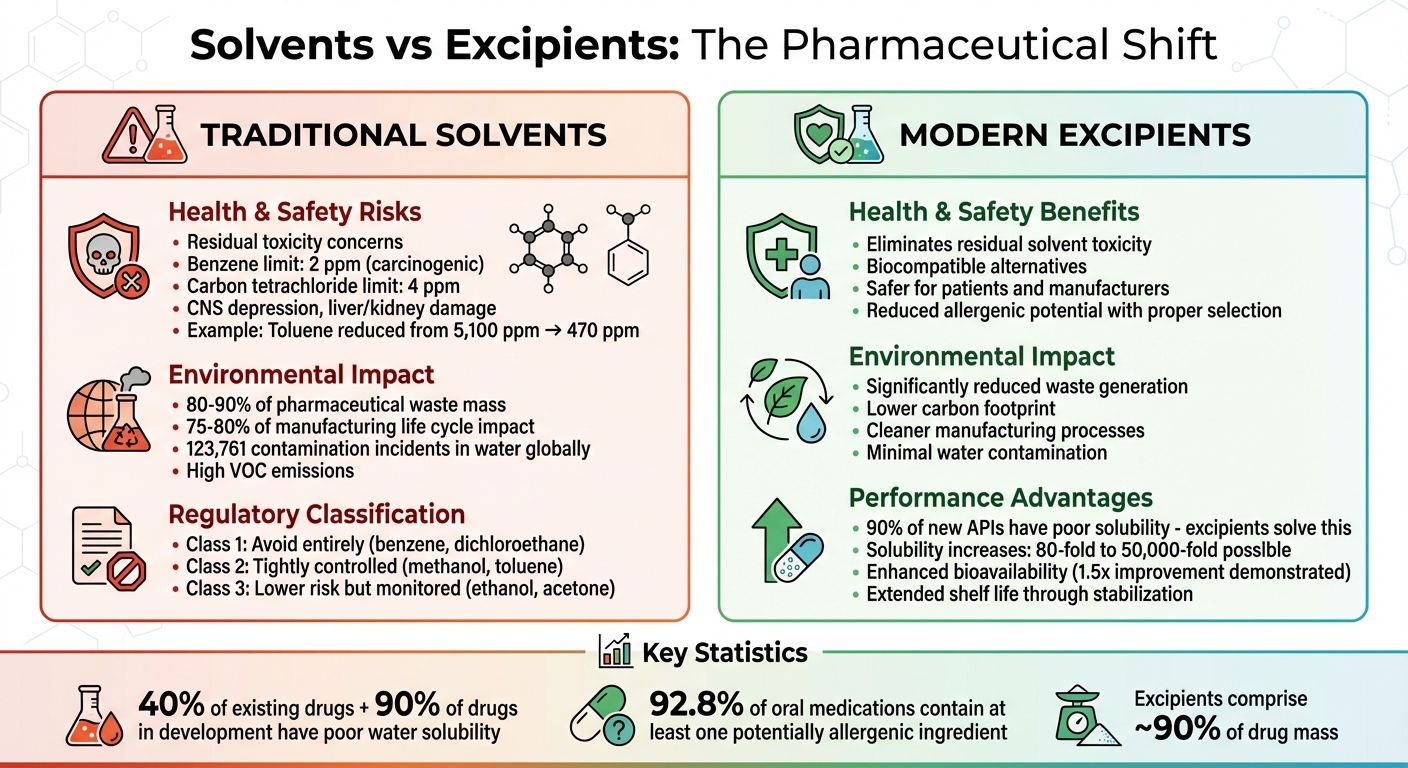
Solvents vs Excipients in Drug Formulation: Safety, Environmental Impact, and Regulatory Comparison
Introduction to Pharmaceutical Excipients
sbb-itb-aa4586a
Problems with Solvents in Drug Formulation
Organic solvents have long been a cornerstone in pharmaceutical manufacturing, but their use comes with serious concerns. These range from health risks to regulatory hurdles and environmental challenges. Below, we break down these issues to highlight why formulators are seeking safer alternatives.
