Specific gravity measures a material’s density relative to water, typically at a reference temperature of 25°C. This property is essential across industries like pharmaceuticals, food production, and electronics, where precision directly impacts quality and compliance. For example:
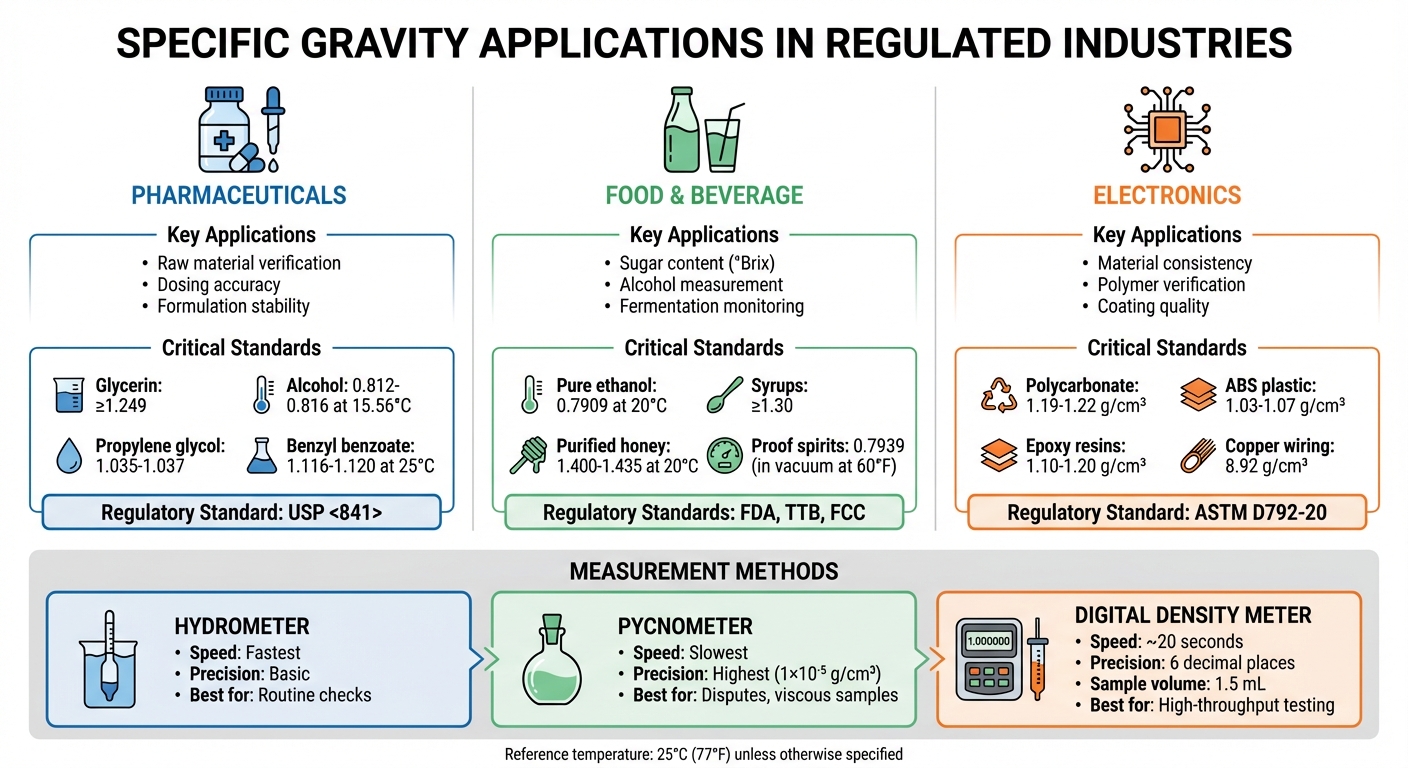
- Pharmaceuticals: Ensures proper dosing, raw material purity, and formulation stability. Glycerin, for instance, must meet a specific gravity of not less than 1.249.
- Food and Beverage: Used to measure sugar (°Brix) and alcohol content, ensuring product consistency and safety. Pure ethanol has a specific gravity of approximately 0.7909 at 68°F.
