Lyophilization, or freeze-drying, stabilizes protein-based drugs by removing water through freezing, sublimation, and desorption. This process minimizes chemical degradation, extends shelf life, and supports precise dosing. However, without protective excipients, proteins can lose activity due to stress from freezing, pH shifts, and water removal.
Excipients like sucrose and trehalose act as cryoprotectants and lyoprotectants, preserving protein structure by forming hydrogen bonds and creating glassy matrices. Studies show trehalose offers better miscibility and stability under high temperatures compared to sucrose. Bulking agents like Mannitol provide structural support during drying but pose risks of crystallization issues that can compromise protein stability. Surfactants reduce aggregation at interfaces, while buffers maintain pH and prevent degradation. Chelators like EDTA combat metal-catalyzed oxidation, enhancing overall stability.
Analytical tools such as Differential Scanning Calorimetry (DSC), Size Exclusion Chromatography (SEC), and solid-state NMR are critical for assessing excipient performance and ensuring long-term protein stability. Selecting the right excipients, monitoring impurities, and using high-purity materials are key to maintaining the integrity of lyophilized protein formulations.
Conformational jamming of protein molecules in the solid state measured by terahertz dynamics
Cryoprotectants and Lyoprotectants: How They Protect Proteins
Excipient Performance Comparison for Lyophilized Protein Stability
Developing an effective excipient strategy relies on understanding protective mechanisms and validating stability through rigorous testing. Disaccharides like sucrose and trehalose safeguard proteins in two key ways: they create a rigid, glassy amorphous matrix that limits molecular movement, and they substitute the protein’s hydration shell by forming hydrogen bonds with the protein surface as water evaporates [6][8].
The success of these sugars lies in their ability to intimately mix with proteins at the molecular level. Trehalose, in particular, demonstrates better miscibility with proteins at a 2–5 nm scale, allowing closer interaction compared to larger molecules like polysaccharides [8]. This close interaction enables trehalose to more effectively slow protein dynamics by tightly coupling with residual hydration water [9].
"The better stabilizing properties of trehalose are then assumed to be due to a stronger dynamic coupling of trehalose to water, which, in turn, leads to a stronger reduction of the protein dynamics." – Jan Swenson, Division of Nano-Biophysics, Chalmers University of Technology [9]
In a study conducted in September 2007 at Dongduk Women’s University, researchers tested recombinant human serum albumin (rHSA) with different excipients. After 4 months of storage at 95°F (35°C), formulations containing sucrose and trehalose showed no detectable aggregation, while mannitol-only formulations exhibited visible protein clumping. Differential scanning calorimetry confirmed that the disaccharides formed complete amorphous states, fully protecting the protein during storage [7]. These findings are further supported by quantitative stability studies.
Stability Data for Cryoprotectants
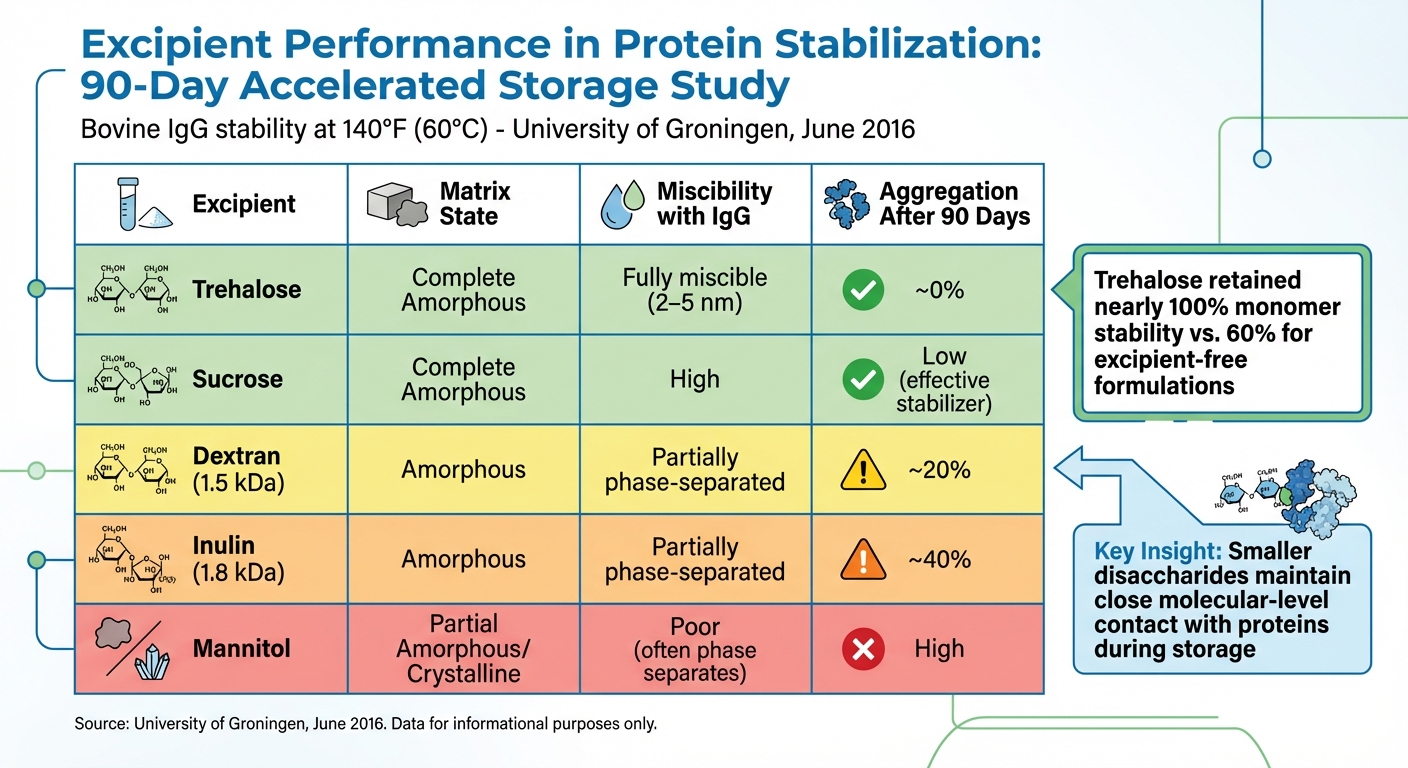