Health and Safety Risks
Residual solvents in drugs can pose serious health hazards. These chemicals can enter the body through inhalation, skin contact, or ingestion, potentially leading to harmful effects if they reach the bloodstream [7][8]. For example, solvent vapors can depress the central nervous system, causing anything from mild headaches and dizziness to severe effects like unconsciousness [6][8].
Long-term exposure introduces even graver risks. Certain solvents, like benzene and carbon tetrachloride, are known carcinogens and can cause irreversible liver and kidney damage over time [6][7][9]. Regulatory limits reflect these dangers: the FDA restricts benzene residues to 2 ppm and carbon tetrachloride to 4 ppm due to their toxicity [9]. Other solvents, such as hexane and methyl n-butyl ketone, can damage peripheral nerves, leading to motor impairments or permanent numbness [7].
"Health hazards associated with solvent exposure include toxicity to the nervous system, reproductive damage, liver and kidney damage, respiratory impairment, cancer, and dermatitis." – OSHA [6]
One example of mitigating these risks comes from a U.S.-based API manufacturer that reduced toluene levels in its product from 5,100 ppm to 470 ppm by modifying vacuum drying processes. This adjustment not only improved safety but also enabled regulatory approval for a New Drug Application. While health risks are a critical concern, solvents also bring significant regulatory and environmental challenges.
Regulatory and Environmental Issues
The FDA categorizes solvents into three risk-based groups:
- Class 1 solvents (e.g., benzene, 1,2-dichloroethane): These should be avoided entirely due to their toxicity and environmental harm.
- Class 2 solvents (e.g., methanol, toluene, acetonitrile): Use of these is permitted but must be tightly controlled.
- Class 3 solvents (e.g., ethanol, acetone): These pose lower toxicity risks but still require quality oversight [9].
"Solvents in Class 1 should not be employed in the manufacture of drug substances, excipients, and drug products because of their unacceptable toxicity or their deleterious environmental effect." – FDA Q3C Guidance [9]
The environmental impact of solvent use is staggering. Solvents account for 80–90% of waste mass in pharmaceutical production and contribute 75–80% of the life cycle impact of manufacturing processes [12]. Volatile Organic Compound (VOC) emissions are a major regulatory focus, prompting manufacturers to adopt recovery and reuse systems instead of traditional solvent destruction methods to reduce greenhouse gas emissions [11]. A global survey reported 123,761 incidents of pharmaceutical residues in sewage and surface water, highlighting the widespread contamination caused by solvent use [10]. Regulatory bodies, particularly in the EU, are tightening rules to address these environmental concerns, from production to disposal [10].
These issues emphasize the importance of finding safer, non-toxic alternatives, like excipients, to replace traditional solvents in drug formulation.
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
How Excipients Work as Solvent Replacements
Excipients provide pharmaceutical formulators with safer alternatives to toxic organic solvents by improving solubility and stability in drug formulations. By understanding their mechanisms, manufacturers can create effective products that meet stricter safety standards. Below, we dive into the types of excipients, their functions, and the testing required to ensure their compatibility with active pharmaceutical ingredients (APIs).
Common Excipient Types
Different excipients tackle solubility challenges using unique chemical and physical properties:
- Cyclodextrins: These cyclic oligosaccharides feature a hydrophobic inner cavity and a hydrophilic outer surface. They encapsulate poorly soluble drug molecules, forming water-soluble complexes. For example, Hydroxypropyl-β-cyclodextrin (HP-β-CD) and Sulfobutylether-β-cyclodextrin (SBE-β-CD) are widely used in injectable formulations due to their reduced kidney toxicity [2][13].
- Surfactants and Amphiphilic Polymers: These reduce surface tension and form micelles at critical concentrations. Examples include Poloxamers, polysorbates like Tween 80, and poly(amino acid) polymers. Notably, Apisolex™ polymer has been shown to increase the solubility of hydrophobic APIs by up to 50,000-fold, offering a safer alternative to PEG-based enhancers for injectable drugs [14].