- Electronics: Validates material consistency in polymers and coatings, ensuring thermal stability and structural integrity.
Regulatory standards like USP <841> and ASTM guidelines govern specific gravity testing methods, such as pycnometers and digital density meters. These methods ensure accuracy, with digital meters offering results in seconds and precision up to six decimal places. High-quality, compendial-grade chemicals are critical for calibration and testing accuracy, supporting compliance and product reliability.
Specific Gravity Standards and Measurement Methods Across Regulated Industries
How Do You Measure Specific Gravity? – Chemistry For Everyone
Specific Gravity in Pharmaceutical Manufacturing
In pharmaceutical manufacturing, specific gravity plays a crucial role in ensuring that every ingredient meets stringent quality standards before entering production. This measurement serves as a unique identifier, or "fingerprint", for pure substances, helping quality control teams confirm material identity and detect potential contamination or adulteration. By comparing measured values to USP monograph specifications, manufacturers verify compliance. For instance, testing a glycerin shipment ensures it meets the USP requirement of not less than 1.249 [1]. This level of precision in raw material verification forms the foundation for subsequent quality assurance processes.
Verifying Raw Material Quality
Specific gravity testing is essential for confirming the identity and purity of solvents, excipients, and active pharmaceutical ingredients (APIs) before they are incorporated into formulations. Each material has a defined range of acceptable values: alcohol must fall between 0.812 and 0.816 at 60.01°F (15.56°C), propylene glycol between 1.035 and 1.037, and benzyl benzoate between 1.116 and 1.120 at 77°F (25°C) [1]. This testing also measures solute concentrations in solvents, such as sugar or alcohol levels, which directly influence dosing accuracy in liquid formulations [1]. Substances like ethanol, acetone, glycerin, and castor oil are routinely tested upon receipt. Any deviation from specified ranges may indicate contamination or incorrect material identification. Once raw materials pass these tests, the focus shifts to ensuring formulation stability.
Formulation Stability and Solvent Selection
Specific gravity is a key factor in selecting solvents and carriers, as it helps predict how materials will interact and settle in formulations [1]. In oral and injectable suspensions, it ensures uniformity by controlling particle settling and maintaining homogeneity until administration. For topical products like creams, lotions, and gels, specific gravity affects spreadability and prevents emulsion separation over the product’s shelf life [1]. It also ensures consistency in liquid dosage forms, such as injectables and oral solutions, where precise active ingredient concentrations are critical for therapeutic effectiveness. This is especially important for high-potency drugs, where even slight variations can lead to dosing errors. In metered-dose inhalers (MDIs), specific gravity influences the pressure and flow characteristics of aerosol formulations, ensuring accurate dosing with each use [1].
Meeting Compendial Standards
USP General Chapter <841> defines specific gravity as the ratio of the weight of a liquid in air at 77°F (25°C) to the weight of an equal volume of water at the same temperature [4]. To meet these standards, manufacturers rely on either Method I (Pycnometer) or Method II (Oscillating Transducer Density Meter) [4]. The pycnometer method is the most precise, making it the preferred choice for samples that are too viscous or in cases of dispute [3]. Digital density meters, on the other hand, provide results in approximately 20 seconds with six-decimal precision [1]. However, for samples with viscosities exceeding 10 mPa·s, density corrections are necessary due to dampened oscillations [6]. Calibration for Method II requires the instrument to measure 0.9970 ± 0.0001 g/cm³ in degassed water at 77°F (25°C) [6]. Additionally, when using digital meters, manufacturers must ensure the software complies with FDA 21 CFR Part 11, which includes features like audit trails and electronic signatures to maintain data integrity [1].
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Specific Gravity in Food and Beverage Production
Specific gravity plays a crucial role in food and beverage manufacturing by ensuring product consistency, monitoring processes, and meeting regulatory requirements. This measurement helps determine the concentration of dissolved substances like sugar and alcohol, which directly affect the taste, safety, and market appeal of products [11]. Since specific gravity is a fixed property of pure substances under consistent temperature and pressure, any deviation can indicate the presence of fillers or impurities [9][10]. These principles form the foundation for advanced measurement techniques discussed later.
