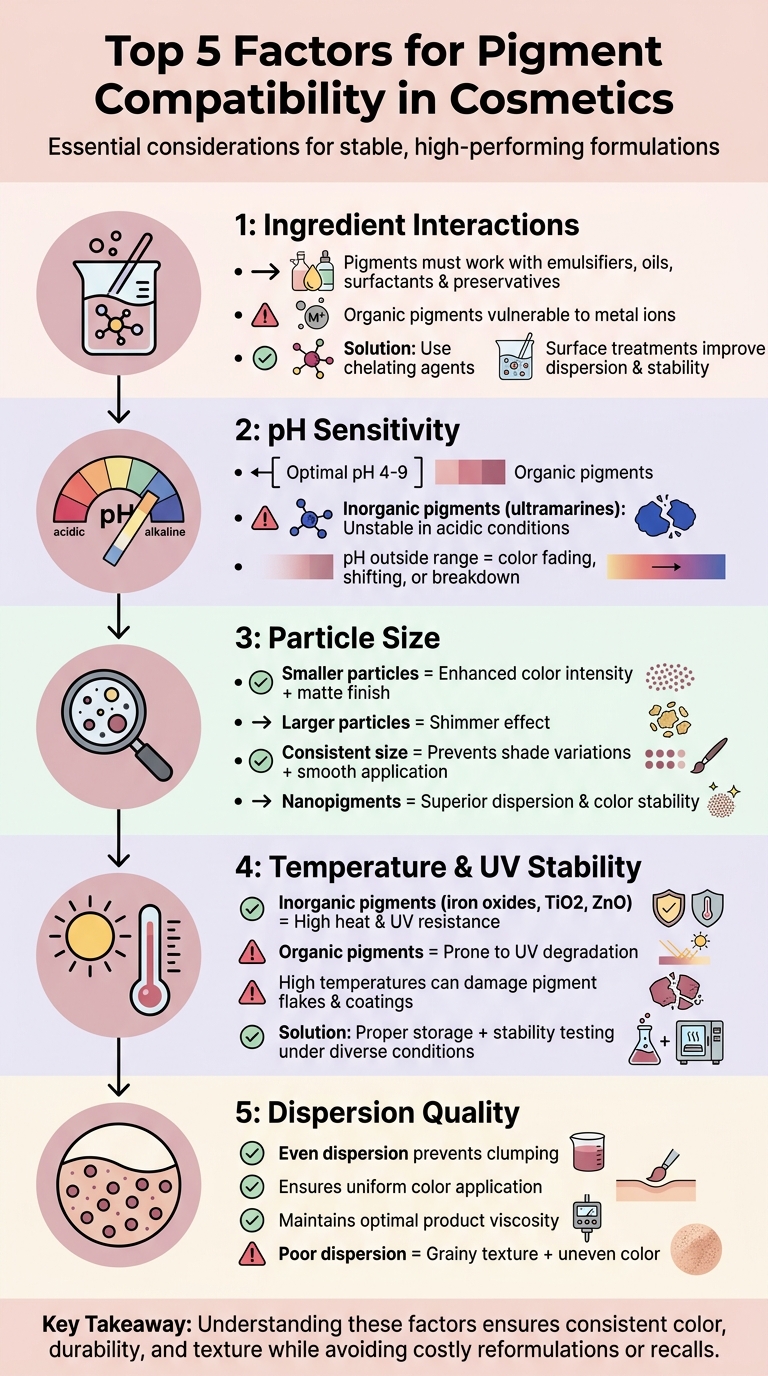
When formulating cosmetics, pigment compatibility is critical to achieving stable, high-performing products. Incompatible pigments can lead to issues like color fading, uneven application, or product separation. Here’s what you need to know:
- Ingredient Interactions: Pigments must work well with emulsifiers, oils, surfactants, and preservatives. Organic pigments are prone to degradation from metal ions, which can be mitigated with chelating agents. Surface treatments improve pigment dispersion and stability.
- pH Sensitivity: Organic pigments perform best between pH 4–9, while some inorganic pigments, like ultramarines, destabilize in acidic conditions.
- Particle Size: Smaller particles enhance color intensity and create matte finishes, while larger ones add shimmer. Consistent particle size prevents shade variations and ensures smooth application.
