Protein-based biopharmaceuticals are highly effective yet fragile, requiring stabilization to maintain their therapeutic properties and ensure patient safety. Without proper stabilizers, proteins can degrade, aggregate, or lose potency, leading to reduced effectiveness and potential immune responses. Stabilizers like Sucrose, Trehalose, and Recombinant Human Serum Albumin (rHSA) play a critical role in protecting proteins from physical and chemical stressors during production, storage, and administration.
Key Stabilizers and Their Functions:
- Sucrose: Protects proteins during freeze-drying by replacing water molecules. Common in liquid and lyophilized formulations but may cause glycation at high temperatures.
- Trehalose: Offers better thermal stability than sucrose, ideal for heat-sensitive formulations. However, it may crystallize during freeze-thaw cycles.
- rHSA: Prevents aggregation and provides antioxidant protection. Suitable for specialized applications like radiolabeled drugs but is costly and complex to regulate.
Emerging Stabilizer Technologies:
- Designer Sugars: Combine disaccharides with polysaccharides for enhanced hydrogen bonding and stability.
- Amino Acid-Based Ionic Liquids: Biocompatible solvents that protect proteins under extreme conditions.
- Stimuli-Responsive Polymers: Adjust protective mechanisms based on environmental changes, offering tailored protein stability.
Stabilizer selection depends on the protein’s specific challenges, such as thermal stress, aggregation risk, or storage conditions. Proper formulation ensures the drug’s effectiveness and safety, supported by high-purity stabilizers sourced from reliable suppliers.
Mechanisms of aggregation and stabilisation
Main Types of Protein Stabilizers
Protecting protein-based biopharmaceuticals requires careful selection of stabilizers, each tailored to address specific challenges like aggregation, oxidation, or thermal stress. Whether in liquid or lyophilized formulations, these stabilizers play a critical role in maintaining protein stability. Below is an overview of commonly used stabilizers and their mechanisms.
Sucrose: A Versatile Sugar-Based Stabilizer
Sucrose is widely used to protect proteins, particularly during freeze-drying processes. It forms hydrogen bonds with protein surfaces, effectively replacing water. In liquid formulations, sucrose enhances the hydration shell through a process called preferential exclusion. For example, Abatacept formulations utilize sucrose in liquid form and maltose in lyophilized form, showcasing sucrose’s adaptability [5].
In lyophilized formulations, a typical stabilizer-to-protein molar ratio is 360:1 for antibodies [5]. However, sucrose does have limitations. It can hydrolyze into reducing sugars, which may lead to glycation. A study on a recombinant monoclonal IgG2 antibody revealed glycation within one month at 99°F in sucrose-based formulations [5]. Additionally, some sucrose batches contain nanoparticulate impurities that might either mimic protein aggregates during analysis or even promote aggregation [1].
Trehalose: Thermal Stability at Higher Temperatures
Trehalose offers a compelling alternative to sucrose, particularly in situations involving heat stress. With a glass transition temperature (Tg) of approximately 176°F – significantly higher than sucrose’s 138°F – it maintains its protective properties at elevated temperatures [5]. This makes trehalose especially effective in forming a rigid glass matrix that limits protein unfolding by restricting molecular motion [1].
Trastuzumab (Herceptin) incorporates 60 mM trehalose as a lyoprotectant, benefiting from its higher Tg to enable more energy-efficient lyophilization [5].
Unlike sucrose, trehalose is chemically stable and does not cause glycation. However, it has a drawback: trehalose can crystallize during freeze-thaw cycles, whereas sucrose remains amorphous under similar conditions [5]. For optimal stability, lyophilized products containing trehalose should be stored at temperatures roughly 54°F below its Tg [1].
Recombinant Human Serum Albumin (rHSA): A Multi-Functional Option
Recombinant human serum albumin (rHSA) serves as a versatile stabilizer, offering multiple protective mechanisms. As a naturally occurring protein in human plasma, rHSA prevents aggregation by blocking adsorption onto vial surfaces and air-liquid interfaces [3]. It also functions as an antioxidant, making it particularly effective for stabilizing radiolabeled drugs and improving the solubility of poorly soluble peptides [3].
"As a result of this multifunctionality, in some cases, it is possible to reduce the number of excipients required to stabilize a biologic drug." – Jens T. Bukrinski, Formulation Scientist, Novozymes [3]
rHSA’s natural origin reduces immunogenic risks compared to synthetic surfactants. Unlike polysorbates, which are prone to hydrolysis, auto-oxidation, and contamination with trace peroxides, rHSA offers a safer alternative [3][4]. It is especially useful in cases where traditional detergents fail, such as stabilizing virus-like particles, where polysorbates may disrupt protein complexes [3].