Additional data highlights the protective effects of cryoprotectants under accelerated conditions. In a June 2016 study from the University of Groningen, researchers used solid-state NMR to assess the stability of bovine IgG. After 90 days at 140°F (60°C), trehalose formulations retained nearly 100% monomer stability with minimal aggregation. By contrast, excipient-free formulations retained only about 60% monomer stability, while larger sugars like dextran (1.5 kDa) and inulin (1.8 kDa) resulted in approximately 20% and 40% aggregation, respectively [8].
| Excipient | Matrix State | Miscibility with IgG | Aggregation After 90 Days at 140°F (60°C) |
|---|---|---|---|
| Trehalose | Complete Amorphous | Fully miscible (2–5 nm) | ~0% |
| Sucrose | Complete Amorphous | High | Low (effective stabilizer) |
| Dextran (1.5 kDa) | Amorphous | Partially phase-separated | ~20% |
| Inulin (1.8 kDa) | Amorphous | Partially phase-separated | ~40% |
| Mannitol | Partial Amorphous/Crystalline | Poor (often phase separates) | High |
This data underscores the superior long-term stability provided by smaller disaccharides, which maintain close molecular-level contact with proteins during storage. The choice between sucrose and trehalose often depends on storage conditions, as trehalose’s higher glass transition temperature (~230°F or 110°C compared to sucrose’s ~140°F or 60°C) makes it a better option for warmer environments [10].
This information is for educational purposes only. Always consult regulatory guidelines and qualified professionals before making formulation or sourcing decisions.
Bulking Agents: Structure Support and Crystallization Issues
Disaccharides may stabilize proteins at the molecular level, but bulking agents like Mannitol play a different role. They provide structural support, which is crucial during lyophilization to maintain the integrity of the lyophilized cake. Mannitol, in particular, crystallizes during freezing, forming a rigid framework that prevents collapse during drying. This crystalline structure allows for higher product temperatures during the primary drying phase, speeding up sublimation [11].
A 2018 study by Wolfgang Friess illustrated this advantage using a formulation with 40 mg/mL Mannitol and 10 mg/mL Sucrose. The crystalline scaffold formed by Mannitol enabled drying at 14°F (-10°C), cutting the primary drying time by 85% – from 47 hours to just 7 hours – compared to a more conservative approach at -39°F (-39°C) [11].
However, Mannitol’s crystallization isn’t without challenges. It can produce three anhydrous polymorphs (α, β, δ) and a metastable hemihydrate. The hemihydrate poses a particular problem because when it dehydrates during storage, the released water can lead to protein aggregation and degradation [18, 19].
"Mannitol’s multiple crystalline modifications can compromise product reproducibility and long-term stability." – Stefan Schneid, Xenia Riegger, and Henning Gieseler, PhD [12]
To address these issues, formulators often use a Mannitol-to-Sucrose ratio of at least 4:1 to ensure complete crystallization. They also incorporate an annealing step – holding the product at around 5°F (-15°C) for several hours during freezing – to promote the formation of stable polymorphs. The following section delves into the specific crystallization risks of Mannitol and the analytical tools used to monitor them.
