Electrochemical stability and thermal stability are two essential properties that determine how materials perform under electrical and thermal stress. Electrochemical stability refers to a material’s resistance to degradation when exposed to voltage and current, while thermal stability measures its ability to withstand high temperatures without breaking down. Both are critical for safety and performance in industries like energy storage, electronics, and pharmaceuticals.
Key points:
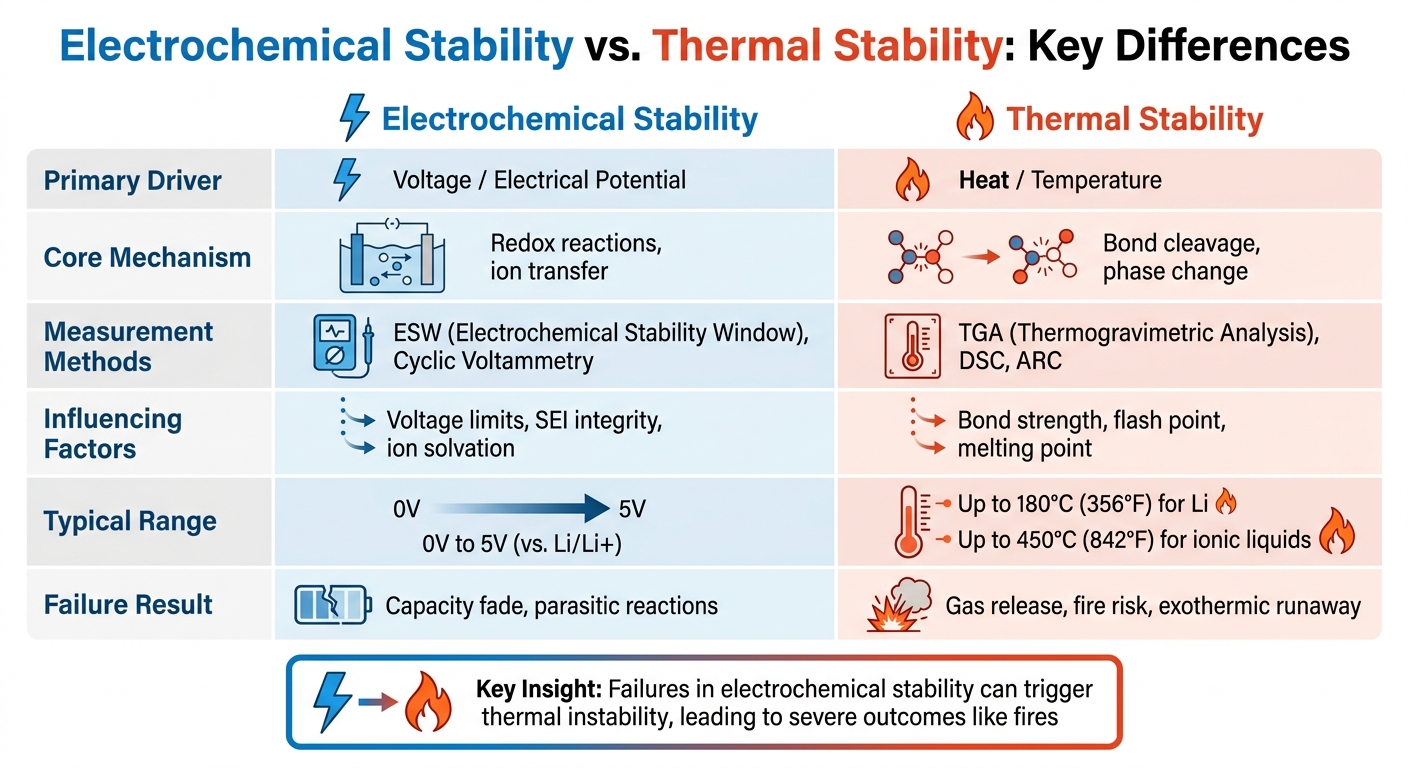
- Electrochemical stability is defined by the electrochemical stability window (ESW), which measures the voltage range where a material remains stable.
- Thermal stability focuses on decomposition temperature and is commonly assessed using Thermogravimetric Analysis (TGA).
- Failures in electrochemical stability can trigger thermal instability, leading to severe outcomes like fires.