This information is provided for educational purposes. Always consult regulatory guidelines and professionals for sourcing and formulation decisions.
New Stabilizer Technologies
The biopharmaceutical industry is advancing beyond traditional stabilizers like sucrose and trehalose. New engineered molecules are now providing stronger protection, better thermal stability, and improved performance under challenging conditions. These advancements address the limitations of conventional formulations, especially for high-value protein drugs that require long shelf lives or need to endure harsh environments. By building on hydrogen-bonding principles, these approaches tackle modern formulation challenges. Below are three approaches that significantly improve protein stabilization compared to traditional methods.
Designer Sugars with Enhanced Hydrogen Bonding
Modified sugar systems are now surpassing single-component stabilizers. A notable method blends small disaccharides with larger polysaccharides such as inulin, dextran, or pullulan. This combination leverages the high glass transition properties of polysaccharides and the robust hydrogen bonding capabilities of smaller sugars. Mensink et al. explain, "Smaller-sized sugar was found to be more flexible molecularly, thereby forming larger hydrogen bonds, which provided better stability and were less affected by steric obstruction" [7].
Alkylsaccharides, which pair sugars with long-chain fatty acids, are emerging as strong alternatives to traditional surfactants. For instance, polysorbates are used in 90% of approved monoclonal antibody formulations [1]. Edward T. Maggio, President and CEO of Aegis Therapeutics, highlights that "Alkylsaccharides are as effective, and in some cases, more effective than the polysorbates for the prevention of aggregation, and they are completely free of any oxidative damage problems" [3]. Another innovative sugar-based stabilizer is (2-hydroxypropyl)-β-cyclodextrin, a cyclic oligosaccharide with a hydrophobic cavity and hydrophilic surface that protects proteins from mechanical stress and surface-induced aggregation.
Beyond these engineered sugars, ionic liquids present a groundbreaking stabilization approach.
Amino Acid-Based Ionic Liquids
Amino acid-based ionic liquids are redefining protein stabilization, particularly under extreme conditions. These biocompatible compounds interact with proteins through van der Waals forces and hydrogen bonding, offering effective protection. For example, choline valinate increased insulin’s melting temperature by nearly 13°C, while choline dihydrogen phosphate raised the melting temperature of trastuzumab by over 21°C [6]. Banerjee et al. demonstrated in 2018 that choline geranate (CAGE) could stabilize an oral insulin formulation for four months under refrigeration and two months at room temperature [6]. With over one million possible cation–anion combinations, these designer solvents can be tailored to meet the needs of specific protein drugs. At a concentration of 50 mM, choline valinate completely suppressed amyloid and fibril formation in human recombinant insulin [6]. These choline-based ionic liquids are biodegradable and exhibit low cytotoxicity compared to conventional imidazolium-based options.
Stimuli-Responsive Polymers
Stimuli-responsive polymers and chemical chaperones, such as betaine and specific trehalose derivatives, adjust their protective mechanisms based on changes in temperature, pH, or osmotic stress, helping to preserve native protein structures [2]. Synthetic amphiphilic polymers are also being developed to replace polysorbates, offering protection against interface-mediated unfolding without the risks of hydrolysis or auto-oxidation. These responsive systems hold promise for creating self-adjusting formulations that provide consistent protection throughout storage and administration.