- Temperature & UV Stability: High temperatures or UV exposure can degrade organic pigments. Proper storage and testing under diverse conditions help maintain pigment quality. This is especially critical when you formulate long-wear cosmetic colorants that require high stability.
- Dispersion Quality: Even pigment dispersion prevents clumping, ensures uniform color, and maintains optimal product viscosity.
Understanding these factors ensures cosmetic products deliver consistent color, durability, and texture while avoiding costly reformulations or recalls.
5 Critical Factors for Pigment Compatibility in Cosmetic Formulations
1. How Pigments Interact with Other Ingredients
Interaction with Formulation Components
To create stable and effective formulations, it’s crucial to understand how pigments interact with other ingredients. Pigments need to work seamlessly with emulsifiers, oils, surfactants, and preservatives to maintain stability. Organic pigments, for instance, are particularly vulnerable to metal ions commonly present in formulations. These ions can cause pigments to degrade or become unstable. To counteract this, formulators often use chelating agents, which bind to metal ions and help preserve pigment integrity.
Surface treatments also play a major role in enhancing pigment compatibility. Techniques such as coating pigments with lecithin, esters, lauroyl lysine, or zinc and magnesium stearates improve their ability to work with both oil and water phases in emulsions. Additional chemical modifications, like those involving nylon or polyacrylates, further enhance pigment dispersion and improve texture. These treated pigments make formulation easier by blending more uniformly, dispersing consistently, and resisting moisture absorption. It’s also important to consider formulation conditions, particularly pH, as they significantly affect pigment stability.
Stability Under pH and Environmental Conditions
The stability of pigments depends not only on their interactions with other ingredients but also on maintaining the right pH and environmental conditions. For example, organic pigments generally perform best in formulations with a pH between 4 and 9 [1]. Outside this range, problems like color fading, shifting, or breakdown can occur. On the other hand, inorganic pigments, such as ultramarines, are particularly sensitive to acidic environments and become unstable at low pH levels [1]. A foundation with a pH of 3.5, for instance, could destabilize ultramarine pigments, leading to noticeable color changes over time.
This sensitivity isn’t limited to pH. Preservatives, antioxidants, and other ingredients containing trace metals can also affect pigment performance. To avoid such issues, it’s essential to finalize the formulation of the base product – including emulsifiers, oils, and surfactants – before selecting pigments. This ensures that the pigments deliver the intended opacity and shimmer without compromising the stability of the overall product [1].
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
sbb-itb-aa4586a
2. Color Strength, Shade Accuracy, and Particle Size
Particle Size and Dispersion Quality
When it comes to creating flawless color in cosmetic formulations, understanding particle size and how well pigments disperse is key. Pigments are insoluble molecules that need a binder to distribute evenly. Their color comes from their chemical structure and how they absorb or reflect light [4]. Smaller pigment particles provide more intense color and precise shades, while larger particles can result in uneven coverage and duller finishes.
Nanopigments, which are ultra-fine particles, excel at even dispersion, improving color stability, texture, and adaptability in formulations [3]. However, achieving this even dispersion is critical. Poorly dispersed pigments can clump together, leading to a cakey texture and uneven color application [2]. This makes dispersion quality a cornerstone of effective pigment performance.
For inorganic pigments like Titanium Dioxide, particle size plays a pivotal role in delivering opacity and brightness by reflecting and scattering light [3]. To achieve a smooth texture and even application, pigment dispersions must be tailored to the specific formulation base [3]. Consistent particle size also helps prevent shade variations between production batches. Many manufacturers now use advanced tools like color formulation software and digital color standards to monitor sample history, assess consistency across different lighting conditions, and detect potential color shifts during early stability testing [2]. High-quality dispersion not only ensures vibrant and uniform color but also strengthens the formula’s resilience to environmental changes.
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Working with Colorants and Pigments
3. pH Stability and Chemical Resistance
When developing cosmetic formulations, understanding both pH stability and chemical resistance is crucial for maintaining pigment performance. The pH of a formula directly influences how well pigments retain their color and structure. Organic colorants are most stable in moderate pH ranges [1]. While inorganic pigments are generally more durable, they can still degrade or change color under extreme pH conditions [1].