Quick Comparison:
| Metric | Electrochemical Stability | Thermal Stability |
|---|---|---|
| Primary Driver | Voltage / Electrical Potential | Heat / Temperature |
| Core Mechanism | Redox reactions, ion transfer | Bond cleavage, phase change |
| Measurement Methods | ESW, Cyclic Voltammetry | TGA, DSC, ARC |
| Failure Result | Capacity fade, parasitic reactions | Gas release, fire risk |
Understanding these differences helps in selecting materials that balance both properties for high-performance and safe industrial applications.
Electrochemical vs Thermal Stability: Key Differences Comparison Chart
Linking multi-physics mechanisms to the performance & degradation of solid-state batteries – MC Pang
sbb-itb-aa4586a
What is Electrochemical Stability?
Electrochemical stability refers to a material’s ability to resist redox decomposition when exposed to an electrical potential. This property is often defined by the electrochemical stability window (ESW), which is the voltage range within which an electrolyte or solvent remains stable without undergoing chemical changes [9]. According to Philippe Bühlmann, a Chemistry Professor at the University of Minnesota:
"The electrochemical window is the potential range in which an electrolyte/solvent system does not get reduced or oxidized" [9].
The ESW is calculated by subtracting the reduction (cathodic) potential from the oxidation (anodic) potential. For instance, lithium bis-(trifluoromethanesulfonyl)imide (LiTFSI, CAS No. 90076-65-6) exhibits an ESW of about 3.0 V, functioning effectively within a range of 1.9 to 4.9 V [11]. If this range is exceeded, the electrolyte can break down, leading to reduced efficiency and possible device failure.
Definition and Measurement
Measuring electrochemical stability can be challenging. A common method, linear sweep voltammetry (LSV), gradually increases the potential until a specific cut-off current (typically 0.01 to 5.0 mA/cm²) is reached. However, this approach can produce ESW values that vary by as much as 0.9 V [9].
An alternative technique, the linear fit method, determines the ESW by identifying the point where the background current intersects with the decomposition current slope. This approach avoids arbitrary thresholds and reduces the influence of mass transport effects, providing more consistent results – especially when working with high-surface-area porous electrodes. For high-temperature applications, specialized methods like high-temperature cyclic voltammetry (HT-CV) are used, particularly with solid-state cells, to study oxide stability and redox processes at temperatures above 250°C (482°F) [8].
These advanced measurement techniques are essential for ensuring the reliability of materials in demanding energy systems.
Where Electrochemical Stability Matters
Accurate electrochemical stability measurements are crucial for high-performance technologies such as batteries, supercapacitors, and fuel cells. For example, in lithium-metal batteries, electrolytes must endure voltages up to 4.8 V and temperatures as high as 300°C (572°F) without degrading [10]. While organic electrolytes typically degrade at voltages above 4.0 V, ionic liquids offer a broader stability range, often between 4 and 5 V [11].
In fuel cells and other electronic devices, maintaining electrochemical stability is critical to preventing the degradation of components like platinum catalysts, which often operate in highly corrosive environments and under significant cathodic potentials [5]. A striking example of this importance is NASA‘s Mars 2020 Perseverance Rover. In 2021, its Mars Oxygen In Situ Resource Utilization (MOXIE) experiment used high-temperature electrolysis with zirconia cells to produce 5.37 grams of oxygen per hour from the Martian atmosphere. This process relied on materials capable of maintaining their electrochemical stability under extreme conditions [7].
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
What is Thermal Stability?
Thermal stability refers to a material’s ability to resist decomposition when exposed to heat. Unlike electrochemical stability in mixed environments, which deals with degradation caused by voltage extremes, thermal stability focuses on how heat impacts a material’s structure. According to ASTM E2550, this property is defined by the temperature at which decomposition starts and the associated mass change, typically measured using thermogravimetry [12].
At the molecular level, thermal stability is influenced by the strength of chemical bonds. Some bonds are more vulnerable to heat, leading to structural breakdown or unintended chemical reactions. For instance, acetylsalicylic acid (Aspirin, CAS No. 50-78-2) begins decomposing around 102°C (216°F) in a nitrogen atmosphere, as determined by ASTM E2550 testing [12].
Definition and Testing Methods
Thermogravimetric Analysis (TGA) is the primary method for assessing thermal stability. It measures weight loss as a material is heated, identifying the temperature at which decomposition begins [12]. However, TGA results can vary based on factors like sample mass, heating rate, crucible type, and the surrounding atmosphere. To ensure accurate comparisons, tests must be conducted under consistent conditions. ASTM E2550 also recommends zooming in on TGA curves to a scale of 1% to 2% mass change to clearly pinpoint the onset of decomposition. It’s worth noting that TGA is specific to materials that decompose or react and cannot evaluate stability against processes like sublimation or vaporization [12]. This makes TGA a critical tool for selecting materials suited to high-temperature environments.
