Industrial surfactants are chemical compounds designed to reduce surface and interfacial tension in a variety of applications, such as emulsification, wetting, dispersion, solubilization, and foam management. Selecting the right surfactant is critical to ensure consistent performance, avoid formulation failures, and meet regulatory standards. Here’s a quick guide to simplify the process:
- Define Application Needs:
- Identify the surfactant’s primary role (e.g., cleaning, emulsifying, wetting).
- Consider the substrate material and end-use requirements.
- Use the Hydrophilic-Lipophilic Balance (HLB) scale to match surfactant behavior to your formulation.
- Choose the Right Chemistry:
- Select from anionic, cationic, nonionic, or amphoteric surfactants based on charge and functionality.
- Match HLB values to your system (e.g., 3–6 for water-in-oil emulsions, 8–18 for oil-in-water).
- Test Compatibility and Stability:
- Evaluate performance under varying conditions (e.g., temperature, pH, water hardness).
- Check for interactions with other ingredients and equipment materials.
- Verify Regulatory Compliance:
- Ensure surfactants meet biodegradability and toxicity standards (e.g., EPA Safer Choice criteria).
- Review the Safety Data Sheet (SDS) for proper handling and safety protocols.
- Ensure Quality and Supply Reliability:
- Request Certificates of Analysis (COAs) for batch consistency.
- Confirm supplier reliability and lead times to avoid production delays.
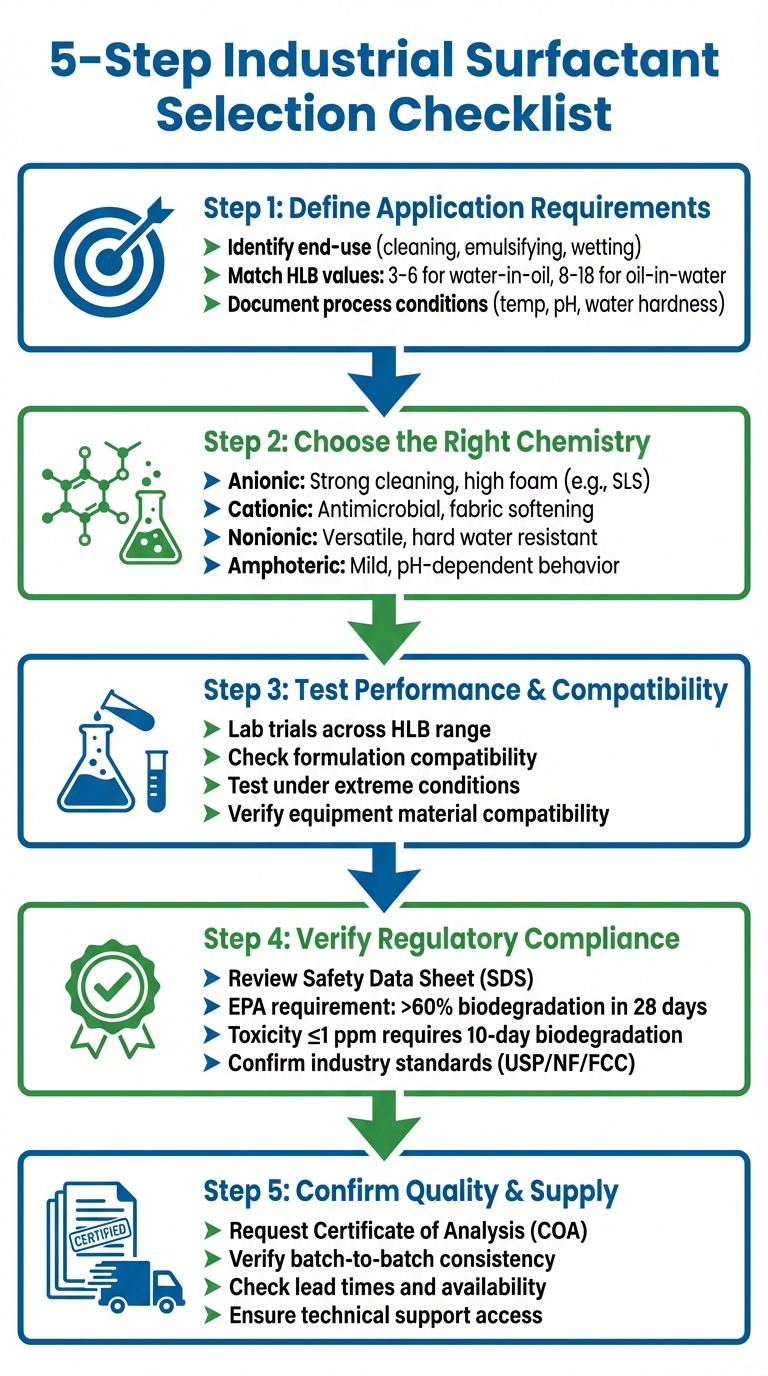
5-Step Industrial Surfactant Selection Process Checklist
Step 1: Define Application Requirements
Choosing the right surfactant starts with clearly defining the specific needs of your application. Skipping this step can lead to selecting a product that doesn’t align with your process, potentially causing inefficiencies or failures. Begin by documenting the functional requirements, setting measurable performance goals, and noting key process parameters. This clarity ensures your surfactant choice matches the intended purpose.