Crystallization Risks in Lyophilized Proteins
Incomplete crystallization of Mannitol can lead to moisture-related instability, as highlighted in a 2008 study from the University of Erlangen. Researchers observed that adding 1% Sucrose to a Mannitol formulation raised residual moisture levels from 0.2% to 2.4%. X-ray powder diffraction (XRPD) confirmed that these Sucrose-containing formulations showed significant amounts of Mannitol hydrate, which is associated with increased moisture-related instability [12].
Analytical methods are essential for understanding how Mannitol’s crystallization impacts protein stability. Techniques like Fourier-transform infrared spectroscopy (FTIR) and size-exclusion chromatography (SEC) help assess protein structure and aggregation. Meanwhile, XRPD and differential scanning calorimetry (DSC) are used to identify specific crystalline forms in the final product. Among these, the δ-form is often favored, though it carries some instability risks [17, 19]. Monitoring for Mannitol hemihydrate is particularly critical, as its conversion to an anhydrous form during storage can jeopardize long-term protein stability.
Striking the right balance between Mannitol’s advantages and its risks is key to creating optimal formulations. While Mannitol provides a strong cake structure and enables faster drying, it does not stabilize protein conformation once crystallized. For this reason, it is typically paired with amorphous stabilizers like Sucrose or Trehalose to keep proteins in a glassy, stable state [15, 17].
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Surfactants and Buffers: Preventing Aggregation and Maintaining pH
Proteins naturally shield their hydrophobic cores, but during lyophilization, they face challenges at air–water and ice–water interfaces. Surfactants, like PS20 and PS80, play a key role in reducing protein aggregation by preferentially adsorbing at these interfaces [13]. Hanns-Christian Mahler explains:
"Competitive interaction of nonionic surface-active components at the interface (such as the air–water or ice–water interface) is the primary mechanism for preventing interface-induced aggregation." [13]
However, PS20 and PS80 come with their own issues. These surfactants are susceptible to thermal autoxidation and hydrolysis, which can generate peroxides that oxidize sensitive amino acids like methionine [17][18]. For those looking to avoid oxidative damage, alkylsaccharides provide a safer alternative, offering similar anti-aggregation properties without the risk of peroxide formation [18]. These surfactants work hand-in-hand with buffers, which maintain protein stability by regulating pH.
Buffers stabilize proteins by controlling pH, which is critical for minimizing both chemical degradation (e.g., deamidation, oxidation) and physical instability (e.g., aggregation, denaturation) [15][5]. In the solid state, the "microenvironmental pH" – the ionization state of protein functional groups within the dried matrix – becomes particularly significant [15]. During freezing, however, some buffer components can crystallize or phase separate, causing pH fluctuations that destabilize proteins [14][16]. Potassium phosphate is often preferred over sodium phosphate for this reason. Sodium phosphate, especially in its dibasic form, tends to crystallize during freezing, creating an acidic environment that can harm protein stability [15][16].
How Buffers and Chelators Affect Stability
Buffers do more than just regulate pH – they also influence long-term protein stability. In June 2022, researchers from Purdue University and Merck & Co. studied Bovine Serum Albumin (BSA) formulations using 200 mM buffers (phosphate, succinate, and citrate). Their findings, based on solid-state NMR, revealed that phase separation in succinic buffer formulations was directly linked to reduced stability during a 90-day accelerated storage test at 104°F (40°C). Data from ssHDX-MS showed a strong correlation between phase separation and the loss of monomeric protein area (R² = 0.8722 for spray-dried and 0.8428 for lyophilized formulations) [14]. This underscores the importance of advanced techniques like ssNMR, which can detect issues like phase separation that traditional methods, such as ssFTIR, might miss.
In addition to buffers, chelators like EDTA enhance formulation stability by preventing metal-catalyzed oxidation. Trace amounts of transition metals, often introduced through excipients or equipment, can trigger the formation of reactive oxygen species via the Fenton pathway [5]. EDTA works by binding metals such as iron and copper, blocking this degradation process. However, chelators can sometimes interact with and destabilize proteins [18]. Even with moisture levels below 1%, chemical degradation (e.g., deamidation, oxidation) remains a concern, making it essential to carefully balance buffers and chelators for optimal stability [5].