Where Thermal Stability Matters
Thermal stability plays a crucial role in industries that operate under high-temperature conditions. Whether in pharmaceuticals, electronics, or ceramics, understanding a material’s behavior under heat ensures safety and reliability [12][13].
In pharmaceuticals, thermal stability determines the temperature at which active ingredients start to break down, directly affecting a drug’s safety and shelf life [12]. In electronics and battery production, it’s vital for preventing hazardous situations. For example, NMC811, a cathode material used in electric vehicle batteries, starts self-heating around 175°C (347°F) and experiences thermal runaway at approximately 188°C (370°F). Charged NMC811 releases about 1,142 J/g of heat, significantly more than materials like ζ-V₂O₅ (361 J/g) and ε-VOPO₄ (406 J/g), which are considered more stable [3]. These insights are essential for ensuring that materials can perform safely and effectively in high-temperature applications.
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Key Differences Between Electrochemical and Thermal Stability
How Each Type of Stability Works
Electrochemical and thermal stability operate through entirely different mechanisms. Electrochemical stability relies on voltage and electron transfer, driven by redox reactions that occur at specific electrode potentials. These reactions can involve subtractive processes like surface atom dissolution and selective dissolution, or additive processes such as intercalation, deposition, and passivation [5]. Thermal stability, by contrast, depends on a material’s resistance to heat and the strength of its chemical bonds. It follows Arrhenius behavior, where decomposition rates increase exponentially as temperature rises [1]. For ionic liquids, the electrochemical stability window (ESW) is influenced by the stability of both the cation and anion, while thermal decomposition is dominated by the less-stable ion, typically the anion [2]. Professor Tiancheng Mu from Renmin University of China notes:
"For thermal stability, the interactions between ions are less important than the individual anions and cations. The decomposition temperature is mainly determined by the less-stable ion, usually the anion" [2].
Despite their differences, these types of stability often intersect. Take layered oxide cathodes as an example: thermal instability here arises from transition metals migrating into lithium sites – a phenomenon known as cation mixing. This process weakens metal-oxygen bonds, leading to oxygen gas release [1]. Similarly, lithium metal’s relatively low melting point of 180°C (356°F) represents a critical thermal stability threshold. Once melted, lithium can migrate uncontrollably, potentially causing internal shorts [1]. Understanding these mechanisms is essential for designing materials suitable for high-temperature systems, as both stability types directly impact safety and performance. The contrast between voltage-driven redox reactions and heat-induced bond cleavage provides a clear framework for comparing these mechanisms.
Side-by-Side Comparison
The table below outlines the main differences between electrochemical and thermal stability:
| Metric | Electrochemical Stability | Thermal Stability |
|---|---|---|
| Primary Driver | Voltage / Electrode Potential | Heat / Temperature |
| Core Mechanism | Redox reactions, ion (de)intercalation | Bond cleavage, phase transitions |
| Measurement | Electrochemical Window (ESW), Cyclic Voltammetry | TGA, DSC, ARC |
| Influencing Factors | Voltage limits, SEI integrity, ion solvation | Bond strength, flash point, melting point |
| Typical Range | 0V to 5V (vs. Li/Li+) | Up to 180°C (356°F) for Li; up to 450°C (842°F) for ionic liquids |
| Failure Result | Parasitic reactions, capacity fade | Exothermic runaway, gas evolution, fire |
Electrochemical stability is commonly assessed through methods like the electrochemical stability window and real-time monitoring of voltage, current, and impedance to detect internal issues [4]. Thermal stability, on the other hand, is measured using specialized techniques such as Thermogravimetric Analysis (TGA), Differential Scanning Calorimetry (DSC), and Accelerating Rate Calorimetry (ARC). ARC, for instance, employs the "Heat-Wait-Search" method to pinpoint self-heating onset and thermal runaway thresholds [3].
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Performance in High-Temperature Applications
This section delves into how stability mechanisms function when subjected to high-temperature environments.
Trade-Offs in Ionic Liquids
Ionic liquids strike a delicate balance in high-temperature electrochemical systems. These solvents, known for being nonvolatile and noncombustible, provide better thermal stability than flammable carbonate-based electrolytes. However, studies suggest that their higher thermal stability can lead to increased heat production due to exothermic reactions at the electrodes [14].