- Polymers for Solid Dispersions: Polymers such as polyvinylpyrrolidone (PVP), hydroxypropyl methylcellulose (HPMC), and hydroxypropyl methylcellulose acetate succinate (HPMCAS) keep drugs in an amorphous state, preventing the formation of slow-dissolving crystals.
- Lipid-Based Systems: Medium-chain triglycerides and phospholipids are used to create emulsions or lipid nanoparticles that encapsulate hydrophobic drugs.
- pH Modifiers: Compounds like meglumine, citric acid, and sodium carbonate adjust the local environment around drug particles to enhance ionization, boosting solubility for weak acids and bases [2][13][18].
Each excipient type employs a distinct strategy to improve solubility, ensuring flexibility in formulation design.
How Excipients Function
The mechanisms behind excipient functionality directly address safety and environmental concerns associated with traditional solvents:
- Molecular Encapsulation: Cyclodextrins encapsulate non-polar APIs within their hydrophobic cavity, making them water-soluble. For example, complexing ibuprofen with HP-β-CD at a 1:10 ratio increased its solubility 80-fold at pH 5.0 [13]. Similarly, SBE-β-CD has enhanced itraconazole solubility by up to 125-fold [13].
- Amorphization: Solid dispersions disrupt the crystalline structure of drugs, keeping them in a more soluble, high-energy state. Studies show that using HPMC-AS matrices for posaconazole formulations improves both solubility and pH-independent absorption [15]. Hemlata Patil from Catalent explained:
"The molten polymer should have a high capacity for solubilizing the drug, which can be especially beneficial when the drug’s melting point or decomposition temperature is a concern" [16].
- Micellar Solubilization: Amphiphilic block copolymers self-assemble into micelles, where hydrophobic drug molecules are sequestered in the core while hydrophilic portions interact with water. This approach offers a safer profile compared to small-molecule surfactants like Cremophor EL, which are associated with hypersensitivity reactions [14][18].
- Cosolvency and pH Modulation: Water-miscible organic excipients like PEG 400 or acidic/basic additives adjust the polarity or ionization state of the API, improving solubility [13][15][17]. In some cases, in-situ salt formation occurs during processes like hot-melt extrusion, where excipients react with the API to form soluble salts directly within the formulation matrix [16].
Testing Drug-Excipient Compatibility
Ensuring the success of a formulation requires rigorous testing to confirm miscibility, stability, and compatibility between the drug and excipients. Key considerations include:
- Crystallization Inhibition: Tests like Differential Scanning Calorimetry (DSC) and X-ray Powder Diffraction (XPRD) verify that excipients prevent recrystallization, maintaining the drug’s amorphous state.
- Chemical Stability: Compatibility testing identifies potential chemical interactions, such as hydrolysis, oxidation, or Maillard reactions, which could compromise the drug’s stability.
- Solubility Optimization: For cyclodextrin complexes, solubility phase diagrams help determine the ideal API-to-excipient ratio, maximizing solubility without wasting materials.
These tests ensure that the final formulation delivers consistent drug concentration, dissolution rates, and shelf life, all of which are critical for regulatory approval and patient safety.
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Factors to Consider When Choosing Excipients
Choosing the right excipient to replace a solvent is a balancing act involving patient safety, regulatory demands, and cost considerations. Unlike active pharmaceutical ingredients (APIs), excipients are functional components that interact with the drug, the manufacturing process, and the patient. A poor choice can lead to adverse reactions, production issues, or regulatory hurdles, all of which can have serious consequences for both patients and manufacturers.
Safety and Toxicity
Excipients, although labeled "inactive", can cause unexpected reactions. Since they often make up about 90% of a drug’s total mass, their safety profile is critical. Alarmingly, 92.8% of oral medications contain at least one ingredient with allergenic potential [5]. This highlights the need for thorough safety evaluations beyond simply checking if an excipient is on an approved list.
Certain excipients pose higher risks for specific populations. For instance, neonates are particularly vulnerable to substances like benzyl alcohol and propylene glycol. Back in the 1980s, trace hydrazine contamination in povidone – a commonly used binder – brought attention to the dangers of toxic byproducts. This discovery led to the establishment of strict hydrazine limits in pharmacopeial monographs [5].