Measuring Sugar and Alcohol Levels
Dissolved sugar increases specific gravity above 1.000, typically measured in °Brix [5]. For instance, USP monographs require syrups and caramel flavorings to have a specific gravity of at least 1.30, while purified honey should range between 1.400 and 1.435 at 68°F (20°C) [1]. Alcohol, on the other hand, behaves differently – pure ethanol has a specific gravity of about 0.7909 at 68°F (20°C), so as alcohol concentration rises, the solution’s specific gravity decreases [5]. According to the Alcohol and Tobacco Tax and Trade Bureau (TTB), proof spirits are defined as liquids containing half their volume in ethyl alcohol, with a specific gravity of 0.7939 in a vacuum at 60°F [12]. For spirits with dissolved solids exceeding 400 mg/100 mL, manufacturers must adjust for solids, as they increase specific gravity and can obscure the true alcohol proof [12].
Fermentation Control and Adulteration Detection
Specific gravity is a key metric during fermentation, as it helps track progress and pinpoint when the process is complete. During fermentation, yeast converts dense sugars into less dense alcohol and carbon dioxide, causing the specific gravity to drop. This allows brewers and winemakers to monitor and control the process with precision [5]. It’s important to remove carbon dioxide before measuring, as gas bubbles can distort readings [7]. Beyond process control, specific gravity testing is a powerful tool for detecting adulteration. Deviations from expected values based on Food Chemicals Codex or USP standards can reveal unauthorized substances or significant contamination in raw materials [9][10]. Instruments like oscillating U-tube meters provide precision to five or six decimal places, making it possible to detect even minor adulteration [9].
FDA and Food Industry Standards

Compliance with industry standards is essential for quality assurance. The FDA mandates specific gravity data for direct food additives, while the TTB requires hydrometers to meet ASTM E 100-72 standards, with a maximum error margin of 0.0005 [8][10]. When submitting food additive petitions, manufacturers must include specific gravity as part of the substance’s "fingerprint", alongside properties like refractive index and pH [10]. Digital density meters, often used in regulated settings, feature software capabilities that align with FDA 21 CFR Part 11, ensuring data integrity through audit trails and electronic signatures [1]. Many manufacturers are moving away from manual hydrometer testing and adopting online vibration liquid density meters to cut operational costs [11].
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Specific Gravity in Electronics Manufacturing
Specific gravity testing plays a key role in quality control within electronics manufacturing, much like its application in pharmaceuticals and food production. This process ensures that critical materials such as polymers, ceramics, and coatings meet stringent density requirements. These specifications are vital for maintaining thermal stability and structural integrity in electronic components. According to ASTM D792-20, specific gravity testing helps "identify a material, to follow physical changes in a sample, to indicate degree of uniformity among different sampling units or specimens, or to indicate the average density of a large item" [15].
Assessing Material Consistency
Specific gravity testing provides insights into the uniformity of production batches, catching subtle inconsistencies that visual inspections might miss. For instance, polycarbonate used in electronic housings typically falls within a density range of 1.19–1.22 g/cm³, while ABS, common in consumer electronics, ranges from 1.03–1.07 g/cm³ [13]. These tests can reveal structural issues such as voids or uneven filler distribution that could compromise component durability. In polymers and coatings, specific gravity measurements help identify impurities, filler mismatches, or plasticizer loss, all of which can affect performance. For ceramic slurries used in high-performance substrates, monitoring specific gravity ensures the material’s consistency before firing and confirms that the final product meets density specifications needed for thermal performance and strength [14].
Component Quality Control
Beyond consistency checks, specific gravity testing serves as a quick screening tool before more detailed performance evaluations. Standards like ASTM D792 for solid plastics and ASTM D891 for liquid chemicals and coatings guide these processes [15][3]. Advanced digital U-tube density meters can measure specific gravity with precision up to five or six decimal places using as little as 1.5 mL of material [9][5].