Identify the End-Use Application
The first step is to determine the primary role your surfactant will play. Will it clean industrial parts, stabilize an emulsion, wet a textile surface, or manage foam in a pharmaceutical product? Each task requires a different surfactant chemistry. For example:
- Industrial detergents often rely on anionic surfactants for their strong cleaning power and foaming properties.
- Fabric softeners and disinfectants typically use cationic surfactants, which provide antimicrobial and antistatic benefits.
- Nonionic surfactants are valued for their versatility, excelling in wetting and emulsification across a wide pH range, making them ideal for agrochemical sprays or pharmaceutical solubilization [1].
To guide your selection, consider the Hydrophilic-Lipophilic Balance (HLB) scale. Surfactants with HLB values of 3–6 are effective for water-in-oil emulsions, while those in the 8–18 range are better suited for oil-in-water systems. For textile and dyeing applications, wetting agents typically require HLB values between 7 and 9 [1]. Additionally, take into account the substrate material being treated and the cleanliness level required for subsequent steps. As outlined in ASTM D6361, the process must align with both the material and any follow-up processes [5].
Set Performance Targets
Once the application is defined, establish clear performance expectations. For instance, if you’re working with oil-solubilizing cleansers, refer to the HLB guidelines mentioned earlier. If foam generation is a priority – such as in personal care products – document the desired foam height and how long it should remain stable. On the other hand, if foam is problematic in your industrial process, opt for a low-foaming or defoaming surfactant.
Environmental compliance should also be factored in early. To meet EPA Safer Choice standards, surfactants typically need to achieve over 60% mineralization into CO₂ and water within 28 days. Additionally, if a surfactant has high acute aquatic toxicity (L/E/IC50 ≤1 ppm), it must biodegrade within a strict 10-day period [2]. For applications like electronics or precision cleaning, consider whether a residue-free performance is required after rinsing or evaporation [6].
Document Process Conditions
The performance of a surfactant can be heavily influenced by the conditions in which it operates. Record key factors such as temperature, pH, salinity, and water hardness. These parameters affect solubility and stability. For example:
- Ionic surfactants tend to dissolve better at higher temperatures.
- Nonionic surfactants may face challenges at their "cloud point", where they phase-separate and become insoluble [1][4].
- In hard water containing calcium or magnesium ions, nonionic surfactants like fatty alcohol ethoxylates are often preferred due to their resistance to precipitation [1].
Extreme pH levels or high salinity can destabilize ionic surfactants, causing them to precipitate or lose effectiveness. Amphoteric surfactants, which change charge depending on pH, may also be impacted [1][4]. Additionally, if your process involves high shear forces – such as mixing, pumping, or application – ensure the surfactant can withstand these conditions without causing agglomeration. For instance, in adhesive formulations, an unsuitable surfactant may increase water uptake by up to 120%, compromising performance [3].
Finally, document any exposure to oxidizers, mechanical stress, or other factors that could affect the surfactant’s stability. This thorough documentation will streamline the selection process and help prevent costly reformulations later on.
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
Step 2: Choose the Right Surfactant Type and Chemistry
Once you’ve clarified your application requirements, the next step is to identify a surfactant whose chemical properties align with your process conditions. Selecting the right surfactant chemistry ensures that your formulation performs as intended.