Chemical incompatibilities are another concern. For example, the Maillard reaction can occur between APIs with amine groups and reducing sugars like lactose. Similarly, APIs sensitive to oxidation require excipients with low peroxide levels, such as high-purity povidone or polysorbates.
Toxicity risks also vary by administration route. While Vitamin E is considered safe for oral use, it has been linked to lung toxicity when inhaled. Additionally, lactose intolerance affects 65–80% of the global adult population, with 30–50 million people in the U.S. alone requiring lactose-free alternatives [5]. As Dorothy Chang, MD, and Rong-Kun Chang, PhD, stated:
"Excipients are not completely inert. Even commonly used excipients that are deemed to be pharmaceutically inactive and nontoxic may cause adverse reactions" [20].
| Excipient | Potential Safety/Toxicity Concern | Common Alternative |
|---|---|---|
| Lactose | Gastrointestinal distress in intolerant patients; Maillard reaction with amines | Mannitol, Dextrates, Microcrystalline Cellulose |
| Benzyl Alcohol | Toxic to neonates; systemic allergic reactions | Preservative-free single-dose vials |
| Tartrazine (Yellow 5) | Urticaria (hives) and asthma aggravation | Natural colorants or dye-free formulations |
| Wheat Starch | Gluten sensitivity in Celiac patients | Corn starch, Potato starch |
| Parabens | Delayed hypersensitivity; contact dermatitis | Thymol, Glycine (specific formulations) |
These safety concerns underscore the importance of rigorous validation and testing during excipient selection.
Meeting Regulatory Standards
After addressing safety, regulatory compliance becomes the next critical factor. Replacing solvents can improve safety while aligning with stringent regulatory frameworks. Manufacturers often prefer compendial excipients – those meeting the standards of the US Pharmacopeia–National Formulary (USP–NF) [20]. For noncompendial excipients, suppliers must maintain a Type IV Drug Master File (DMF) with regulatory agencies to ensure quality and consistency [20].
Regulatory acceptance can be verified through resources like the FDA Inactive Ingredients Guide, which lists permissible amounts in approved drugs. The Food Chemicals Codex and the Code of Federal Regulations (21 CFR parts 182 and 184) also provide guidance on substances generally recognized as safe (GRAS) [20]. For example, GRAS-listed oleic acid has shown promise for therapeutic use.
Vendor qualification is equally important. Suppliers should operate in facilities inspected by the FDA or the International Pharmaceutical Excipient Audit (IPEA). For materials derived from animals, prion-free certifications are necessary to address concerns about diseases like Transmissible Spongiform Encephalopathy (TSE).
In continuous manufacturing, standard pharmacopeial monographs may not fully address the complexities of production. Detailed quality agreements with suppliers should include specific tests and specifications tailored to the manufacturing process. As I. Silverstein explained:
"API quality is improved by reducing the presence of all materials other than the desired chemical… Excipient quality is described quite differently… a higher assay is not always better" [19].
The FDA’s PRIME Program encourages the development of novel excipients by allowing independent safety assessments. However, the high costs of toxicology testing often deter innovation, with most new excipients being modifications of existing materials [20].
Cost and Production Scale
Excipient selection also impacts production scalability and costs. Developing new excipients requires significant investment, with nonclinical studies mirroring those for new molecular entities. High costs and risks are particularly associated with noncompendial excipients included in New Drug Applications (NDAs) [4][22].
Physical properties like bulk density, flowability, and compressibility are crucial for large-scale production. For instance, light powders may require pre-blending, adding time and expense [21]. Early evaluation of feeder design compatibility can prevent costly disruptions.
Co-processed excipients (CPEs) simplify production by reducing the number of feeders required on a production line, potentially from six to two. This streamlines control strategies and lowers equipment costs [21]. However, reliance on a single supplier for CPEs introduces supply chain risks, which must be addressed during procurement [4][21]. As Krizia M. Karry, Brian Carlin, and Nigel Langley emphasized:
"The shift from batch-based to continuous production hinges on one critical factor: understanding and controlling the material properties of the raw materials involved" [21].