The cation structure in ionic liquids often dictates a trade-off between thermal stability and electrochemical performance. While anions like TFSI (Tf₂N⁻) can remain stable at temperatures exceeding 420°C (788°F) [16], cations tend to be the weak point. For instance, adding aliphatic groups – methyl, methylene, or methine – improves electrochemical properties but introduces reactive [C(sp³)–H] bonds that are more prone to oxidation compared to the more stable aromatic [C(sp²)–H] bonds [16].
In TFSI-based systems, Hofmann elimination emerges as the primary failure mechanism when oxygen is present [14]. James H. Davis Jr. from the University of South Alabama highlights this vulnerability:
"cations [are] commonly being the thermal weak link of organic salts" [16].
One potential solution lies in peraryl cations, such as PPh₄⁺, which have demonstrated impressive thermal stability. These cations can withstand air exposure at 300°C to 400°C (572°F to 752°F) for extended periods, with PPh₄⁺Tf₂N⁻ remaining stable up to approximately 425°C (797°F) [16]. However, this enhanced thermal durability may come at the cost of reduced electrochemical flexibility.
The trade-offs seen in ionic liquids mirror those in battery cathode materials, which are discussed below.
Comparing Battery Cathode Materials
Battery cathode materials also face compromises between energy density and thermal stability. Polyanion-type cathodes exemplify this balance. In September 2025, researchers Chanmonirath Chak, Vadim Shipitsyn, and Lin Ma from the University of North Carolina at Charlotte compared LFP/graphite and LMFP/graphite pouch cells using a 1.2 m LiFSI-based electrolyte. Their findings showed that LMFP offered a higher gravimetric energy density (169 Wh/kg versus 154 Wh/kg for LFP) but had a lower thermal onset and slightly shorter cycling stability. After 275 cycles at 40°C, LFP retained 91% capacity, while LMFP retained 89% [15].
Thermal performance differences are particularly notable. LFP has a thermal onset temperature of 140°C (284°F), while LMFP begins exothermic reactions at 125°C (257°F). Nonetheless, both materials exhibit low self-heating rates – below 0.1°C/min – indicating strong safety characteristics [15]. The reduced thermal stability of LMFP is linked to Jahn-Teller distortion in its manganese-based structure, which increases charge transfer resistance to 112.2 Ω·cm² after extended cycling, compared to 69.52 Ω·cm² for LFP [15].
| Metric | LiFePO₄ (LFP) | LiMn₀.₆Fe₀.₄PO₄ (LMFP) |
|---|---|---|
| Gravimetric Energy Density | 154 Wh/kg | 169 Wh/kg |
| Volumetric Energy Density | 355 Wh/L | 360 Wh/L |
| Thermal Onset Temperature | 140°C (284°F) | 125°C (257°F) |
| Capacity Retention (275 cycles at 40°C) | 91% | 89% |
| Charge Transfer Resistance (after cycling) | 69.52 Ω·cm² | 112.2 Ω·cm² |
| Self-Heating Rate | < 0.1°C/min | < 0.1°C/min |
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Implications for Industrial Applications
Materials for High-Temperature Electrochemical Systems
Industries relying on high-temperature electrochemical systems face the challenge of ensuring stability in both electrochemical and thermal environments. This balance becomes especially critical in energy storage and electronic devices, where manufacturing often involves sintering at temperatures ranging from 600°C to 1,200°C (1,112°F to 2,192°F) to produce dense ceramic components [17]. These extreme conditions can lead to mechanical cracks caused by mismatched thermal expansion rates among components, which disrupt ion transport and degrade device performance. Such issues are particularly evident at electrode–electrolyte interfaces, where thermal expansion mismatches can significantly impair functionality.
The choice between single-crystalline and polycrystalline materials highlights the complexity of this problem. For instance, studies on solid-state batteries reveal that polycrystalline NMC631 combined with LLZTO at 900°C (1,652°F) suffered a 63% capacity loss due to resistive interfacial phases forming at the electrode-electrolyte boundary. In contrast, single-crystalline versions of the same material reduced this loss to 48% while retaining structural integrity at temperatures as high as 1,000°C (1,832°F) [18]. Polycrystalline materials tend to form these resistive phases at lower temperatures, further complicating their use in high-temperature applications. These findings emphasize the delicate trade-offs manufacturers must navigate during high-temperature processing to maintain performance and durability.