For adhesives and coatings, such as epoxy resins, maintaining a density of 1.10–1.20 g/cm³ is critical for effective bonding and protecting circuitry. Similarly, copper wiring, with a density of about 8.92 g/cm³, and aluminum housings, at 2.70 g/cm³, must adhere to strict density specifications to ensure proper conductivity and structural strength. These tests are typically conducted at a controlled temperature of 23°C ±2°C [13][9].
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
sbb-itb-aa4586a
Measurement Methods and Standards
Accurate measurement methods are essential for maintaining the consistency and quality required in regulated industries. These industries depend on standardized techniques to meet strict compliance requirements. The choice of measurement method often hinges on factors like the desired precision, the sample’s viscosity, and the specific regulatory framework. Among the most commonly used techniques are the hydrometer, the pycnometer, and the digital density meter – each suited to different needs.
Common Measurement Techniques
The hydrometer method uses a floating, calibrated device to provide readings directly from its stem. This is the simplest and quickest option, making it ideal for routine checks in industrial settings where extreme precision isn’t necessary [2][5].
The pycnometer method involves a calibrated glass flask of known volume, allowing a sample’s mass to be compared with the mass of an equal volume of water. It offers exceptional accuracy, reaching up to 1×10⁻⁵ g/cm³. This method is often used as a "referee method" for resolving disputes or for testing samples with higher viscosity [3][4].
Digital density meters operate using oscillating transducers within a U-shaped borosilicate glass tube. They require only 1.5 mL of sample and deliver results in seconds, with errors as low as 1×10⁻⁵ g/cm³ and repeatability down to 1×10⁻⁶ g/cm³ [4]. Their minimal sample volume speeds up temperature stabilization compared to traditional methods. For high-viscosity samples, these meters apply viscosity compensation [6].
These techniques are critical for ensuring compliance with USP and ASTM standards.
Meeting USP and ASTM Standards
Regulated industries adhere to specific standards to validate measurement results. For example, pharmaceutical manufacturers must follow USP <841>, which defines specific gravity as the ratio of a liquid’s weight in air at 25°C to the weight of an equal volume of water at the same temperature [1][4]. USP <841> outlines two approved methods: Method I, which uses a pycnometer, and Method II, which employs an oscillating transducer or digital density meter. To comply with USP requirements, digital density meters must achieve measurement errors between 1×10⁻³ and 1×10⁻⁵ g/cm³. Daily calibration with degassed water at 25°C is mandatory, using a reference value of 0.997043 g/cm³.
ASTM standards address measurements for industrial chemicals and petroleum products. For instance, ASTM D891 specifies hydrometers (Method A) or pycnometers (Method B) for testing liquid industrial chemicals. Meanwhile, ASTM D4052 governs digital density meters used with petroleum distillates and viscous oils, accommodating viscosities up to 15,000 mm²/s. These standards also define operational limits, such as temperature and vapor pressure (typically below 100 kPa for digital meters), to prevent issues like bubble formation [16]. In highly regulated environments, such as those governed by FDA 21 CFR Part 11, digital meters must include features like audit trails, electronic signatures, and customizable user roles to ensure data integrity [1].
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
Chemical Sourcing for Accurate Testing
Using Compendial-Grade Chemicals
The precision of specific gravity testing hinges on the quality of the chemicals used as reference standards and in production processes. Compendial-grade chemicals – those meeting standards set by USP (United States Pharmacopeia), FCC (Food Chemicals Codex), ACS (American Chemical Society), and NF (National Formulary) – serve as the benchmark materials required to meet regulatory demands. Drug manufacturers and developers must establish specifications for excipients and active ingredients that align with these pharmacopeial requirements [17]. Using chemicals that fall short of these standards can undermine the accuracy of calibration efforts.
"The tests and compliance with these standards detailed in the reference methods constitute a basic requirement for the global manufacture, release, and distribution of pharmaceutical ingredients and specialties." – Sigma-Aldrich [17]
Subpar chemicals can lead to inaccurate measurements, which may result in regulatory issues. In the pharmaceutical industry, errors in specific gravity measurements caused by poor-quality chemicals can have serious consequences, such as incorrect dosing in injectable or oral solutions. These inaccuracies can also affect the density of suspensions and emulsions, potentially reducing therapeutic effectiveness and destabilizing the product [1]. This underscores the importance of sourcing high-quality chemicals, a practice that aligns with the robust strategies provided by Allan Chemical Corporation.