Continuous manufacturing systems, which tie output to runtime, reduce scale-up risks. These systems can tolerate short-term material flow disruptions without exceeding concentration limits, showcasing their efficiency [21]. However, inconsistent excipient properties can still cause interruptions, leading to higher operational costs.
Collaboration between suppliers and pharmaceutical companies is essential for successful excipient integration. As Sherry Ku and Ranga Velagaleti noted:
"The successful use of a novel excipient can only arise from a close partnership between the supplier (i.e., excipient manufacturer) and the user (i.e., pharmaceutical company)" [22].
For large-scale operations, variability between excipient batches must also be managed. Excipients are often produced in large quantities, with batch averages reported on Certificates of Analysis (CoA). Understanding residence time distribution allows manufacturers to track specific batches and address quality issues efficiently [21].
Advantages of Excipients Over Solvents
When considering the practicalities of excipient selection, the benefits of moving away from traditional solvents become apparent. Excipients tackle many challenges associated with organic solvents – such as toxicity and poor bioavailability – while improving safety, effectiveness, and shelf life.
Better Patient Safety
One major advantage of excipients is their ability to eliminate residual solvent toxicity. Organic co-solvents and surfactants, often used to enhance drug loading, can lead to toxic effects or immune responses in injectable formulations. In contrast, polymeric excipients and micelles provide safer, biocompatible options, even at higher concentrations.
Amino acid–based excipients, like LLS Health’s Apisolex, exemplify this safety advantage. Apisolex, a multiblock co-polymer containing a polysarcosine block, leverages sarcosine, a naturally occurring amino acid derivative, to ensure biocompatibility in parenteral formulations with high drug loading requirements [23]. Shreya Shah, Associate Director of Formulation Services at LLS Health, emphasized:
"Safety is always a key concern in the formulation of novel therapeutics, but it is even more crucial for parenteral formulations that bypass the gastrointestinal tract and are delivered directly to receptor sites via the bloodstream" [23].
A practical example of excipient use during the COVID-19 pandemic is Gilead Sciences‘ formulation of remdesivir. By incorporating sulfobutylether-β-cyclodextrin (SBECD), they safely delivered the hydrophobic active pharmaceutical ingredient (API) intravenously [23]. Beyond safety, excipients also play a vital role in enhancing how effectively drugs work.
Increased Drug Bioavailability
Many drugs face a common hurdle: poor water solubility. Around 40% of existing APIs and nearly 90% of those in development fall into this category [23][3]. Excipients offer solutions that organic solvents cannot.
Lipid-based excipients, for example, keep lipophilic drugs in liquid form until they reach the absorption site, bypassing the dissolution step that often limits absorption [24]. Similarly, Amorphous Solid Dispersions (ASDs) use hydrophilic polymers like PVP or HPMC to disperse APIs at a molecular level. This approach places the drug in an amorphous, high-energy state, significantly improving solubility and dissolution rates compared to crystalline forms [2][15].
Research highlights these benefits. In one study with beagle dogs, a carbamazepine formulation using hydroxypropyl-β-cyclodextrin (HP-β-CD) and 0.1% HPMC improved solubility by 95 times and increased bioavailability by 1.5 times compared to standard tablets [2][15]. Similarly, valsartan fast-disintegrating tablets containing 5% w/w crospovidone reduced arterial blood pressure by 60 mm Hg within an hour, outperforming a reference oral suspension, which only achieved a 10 mm Hg reduction over three hours [2][15].
Posaconazole, an antifungal, also highlights excipients’ impact. A solid dispersion formulation with hydroxypropyl methylcellulose acetate succinate (HPMC-AS) maintained drug supersaturation throughout the gastrointestinal tract, enabling effective absorption in a fasted state – unlike the traditional oral suspension, which required food for efficacy [2][15]. Beyond improving bioavailability, excipients also enhance product longevity.
Greater Stability and Longer Shelf Life
Excipients offer robust protection against environmental factors, unlike solvents that are removed during processing. Hygroscopic excipients, for instance, absorb residual moisture, reducing hydrolysis risks and preventing microbial growth [25]. By binding moisture, they effectively manage "water activity", ensuring it cannot degrade the API [26].