Understand Surfactant Classifications
Surfactants are grouped based on the electrical charge of their hydrophilic (water-attracting) head. Here’s a breakdown of the main types:
- Anionic surfactants: These carry a negative charge and are known for their strong cleaning power and foaming ability. They’re commonly found in degreasers and cleaners, with examples including sulfonates, sulfates (like Sodium Lauryl Sulfate), and carboxylates [1][7][9].
- Cationic surfactants: Positively charged, these are often derived from quaternary ammonium compounds. They excel at attaching to negatively charged surfaces, making them ideal for fabric softeners, hair conditioners, and disinfectants. Their antimicrobial, antistatic, and corrosion-inhibiting properties are particularly valuable [1][7][8].
- Nonionic surfactants: These have no charge and gain water solubility through oxygen-rich groups like polyoxyethylene. They’re highly versatile, functioning as emulsifiers and wetting agents. Nonionic surfactants work well across a wide pH range and resist hard water, making them essential in agrochemical sprays, pharmaceutical solubilizers, and food emulsifiers [1][8][9].
- Amphoteric (or zwitterionic) surfactants: These contain both positive and negative charges within the same molecule. Their behavior shifts with pH – acting as cationic in acidic environments and anionic in alkaline ones. Known for their mildness and low skin irritation, they’re widely used in baby shampoos, facial cleansers, and other formulations for sensitive skin [1][7][8].
Nonionic surfactants are compatible with all other types, making them valuable as co-surfactants in blended formulations [7][8].
Select the Appropriate Chemistry Family
Once you’ve identified the surfactant type, focus on specific chemistries within each category:
- Anionic surfactants: Sulfonates are prized for their thermal and chemical stability. Sulfates, such as Sodium Lauryl Ether Sulfate (SLES), provide excellent foaming, while carboxylates offer milder cleansing and improved biodegradability [1][7].
- Cationic surfactants: Quaternary ammonium salts dominate this category, widely used in fabric softeners and disinfectants due to their antimicrobial properties and strong adhesion to fibers [1][8].
- Nonionic surfactants: Common examples include alcohol ethoxylates (effective in hard water), polysorbates (e.g., Polysorbate 80, often used as a solubilizer with an HLB of about 15), and alkyl polyglucosides (APGs), which are naturally derived and biodegradable. Alcohol ethoxylates perform well in metalworking fluids and industrial cleaners, as they resist precipitation caused by calcium and magnesium ions [1][11].
- Amphoteric surfactants: Cocamidopropyl Betaine (CAPB) is a popular choice, offering foam boosting and viscosity-building properties for personal care products. When using betaines, it’s important to monitor the formulation’s pH, as it influences the molecule’s charge and functionality [7][8].
Another critical consideration is the Critical Micelle Concentration (CMC) – the minimum concentration at which surfactant molecules form micelles. A lower CMC indicates higher efficiency, meaning less surfactant is needed to achieve the desired effect [9].
Consider Hydrophilic-Lipophilic Balance (HLB) and Other Key Properties
The Hydrophilic-Lipophilic Balance (HLB), introduced by William C. Griffin in 1949, measures a surfactant’s affinity for water versus oil on a scale from 1 to 20 [10][11].
- Surfactants with HLB values between 1 and 9 are lipophilic, making them suitable for water-in-oil emulsions like rust preventives.
- HLB values between 10 and 20 indicate hydrophilic properties, ideal for oil-in-water systems such as cleaners and lotions [10][11].
Matching a surfactant’s HLB to the "required HLB" of your oil phase is essential for achieving stability and preventing phase separation. For example, surfactants with HLB values of 13–15 are commonly used as detergents, while those in the 15–20 range function as solubilizers for fragrances or active pharmaceutical ingredients (APIs) [10][1].
In alcohol ethoxylates, the HLB value increases with the length of the ethylene oxide (EO) chain, enhancing water solubility. However, these surfactants can become less soluble at high temperatures due to chain dehydration, potentially leading to phase separation at their "cloud point" [1].
Since HLB is a theoretical guideline, always validate your formulation through lab testing. Check for signs of instability, such as creaming, coalescence, or viscosity changes [10][1].