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
sbb-itb-aa4586a
Stabilizer Comparison
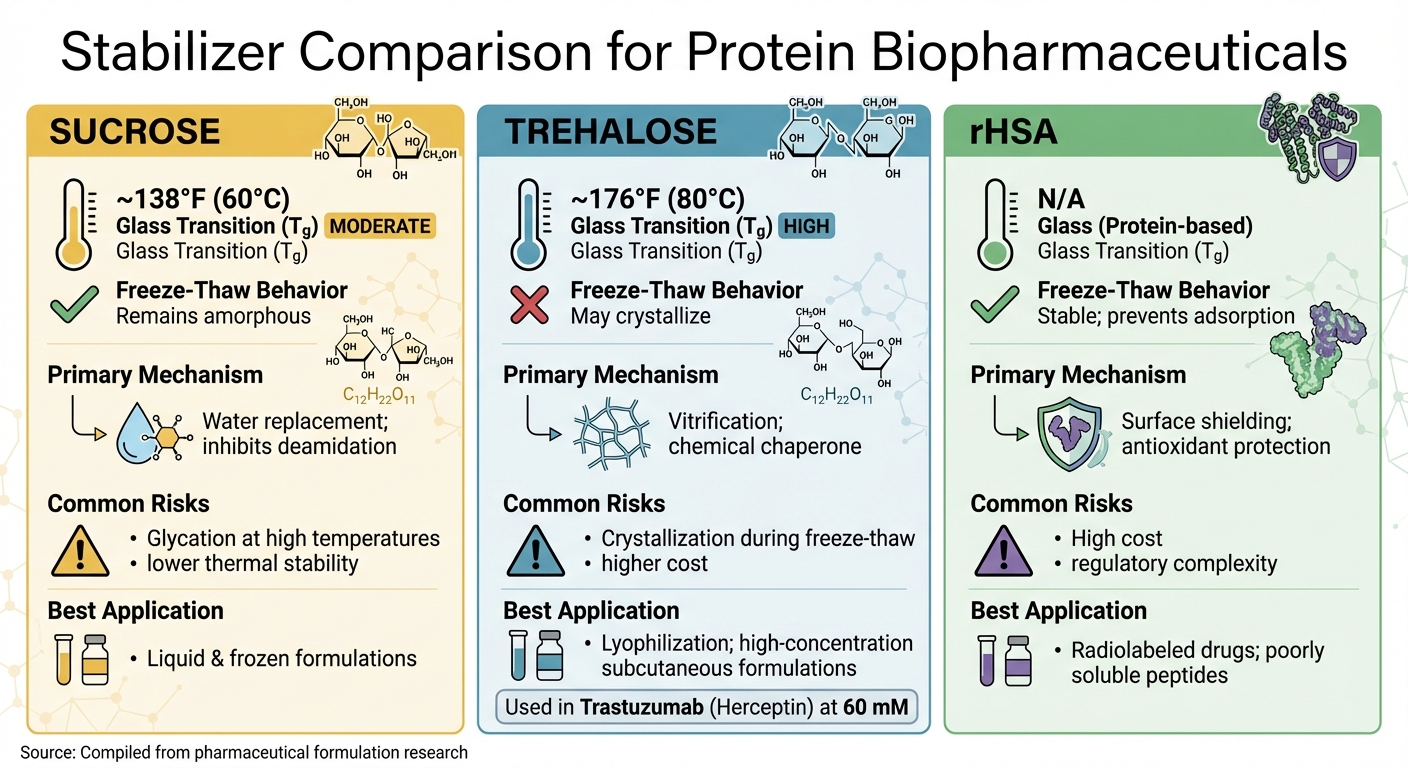
Protein Stabilizer Comparison: Sucrose vs Trehalose vs rHSA
Choosing the right stabilizer for protein biopharmaceuticals means evaluating how each option performs under specific conditions. Sucrose, trehalose, and recombinant human serum albumin (rHSA) are commonly used stabilizers, each offering unique benefits depending on formulation, storage, and processing needs.
Sucrose stands out for its performance during freeze–thaw cycles. Its ability to stay amorphous during these cycles helps protect proteins from damage caused by crystallization [5]. This makes it a great choice for liquid and frozen formulations. However, it does carry a glycation risk at high temperatures [5]. With a glass transition temperature (Tg) of about 138°F (60°C), sucrose provides moderate thermal stability, allowing for versatile use in different applications [5].
Trehalose offers better thermal stability compared to sucrose, with a Tg of approximately 176°F (80°C) [5]. This higher Tg supports more energy-efficient lyophilization (freeze-drying) processes and ensures stability under high-temperature or humid storage conditions. For example, Trastuzumab (Herceptin) uses 60 mM trehalose in its commercial lyophilized formulation to enable quicker and more efficient drying [5]. On the downside, trehalose can crystallize during freeze–thaw cycles and is generally more expensive than sucrose [5].
Recombinant human serum albumin (rHSA) serves as a versatile stabilizer by preventing surface adsorption, reducing protein self-association through preferential hydration, and providing antioxidant protection [3].
"Because rHSA is a natural protein in the body, it is safer than other alternative surfactant excipients, such as synthetic polymers, because it has a low risk for immunogenicity" – Jens T. Bukrinski, Formulation Scientist, Novozymes [3]
Despite these benefits, rHSA’s high cost and regulatory complexities limit its use to specialized applications, such as radiolabeled drugs or formulations for poorly soluble peptides [3].
The table below provides a quick comparison of these stabilizers, summarizing their key properties and use cases.