For example, Ultramarines, a type of inorganic pigment, lose their vibrancy in acidic environments. If the pH drops below 4, these pigments can break down, compromising the final product’s appearance [1]. This makes it essential to finalize the formulation’s base and confirm its pH stability for effect pigments before selection.
Beyond pH levels, chemical interactions within the formulation also pose challenges to pigment stability.
Interaction with Formulation Components
Chemical interactions between pigments and other formulation ingredients can significantly affect pigment integrity. For instance, metal ions can destabilize organic pigments, leading to color fading or changes [1]. To counteract this, adding chelating agents helps stabilize organic pigments without disrupting active ingredients [1].
Effect pigments, known for their unique visual properties, require special handling. Certain solvents, emollients, or surfactants can damage the protective coatings on these pigments. This can result in issues like phase separation, clumping, or unwanted color shifts [5]. Additionally, high-shear mixing or exposure to excessive heat during production can fracture pigment flakes or strip their coatings, reducing both their durability and visual appeal [5].
To preserve the quality of effect pigments:
- Add them after high-temperature or high-shear processing steps [5].
- Pre-wet them in a compatible carrier to ensure even dispersion and prevent clumping [5].
This information is intended for guidance only. Always consult official regulations and experts when making formulation or sourcing decisions.
4. Temperature and Environmental Stability
Managing production temperature and environmental conditions is just as important as understanding ingredient interactions and pH stability when it comes to pigment performance. Temperature fluctuations during manufacturing and storage can significantly impact how pigments perform. For example, a pigment that appears vibrant in a controlled lab setting might lose its luster or stability during large-scale production if temperature controls are neglected. High temperatures during batching can damage pigment flakes or strip away protective coatings, which diminishes the visual quality that consumers expect from effect pigments.
Temperature and UV Resistance
Exposure to heat and light presents major challenges to pigment durability. Inorganic pigments like iron oxides, titanium dioxide, and zinc oxide are well-known for their ability to withstand both high temperatures and UV light, making them highly durable under stressful environmental conditions [2]. On the other hand, organic (synthetic) pigments, while offering vivid and transparent colors, are more prone to UV-induced degradation [2]. This vulnerability can cause gradual color changes over time [2].
"Organic colorants are all transparent on the skin, with various levels of chemical and physical stability."
– Belinda Carli, Author, SpecialChem [1]
To reduce UV-related damage, store master color standards in dark, dry spaces and conduct stability testing under a variety of light sources – both natural and artificial. This ensures that colors remain consistent across different lighting conditions [2]. Additionally, incorporating chelating agents into formulations can help stabilize organic pigments by protecting them from chemical breakdown.
Beyond UV exposure, maintaining stable environmental conditions is crucial in preserving pigment integrity.
Stability Under pH and Environmental Conditions
Temperature and UV exposure aren’t the only factors that can impact pigment performance. Even small environmental changes during production can lead to issues such as phase separation, agglomeration, or undesirable color shifts. Temperature fluctuations, in particular, can worsen compatibility challenges, making it essential to closely monitor and adjust both temperature and mixing speeds during batching to protect the delicate structure of pigments.
"Small measurement errors or environmental variances during formulation can turn a promising lab result into a dull, unstable product in production."
– Vivify Beauty Care [5]
To mitigate these risks, pilot batches should be conducted before full-scale production. This allows manufacturers to identify how temperature variations during scale-up might affect the final product’s appearance and stability. Performing both accelerated and real-time stability tests under different conditions is also key to validating pigment performance. By simulating the final product’s intended environment in the lab, manufacturers can assess how pigments will react to the base’s temperature and pH, ensuring the desired outcome [5].
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
5. Dispersion Quality and Effects on Viscosity
Beyond factors like pH and temperature, the quality of pigment dispersion plays a key role in ensuring pigment stability and controlling viscosity in cosmetic formulations. Achieving even pigment dispersion is essential for vibrant, consistent color in products. Poor dispersion can lead to clumping, resulting in a grainy texture and uneven application. Additionally, improper dispersion often increases viscosity, making the product harder to apply and diminishing overall performance.