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
Step 3: Test Performance and System Compatibility
Once you’ve selected a surfactant based on its chemistry and Hydrophilic-Lipophilic Balance (HLB), it’s time to confirm its effectiveness through thorough lab testing. This step ensures the surfactant performs well under your operating conditions and aligns with the application requirements defined earlier.
Evaluate Compatibility with Formulation Components
Start by examining how your surfactant interacts with other ingredients in your formulation. For instance, cationic surfactants, which carry a positive charge, often conflict with anionic surfactants, which are negatively charged [1]. If your formula includes polymers, builders, biocides, or preservatives, test for any adverse chemical reactions during mixing.
In hard water environments containing calcium and magnesium ions, nonionic surfactants like fatty alcohol ethoxylates often outperform ionic surfactants by resisting precipitation [1]. You can also experiment with blending surfactant types. A mix of nonionic and anionic surfactants, for example, can improve thermal stability, emulsification, and foam control through synergistic effects [1].
Conduct Lab and Stability Testing
Perform lab trials across a range of HLB values to pinpoint the best option for your needs. Start by estimating the required HLB based on the oil phase in your formulation, and test surfactants within that range.
Evaluate your formulation under all expected conditions, including manufacturing, storage, and use. Don’t forget to include extreme scenarios like freeze-thaw cycles if applicable. Look for signs of instability, such as creaming, coalescence, viscosity shifts, or color changes. For applications requiring long shelf lives, accelerated aging studies at elevated temperatures can help predict how the formulation will hold up over time.
Check Material and Equipment Compatibility
After confirming performance, assess how the surfactant interacts with your production setup. Test its compatibility with the metals, plastics, elastomers, and coatings used in your equipment to avoid issues like corrosion, swelling, or material degradation [5]. For example, cationic surfactants derived from quaternary ammonium compounds are often chosen for their ability to inhibit corrosion, making them ideal for metalworking fluids and industrial cleaning applications [1].
Additionally, ensure that foam levels and residual surfactants won’t interfere with downstream processes like coating adhesion or bonding. Following guidelines such as ASTM D6361/D6361M can help verify that your cleaning agent is safe for both the material and subsequent manufacturing steps [5].
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
sbb-itb-aa4586a
Step 4: Verify Regulatory and Safety Compliance
Ensuring that your surfactant complies with U.S. regulations and safety standards is essential for protecting workers, maintaining compliance, and addressing environmental concerns.
Review U.S. Regulatory Requirements
Start by obtaining the surfactant’s Safety Data Sheet (SDS). According to OSHA‘s Hazard Communication Standard (29 CFR 1910.1200), manufacturers and suppliers must provide SDSs in the GHS format, offering consistent hazard information [12][13].
Maintain an up-to-date chemical inventory for all surfactants used in your facility. This includes surfactants for production, cleaning, and maintenance processes [13]. For industries such as pharmaceuticals, food, or cosmetics, confirm that the surfactant complies with specific standards like USP (United States Pharmacopeia), NF (National Formulary), or FCC (Food Chemicals Codex). If the surfactant is used in products that directly impact consumers or the environment, consult the EPA’s Safer Chemical Ingredients List (SCIL) to identify options with lower toxicity and better biodegradability [2].
Once regulatory documentation is in place, shift your attention to environmental concerns and worker safety.
Assess Environmental and Sustainability Criteria
Beyond meeting regulations, evaluate the environmental impact of the surfactant. This is especially important for applications where the surfactant may bypass sewage treatment, such as outdoor use. The EPA assesses surfactants based on their aquatic toxicity, biodegradation rate, and the safety of their degradation products [2]. A surfactant is considered biodegradable if at least 60% of it mineralizes into CO₂ and water within 28 days [2][15].
The EPA Safer Choice program further categorizes surfactants on the SCIL into color-coded tiers:
- Green Circle: Verified low concern based on experimental data.
- Green Half-Circle: Expected low concern but requires additional testing.
- Yellow Triangle: Meets basic criteria but has some hazard concerns.
- Grey Square: Potentially unacceptable and may be removed unless justified within 12 months [14].