This information is intended for educational purposes. Always consult official guidelines and qualified professionals when making formulation or sourcing decisions.
sbb-itb-aa4586a
Amino Acids and Other Excipients: Additional Stability Factors
In addition to sugars and bulking agents, amino acids and other minor excipients play a role in maintaining protein stability. Amino acids like leucine, for instance, serve different purposes depending on the drying method. In spray-drying, leucine’s surface activity helps form a protective shell, improving aerosol performance. On the other hand, in lyophilized formulations, leucine offers less stabilization compared to disaccharides like trehalose or sucrose. This difference arises because, without rapid droplet evaporation, leucine doesn’t concentrate at interfaces or form the strong glassy matrix (vitrification) that disaccharides provide. Furthermore, amino acids tend to crystallize easily, which can disrupt the amorphous state required for long-term protein stability. These findings align with the "water substitute hypothesis", a key concept in protein stabilization during drying.
The "water substitute hypothesis" suggests that stabilizers protect proteins by forming hydrogen bonds to replace those lost when water is removed. While both sugars and amino acids can form these bonds, sugars are more effective at creating a glassy matrix that preserves protein structure and integrity during storage.
Managing impurities is another critical factor in maintaining protein stability. For example, excipients like polysorbates and PEG can introduce peroxides that oxidize sensitive amino acid residues such as methionine, tryptophan, and histidine [5][19]. Transition metal ions, whether from excipients like glycine or from stainless steel processing equipment, can catalyze oxidative reactions even at trace levels [19][20]. A study in the Journal of Pharmaceutical Sciences highlighted:
"Transition metal ions are often present in excipients, and processing in stainless steel equipment can lead to significant iron contamination." [5]
Additionally, while non-reducing sugars like sucrose are generally stable, they may contain metal ions or hydrolyze under low pH conditions, producing reducing sugars like glucose and fructose. These reducing sugars can trigger the Maillard reaction with amino acid residues such as lysine and arginine [5][21]. Another concern is formaldehyde, a byproduct of polysorbate or PEG oxidation, which can lead to protein cross-linking and adduct formation [19]. As Kelly Zhang, a Senior Scientist, explains:
"Reactive impurities in excipients can cause drug product degradation or protein modification even at trace levels, and thus impact drug stability and quality." [19]
Such impurities can alter proteins, diminish potency, and generate toxic degradation products. To address these challenges, combining effective cryo- and lyoprotectants with rigorous impurity control is essential. This includes thorough lot screening for reactive impurities, using metal chelators like EDTA or DTPA to prevent oxidative damage, and sourcing excipients with high-purity or multi-compendial-grade certifications to minimize contamination risks.
Allan Chemical Corporation provides a range of technical-grade and compendial-grade excipients (USP, FCC, ACS, NF) backed by stringent quality systems, supporting pharmaceutical manufacturers in meeting the demanding purity standards for lyophilized protein formulations.
This content is for informational purposes only. Always consult official regulations and qualified professionals when making sourcing or formulation decisions.
Analytical Methods for Evaluating Excipients
Accurate analysis is crucial for choosing excipients that help maintain the stability of lyophilized proteins. These techniques are designed to detect even the smallest changes in protein structure and stability, offering insights into long-term performance and identifying potential formulation challenges early. This analytical approach forms the foundation for selecting excipients, as discussed in earlier sections.
Differential Scanning Calorimetry (DSC) plays a key role in determining two important temperatures: the glass transition temperature (Tg) and the unfolding temperature (Tm). Keeping products below Tg minimizes molecular movement, which is critical for stability [3][22]. Anjali Joshi, Director of Formulation Development at Recipharm, highlights the importance of this method:
"Excipients should therefore be chosen based on their ability to maximize the unfolding temperature, which is best determined using thermal analysis techniques, most commonly differential scanning calorimetry (DSC)" [1].
DSC is also used to identify the eutectic temperature of crystalline bulking agents like mannitol. This information helps in designing lyophilization cycles that prevent collapse during the process [23].
Solid-State Fourier Transform Infrared Spectroscopy (ssFTIR) examines protein secondary structures – such as α-helices and β-sheets – directly in the dried solid state without requiring reconstitution. This is particularly important because any unfolding caused by dehydration may not be obvious once the protein is redissolved [2][3]. John F. Carpenter, a professor at the University of Colorado, underscores this point:
"A native protein secondary structure in the dried solid was necessary for long-term storage stability" [3].
However, ssFTIR has its limitations, especially in detecting subtle structural changes that could still impact the protein’s stability.