Other excipients provide targeted protection. Opaque materials and pigments shield APIs from UV light, while antioxidants neutralize oxygen and free radicals before they can cause damage [25][1]. Buffers help maintain a stable pH, avoiding degradation in acidic or basic conditions. In liquid and parenteral formulations, osmotic adjusters maintain physiological compatibility and chemical stability [1]. As Rohith from Pharma Focus Europe observed:
"Pharmaceutical excipients are no longer considered to be a passive component. These substances affect the performance, stability, safety and patient’s acceptance of drug products significantly" [1].
For moisture-sensitive APIs, excipients with low water activity are essential. Unlike excipients with high free water content, those with bound moisture prevent degradation, preserving the drug’s shelf life [26].
This content is for informational purposes only. Always consult official regulations and qualified professionals for formulation or sourcing decisions.
Conclusion
Excipients have emerged as a powerful alternative to traditional solvents, addressing both safety and regulatory challenges while improving drug formulations. These non-toxic, biocompatible substances eliminate harmful residues and meet stringent standards, simplifying approval processes and reducing health and environmental risks.
By enabling the formation of inclusion complexes and amorphous dispersions, excipients enhance bioavailability and protect active pharmaceutical ingredients (APIs) from degradation. They also eliminate the need for toxic organic co-solvents, offering a safer and more efficient solution. Unlike APIs, excipients are complex mixtures where individual components significantly impact functionality [19].
Sourcing high-quality excipients is essential for successful formulations. A 2020 US Pharmacopeia survey revealed that 28% of formulators faced development roadblocks due to excipient limitations [3]. Compendial-grade excipients (USP/NF) ensure compliance, provide critical safety data for FDA filings, and support the transition from research to GMP manufacturing.
"It is also important to work with a supplier who understands what it takes to scale up the excipient for GMP manufacturing and who can provide all the regulatory and safety/tox testing that the FDA will require."
- Nick DiFranco, Global Market Segment Manager at Lubrizol Life Science Health [3]
Allan Chemical Corporation brings over 40 years of experience in regulated industries, offering pharmaceutical-grade excipients that meet USP, NF, ACS, and FCC standards. With just-in-time delivery, robust technical support, and direct relationships with vetted manufacturers, AllanChem ensures consistent quality and a reliable supply chain. This helps formulators overcome solubility challenges while meeting regulatory demands.
The transition to excipients requires close collaboration between suppliers and pharmaceutical developers. By partnering with trusted providers who understand both technical and regulatory needs, formulators can confidently replace solvents with excipients that enhance drug performance, prioritize patient safety, and ensure market success.
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
FAQs
How do I choose the right excipient to replace a solvent?
Choosing the right excipient requires a careful evaluation of several factors: its ability to improve solubility of the active pharmaceutical ingredient (API), compatibility with other formulation components, safety profile, and compliance with regulatory requirements. For improving solubility, excipients such as Cyclodextrins or specific polymers can be effective. High-throughput screening methods are useful for identifying the most suitable candidates.
It’s essential to ensure that the excipient complies with standards like USP or FCC. For technical-grade solutions, consult reliable suppliers who can provide high-quality materials and detailed technical support.
What lab tests confirm drug–excipient compatibility?
Differential Scanning Calorimetry (DSC) is widely used in laboratories to check drug–excipient compatibility. This test helps detect interactions that could alter the chemical or physical characteristics of a formulation, ensuring the final product remains stable and effective.
Will switching to excipients change bioavailability or shelf life?
Switching to excipients as alternatives to traditional solvents can influence both bioavailability and shelf life, depending on the specific excipient and formulation used. For example, excipients such as solubilizers can boost solubility and absorption, leading to improved bioavailability.
However, excipients also play a role in stability. Factors like phase behavior and chemical interactions within the formulation can directly affect the product’s shelf life. To balance these two critical aspects, careful excipient selection and strict quality control are essential. This ensures that bioavailability and stability are optimized without sacrificing the product’s overall performance.





Comments are closed