Allan Chemical Corporation‘s Support for Regulated Industries
Recognizing the critical role of compendial-grade chemicals in ensuring testing precision, Allan Chemical Corporation has been a trusted partner to regulated industries for over four decades. They supply both technical-grade and compendial-grade chemicals (USP, FCC, ACS, NF) with reliable, just-in-time delivery. Their sourcing-first approach ensures that manufacturers in sectors such as pharmaceuticals, food, and electronics receive high-purity materials exactly when needed, preventing production disruptions. By maintaining direct relationships with carefully vetted manufacturers, they help ensure consistent quality across all batches – an essential factor for precise specific gravity testing.
Allan Chemical Corporation’s specialty chemical solutions align with the requirements of USP <841> and ASTM standards, offering the reliability needed for accurate measurements. This dependable support enables regulated industries to achieve the precision necessary for global product release and distribution.
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
Conclusion
Specific gravity testing plays a key role in ensuring quality control across industries like pharmaceuticals, food and beverage, and electronics manufacturing. In the pharmaceutical sector, it helps maintain dosage accuracy in injectables and oral suspensions while ensuring formulations remain stable to avoid ingredient separation [1]. For food and beverage producers, specific gravity measurements are critical for monitoring sugar levels (Brix) and alcohol content, which are essential for managing fermentation processes [5]. In electronics, it ensures consistency in materials like polymers, ceramics, and coatings. These varied applications highlight the importance of precision in specific gravity measurements across different fields.
Precision in these measurements is not only crucial for product quality but also for compliance with strict regulatory standards. For instance, pharmaceutical manufacturing requires adherence to standards like USP <841>, which governs specific gravity testing for production and distribution [17]. Modern instruments now offer repeatability ranging from 1×10⁻⁴ to 1×10⁻⁶ g/cm³ and include features like audit trails and electronic documentation to support data integrity [4][1].
Accurate specific gravity testing also depends on the use of high-quality, compendial-grade chemicals. Calibration and production using chemicals that meet standards such as USP, FCC, ACS, or NF ensure traceability to international benchmarks. Using lower-grade materials can compromise both product quality and regulatory compliance. Companies like Allan Chemical Corporation supply the high-purity chemicals and reliable delivery schedules that regulated industries rely on to maintain both precision and consistency in testing and production.
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
FAQs
Why is specific gravity important in pharmaceutical manufacturing?
Specific gravity is essential in pharmaceutical manufacturing, serving as a key factor in maintaining the purity, concentration, and uniformity of liquid formulations. It offers a dependable way to confirm that products align with regulatory requirements and meet stringent quality control standards, including those set by the USP.
Through specific gravity measurements, manufacturers can identify variations in raw materials or finished products. This ensures compliance with regulations and safeguards patient safety, playing a crucial role in delivering pharmaceuticals that meet both industry demands and consumer trust.
How is specific gravity used to maintain consistency in food and beverages?
Specific gravity plays a key role in ensuring consistency in food and beverage production by measuring the density of liquids relative to water. This measurement allows producers to confirm ingredient concentrations, maintain quality, and adhere to industry regulations.
For instance, in the beverage industry, specific gravity is used to verify the sugar content in soft drinks or the alcohol levels in spirits. In food manufacturing, it ensures the uniformity of products like syrups and sauces, helping them meet both consumer expectations and regulatory guidelines.
Why is specific gravity essential in electronics manufacturing?
Specific gravity plays a key role in electronics manufacturing, as it directly influences the quality, consistency, and effectiveness of liquids used in critical processes. Take battery production, for example – here, the specific gravity of the electrolyte affects the battery’s voltage, capacity, and overall lifespan. Regular monitoring ensures these parameters stay within optimal ranges, even when operating conditions fluctuate.
Accurate specific gravity measurements also help ensure materials like adhesives, insulating fluids, and electrolytes meet industry standards. They can reveal impurities or contamination, making them essential for quality control. This attention to detail not only improves the reliability of electronic components but also enhances their safety.





Comments are closed