Particle Size and Dispersion Quality
The size of pigment particles significantly affects how well color transfers to the skin, a property often called "color pay-off." Nanopigments have revolutionized this aspect, offering better color stability and versatility compared to larger particles [3]. These tiny particles ensure smoother dispersion, which not only preserves color accuracy but also helps maintain optimal viscosity for improved application. For functional ingredients like Titanium Dioxide and Zinc Oxide, achieving the right particle size and dispersion is critical for consistent color and reliable UV protection [2][3]. Proper dispersion enhances both the color pay-off and the interaction with the formulation base, allowing precise control over viscosity.
Interaction with Formulation Components
Dispersion must be adapted to the formulation base – whether it’s water, oil, or an emulsion – to achieve a uniform texture and maintain viscosity [3]. For example, a dispersion designed for an oil-based product may not perform well in a water-based serum, potentially causing separation or uneven color. Incorporating chelating agents and other additives helps stabilize the dispersion, reducing batch-to-batch inconsistencies and preventing color drift [2]. This careful control ensures a high-quality product with reliable performance.
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Conclusion
Creating successful cosmetic formulations requires careful management of how pigments interact with other ingredients, alongside factors like color strength, particle size, pH stability, temperature resistance, and dispersion quality. These five elements directly influence the appearance, stability, and sensory qualities of the final product. Even small measurement errors can disrupt pigment stability during production, leading to issues like streaky finishes, unstable colors, or "muddy" tones [5].
Precision is crucial throughout the production process. Beyond achieving the desired look, ensuring pigment compatibility helps maintain the intended texture and prevents problems like phase separation, settling, or clumping. For liquid formulations, this can mean avoiding shifts from smooth, easy flow to thick, resistant textures. Stability depends on maintaining an optimal pH – typically between 4 and 9 for organic pigments – and protecting against heat, light, and metal ion degradation [1]. Additionally, particle size and surface treatments play a key role in determining how the product feels on the skin (e.g., velvety or gritty) and the level of shimmer or opacity it delivers [1][5].
Industry experts emphasize the importance of thorough testing:
"The key is testing pigments in your target base under realistic conditions, assessing factors such as pH, temperature, and texture, to identify potential issues early on." – Vivify Beauty Care [5]
A deep understanding of these factors is essential for developing high-quality cosmetic products. Partnering with reliable suppliers for pigments and ingredients is critical to avoid regulatory issues and costly recalls. Allan Chemical Corporation supports this need by offering specialty chemicals and pigments designed to meet stringent quality and stability requirements. With over 40 years of experience in regulated industries, Allan Chemical provides technical-grade and compendial-grade materials (USP, FCC, ACS, NF), along with detailed technical data sheets and expert guidance. This ensures formulators can choose compatible binders, solvents, and surfactants that work seamlessly with specific pigment chemistries, ensuring consistent results and customer satisfaction.
This information is for educational purposes only. Always consult official regulations and qualified professionals when making sourcing or formulation decisions.
FAQs
How do I know if a pigment will react with my preservatives or oils?
To check if a pigment will interact with preservatives or oils, assess its stability across varying pH levels – commonly between 4.5 and 8.5 in cosmetic products – and under different temperature conditions. Techniques such as accelerated aging tests and freeze-thaw cycles help predict how the pigment will perform over time. Opting for pigments with surface treatments that align with your formulation can also enhance stability and reduce potential reactivity problems.
What tests should I run to check pigment stability under heat and UV?
To evaluate how pigments hold up under heat and UV light, you can carry out accelerated aging tests and UV stability tests. Accelerated aging involves subjecting pigments to higher-than-normal temperatures, ranging from 32°F to 100°F, to mimic extreme storage conditions. This process helps identify any shifts in color, intensity, or separation. Meanwhile, UV stability tests expose pigments to ultraviolet light to check for signs of fading, discoloration, or breakdown. These tests are crucial for ensuring pigments maintain their performance in actual cosmetic formulations.
How can I improve pigment dispersion without thickening the formula?
To improve pigment dispersion without making the mixture thicker, consider using wetting agents or dispersants. These help coat pigment particles evenly, reducing clumping. Mechanical techniques such as high-shear mixing or ball milling can also break apart particle agglomerates effectively. Additionally, choosing the right binder, maintaining the appropriate particle size, adjusting the pH, and ensuring the formulation components work well together can enhance stability and uniformity while keeping viscosity unchanged.





Comments are closed