Stabilizer Comparison Table
| Stabilizer | Glass Transition (Tg) | Freeze–Thaw Behavior | Primary Mechanism | Common Risks | Best Application |
|---|---|---|---|---|---|
| Sucrose | Moderate (~138°F / 60°C) [5] | Remains amorphous [5] | Water replacement; inhibits deamidation [8][1] | Glycation at high temp; lower thermal stability [5] | Liquid & frozen formulations [5] |
| Trehalose | High (~176°F / 80°C) [5] | May crystallize [5] | Vitrification; chemical chaperone [2][1] | Crystallization; higher cost than sucrose [5] | Lyophilization; high-concentration subcutaneous formulations [5] |
| rHSA | N/A (Protein-based) | Stable; prevents adsorption [3] | Surface shielding; antioxidant [3] | High cost; regulatory complexity [3] | Radiolabeled drugs; poorly soluble peptides [3] |
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Choosing the Right Stabilizer
Selecting the right stabilizer for your protein formulation involves matching the specific challenges your protein faces with the appropriate excipients. As Jay Kang, Director of Analytical and Formulation Development at Patheon, puts it:
"The quality of the excipients can have a dramatic impact on protein stability." [3]
Proteins can encounter stresses like mechanical agitation, thermal exposure, or drying during formulation and storage. For mechanical stress, surfactants are often the go-to choice. Thermal stress and lyophilization (freeze-drying) are best managed with sugars, which help maintain stability under these conditions [1][3]. High-concentration formulations, which can increase viscosity, often require a mix of amino acids and anionic agents to maintain stability and reduce viscosity [9].
The choice of formulation state – liquid or lyophilized – also plays a pivotal role. Liquid formulations are more prone to chemical and physical instability due to higher molecular mobility. Lyophilized products, on the other hand, must endure the stresses of freezing and drying, which necessitate stabilizers designed for these specific challenges [1][3]. Preformulation studies are essential to identify degradation pathways, fine-tune pH and ionic strength, and determine stabilizer levels using a design-of-experiment (DoE) approach. These studies also help uncover synergistic interactions between excipients [3].
Allan Chemical Corporation‘s Stabilizer Sourcing Services
Allan Chemical Corporation addresses these formulation challenges by providing compendial-grade stabilizers that meet rigorous purity and documentation standards, including USP, FCC, ACS, and NF. With over 40 years of experience in regulated industries, they ensure that every stabilizer sourced comes with comprehensive documentation, such as Certificates of Analysis and Safety Data Sheets, simplifying regulatory compliance and risk management.
Their services are designed to support both early-stage development and large-scale production. Flexible batch sizes and just-in-time delivery help meet the needs of diverse projects. Additionally, their technical support team is available to assist with documentation and provide guidance on stabilizer selection, enabling formulation scientists to tackle quality control and regulatory complexities with confidence. For more information, visit https://allanchems.com to discuss your stabilizer requirements.
This content is for informational purposes only. Always consult official regulations and qualified professionals when making formulation or sourcing decisions.
FAQs
What are the pros and cons of using sucrose as a stabilizer in protein-based biopharmaceuticals?
Sucrose plays a key role as a stabilizer in protein-based biopharmaceuticals, primarily because it helps maintain protein structure. This is achieved through a process called preferential exclusion, where sucrose interacts with the surrounding environment to preserve the protein’s integrity. Additionally, sucrose serves as both a cryoprotectant and lyoprotectant, shielding proteins during freezing, drying, and storage.
That said, sucrose isn’t without its challenges. Pharmaceutical-grade sucrose can sometimes contain nanoparticle impurities. These impurities may trigger protein aggregation or fragmentation, potentially undermining stability and complicating analytical procedures. Even with these drawbacks, sucrose remains a widely used stabilizer in many protein formulations.
How do amino acid-based ionic liquids improve the stability of protein-based biopharmaceuticals?
Amino acid-based ionic liquids serve as stabilizers that help preserve the stability of protein-based biopharmaceuticals. They work by preventing issues like protein unfolding and aggregation, which can compromise the effectiveness of therapeutic proteins. These biocompatible compounds create a protective setting that safeguards the structure and function of proteins during storage and application.
Their adaptability makes them a strong option for advanced formulations, ensuring consistent performance in stabilizing delicate protein-based products.
What makes recombinant human serum albumin (rHSA) such an effective stabilizer for protein-based biopharmaceuticals, even with its high cost?
Recombinant human serum albumin (rHSA) plays a crucial role as a stabilizer, offering protection to proteins against both physical and chemical degradation. It helps reduce non-specific adsorption, limits the formation of amyloid-like aggregates, and provides antioxidant properties. These functions are essential for maintaining the stability and efficacy of biopharmaceutical formulations.
Although rHSA is more expensive, its range of benefits makes it a reliable option for preserving the quality and performance of sensitive protein-based therapeutics.





Comments are closed