Pietro Papa Lopes of Argonne National Laboratory‘s Materials Science Division underscores the importance of considering long-term material stability:
"Material stability focuses on observing changes to the structure and composition over time during operation, which inherently provides greater insight on how to iteratively manipulate the material and interface" [5].
This perspective is particularly relevant in regulated industries, where devices are expected to perform reliably for 10 to 30 years under the demands of high-temperature electrochemical systems [5]. Applications such as pharmaceutical manufacturing equipment, ceramic processing systems, and electronic components require materials that resist degradation into toxic or non-functional byproducts when exposed to prolonged thermal stress.
Working with Chemical Suppliers
In addition to material design, the purity of chemical inputs is essential to maintaining electrochemical and thermal performance. For ceramic electrolyte systems, suppliers must deliver materials with precise crystallographic structures to prevent unwanted reactions during sintering. For example, lithium evaporation at elevated temperatures must be accounted for during synthesis to preserve stoichiometric stability [17]. Even trace impurities can destabilize phases, leading to issues like grain pulverization [17].
Allan Chemical Corporation supports high-temperature applications by providing high-purity technical and compendial chemicals (USP, FCC, ACS, NF) with comprehensive documentation. With over 40 years of experience in regulated industries such as pharmaceuticals, electronics, and ceramics, the company prioritizes sourcing directly from manufacturers to ensure consistent material quality. Their just-in-time delivery and flexible batch sizing further enhance reliability, offering the precision needed for applications where both electrochemical and thermal stability are critical to long-term device performance.
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Conclusion
Electrochemical stability plays a key role in managing voltage-driven degradation, while thermal stability ensures structural integrity. As temperature rises, electrochemical decomposition accelerates, reducing the operational voltage range of systems [6].
To achieve long-term performance, material selection must carefully balance these two stability factors. For high-temperature applications, monitoring changes at active sites during operation is essential to predict operational lifetimes [5]. Pietro Papa Lopes of Argonne National Laboratory highlights this by stating:
"Stability relates to how functional properties change over time during operation" [5].
This insight is particularly important in regulated industries where devices must remain reliable for decades. For example, fuel cells are expected to operate for over 250,000 hours, while pharmaceutical manufacturing equipment must maintain performance across extended timeframes. Both rely on materials that combine electrochemical durability with thermal endurance.
Material selection often involves navigating trade-offs. Advanced options like vanadium-based cathodes and ionic liquids demonstrate how materials can achieve wide electrochemical windows while maintaining thermal stability at temperatures exceeding 400°C (752°F) [6][19].
Allan Chemical Corporation supports these demanding applications with high-purity technical and compendial-grade chemicals (USP, FCC, ACS, NF), accompanied by detailed documentation. With over 40 years of experience serving industries like pharmaceuticals, electronics, and ceramics, the company ensures consistent quality and precision in chemical sourcing – critical for systems where both electrochemical and thermal stability are key to long-term success.
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
FAQs
How do I choose voltage limits that stay inside a material’s ESW?
To determine voltage limits within a material’s Electrochemical Stability Window (ESW), start by identifying the electrolyte’s stability range, which is defined by the breakdown limits of its cations and anions. Set the voltage limits to fall within this range – between the lower and upper bounds – to prevent unwanted oxidation or reduction reactions.
Ensure accuracy by using precise measurement techniques and applying correction factors when necessary. Always validate your findings with dependable methods to avoid misjudging the stability range, which could lead to errors in application.
Why can electrochemical breakdown lead to thermal runaway?
Electrochemical breakdown can lead to thermal runaway by unleashing energy at a rapid pace, producing intense heat. This excessive heat initiates uncontrolled exothermic reactions, causing a sharp temperature rise. As the system continues to overheat, a feedback loop develops, amplifying both the reactions and heat generation. This chain reaction can create hazardous conditions, potentially resulting in fires or explosions due to the uncontrollable temperature escalation.
Which tests should I run to evaluate stability at both high voltage and high temperature?
To assess how materials hold up under high voltage and temperature, thermal stability tests such as thermogravimetric analysis (TGA) are essential. TGA helps measure decomposition behavior and the risk of thermal runaway. For electrochemical stability, techniques like cyclic voltammetry or constant voltage lifetime testing are effective in determining the electrolyte’s voltage limits. Combining these methods provides a detailed understanding of stability under challenging conditions.





Comments are closed