Size Exclusion Chromatography (SEC) remains the go-to method for assessing physical instability by measuring protein aggregation and changes in the monomeric peak after reconstitution [2][22]. For instance, a June 2022 study by Purdue University and Merck & Co. monitored bovine serum albumin (BSA) formulations over 90 days at 104°F (40°C). The SEC results showed that phase separation between buffer components and the protein reduced stability, with R² values exceeding 0.84 [4]. In another study from August 2019, researchers Nathan E. Wilson and Elizabeth M. Topp at Purdue University used SEC to evaluate four model proteins with different excipients. They found that formulations containing mannitol generally showed higher aggregation levels compared to those with sucrose or trehalose [22].
Advanced ssHDX-MS offers heightened sensitivity in detecting minor conformational changes by analyzing hydrogen bonding patterns. Additional tools like X-ray Powder Diffraction (XRPD) identify unwanted crystallization in bulking agents, while solid-state Nuclear Magnetic Resonance (ssNMR) detects phase separation that can lead to localized acidity and reduced stability [22][4]. Nathan E. Wilson from Purdue University explains:
"ssHDX-MS detected subtle changes in conformation and/or matrix interactions for these proteins that were correlated with storage stability, suggesting that the method can be used to design robust solid-state pharmaceutical protein products more rapidly" [2].
By combining these analytical techniques, researchers can make informed decisions about excipient selection, supporting the formulation strategies discussed in earlier sections.
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Conclusion: Selecting the Right Excipients
Choosing the right excipients starts with a well-thought-out design strategy that considers specific interactions between proteins and excipients. Since protein stability depends heavily on weak hydrogen bonds, selecting the appropriate excipients is essential to preserve protein integrity throughout the product’s shelf life [18].
These theoretical principles translate into practical applications through analytical assessments. Techniques like water replacement and vitrification address key degradation pathways identified during preformulation studies [18][1]. As Jay Kang, Director of Analytical and Formulation Development at Patheon, points out:
"A good practice is to use compendial excipients in the development study and control the quality/storage of the excipients from the very beginning, so no surprises are encountered in GMP manufacturing" [18].
Analytical methods play a critical role in excipient selection by identifying issues such as phase separation, crystallization, and trace impurities. For more on these techniques, refer to the Analytical Methods section above. When combined with design-of-experiment (DoE) methodologies, these approaches allow pharmaceutical scientists to fine-tune formulations effectively while adhering to strict development timelines.
A successful formulation also depends on a steady supply of high-quality excipients. Allan Chemical Corporation offers compendial-grade excipients (USP, FCC, ACS, NF) with rigorous impurity controls to ensure consistency from development to GMP manufacturing. With over 40 years of experience in regulated industries and strong relationships with vetted manufacturers, Allan Chemical delivers a reliable supply chain. Their focus on minimizing trace impurities – like metals in sugars or peroxides in polysorbates – helps protect protein stability. Additionally, their just-in-time delivery and technical support ensure that formulation integrity is maintained throughout the product lifecycle.
This content is for informational purposes only. Always consult official regulations and qualified professionals when making sourcing or formulation decisions.
FAQs
How do sucrose and trehalose differ as stabilizers for lyophilized proteins?
Sucrose and trehalose are both common choices for stabilizing lyophilized proteins, but they each bring different strengths to the table. Trehalose is particularly known for forming a more complete amorphous state, which helps protect proteins from aggregation and degradation. This advantage stems from its higher glass transition temperature and stronger interactions with protein molecules.
On the other hand, sucrose is also effective but may not match trehalose in situations where maximum stability is required. Deciding between these two excipients depends on factors like the specific protein being stabilized, the formulation’s requirements, and the desired shelf life. A thorough assessment of their properties is key to ensuring the best possible stabilization.
What role does mannitol crystallization play in the stability of lyophilized protein drugs?
Mannitol crystallization during lyophilization plays a key role in enhancing sublimation rates, which can make the drying process more efficient. However, this same crystallization can trigger phase changes that may jeopardize the long-term stability of protein formulations. For example, the development of a metastable crystalline hemihydrate can weaken the product’s structural integrity over time.
Maintaining stability requires precise control over mannitol’s crystallization during both formulation and processing. This underscores the need to carefully choose excipients and fine-tune lyophilization parameters when working with protein-based drugs.
How do surfactants and buffers help prevent protein aggregation during lyophilization?
Buffers and surfactants are essential for preserving protein stability during lyophilization. Buffers are responsible for maintaining a stable pH, which is crucial for ensuring that proteins keep their structure and function intact. Surfactants serve a different but equally important role by reducing surface tension. This prevents proteins from adhering to container walls or interfaces, which could otherwise cause aggregation.
Together, these excipients shield proteins from both physical and chemical stresses, helping to enhance the stability and extend the shelf life of lyophilized products.





Comments are closed