For applications involving direct environmental release – like boat cleaners or outdoor equipment – stricter standards apply. Surfactants with aquatic toxicity below 10 mg/L are prohibited due to their immediate danger to aquatic life [15]. The balance between toxicity and biodegradability is critical: surfactants with acute aquatic toxicity of 1 ppm or less are only acceptable if they fully biodegrade within 10 days [2].
| Acute Aquatic Toxicity (L/E/IC50) | Required Rate of Biodegradation |
|---|---|
| ≤1 ppm | Within a 10-day window (without concerning byproducts) |
| >1 ppm and ≤10 ppm | Within a 10-day window |
| >10 ppm | Within 28 days |
Ensure Worker Safety and Handling Procedures
After regulatory and environmental assessments, verify that your safety protocols align with the surfactant’s SDS. OSHA requires 16 standardized sections in the SDS, with Sections 1–11 and Section 16 being mandatory [16]. Pay close attention to:
- Section 7 (Handling and Storage): Details safe handling practices and chemical incompatibilities.
- Section 8 (Exposure Controls): Lists OSHA Permissible Exposure Limits (PELs), ACGIH Threshold Limit Values (TLVs), and recommended Personal Protective Equipment (PPE) [16].
Develop emergency procedures based on SDS Sections 4 (first-aid measures), 5 (fire-fighting guidance), and 6 (spill response). Section 14 includes transport-related details like UN numbers, proper shipping names, and hazard classifications for safe delivery and storage [16].
To prioritize risk reduction, use hazard databases like ChemView, RISCTOX, or PubChem [13]. These tools rank surfactants based on hazard and exposure levels, from 1 (low) to 5 (very high) [13]. Additionally, create process flow diagrams to identify areas where hazardous material exposure can be minimized [13].
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
Step 5: Confirm Quality, Documentation, and Supply Reliability
Once regulatory and performance standards are met, the next step is to focus on maintaining consistent quality and ensuring a dependable supply chain. Reliable surfactant quality and timely delivery are essential to keep production on track and meet expected performance benchmarks.
Ensure Quality and Batch-to-Batch Consistency
To maintain quality, always request a Certificate of Analysis (COA) for each batch. This document provides actual test results, such as assay/purity levels, water content (commonly measured through Karl Fischer Titration), and trace metal impurities, rather than just theoretical specifications [17]. It’s crucial to verify that the lot number on the COA matches the one on the container to ensure proper traceability [17].
Selecting the right chemical grade for your application is equally important. For instance:
- Technical grade surfactants (85% to 95% purity) are well-suited for general manufacturing and industrial uses.
- ACS Reagent grade chemicals (95% to 99.9%+ purity) are ideal for analytical purposes.
- USP (United States Pharmacopeia), NF (National Formulary), and FCC (Food Chemicals Codex) grades are required for pharmaceutical, cosmetic, and food applications.
Cost considerations play a role here as well. Technical grade chemicals are often 30% to 50% less expensive than ACS reagent grade options, making the choice of grade a practical way to manage costs [17].
"The right chemical grade can be the difference between success and failure in a process." – Andre Taki [18]
Consistency is key. Compare COAs from different batches to identify trends. A supplier that consistently delivers results within specification limits shows strong process control and reliability [17]. When reviewing documentation, prioritize COAs over Certificates of Conformance (CoC). Unlike a COA, a CoC only confirms compliance without providing specific test data [17].
Evaluate Supply Chain Reliability
A dependable supply chain is just as important as product quality. Ensure your supplier can deliver surfactants on time and in the quantities you need. Check lead times, stocking options, and packaging capabilities to avoid delays. Just-in-time delivery can also help reduce inventory costs and minimize risks like material expiration.
For critical applications, work with suppliers offering flexible batch sizes and quick delivery options. This flexibility is especially valuable during demand fluctuations or when testing new formulations. A strong supply chain ensures uninterrupted operations and smooth collaboration with your chemical provider.
Partner with a Reliable Specialty Chemical Provider
Collaborating with a trusted supplier like Allan Chemical Corporation (https://allanchems.com) can simplify sourcing high-quality surfactants. With over 40 years of experience, they specialize in providing technical-grade and compendial-grade surfactants tailored for regulated industries, including pharmaceuticals, food, cosmetics, and electronics. Their documented systems ensure consistent quality and compliance with environmental requirements.
A reliable supplier also provides technical support for documentation and regulatory questions, helping you navigate complex requirements. Before committing to large-scale purchases, request sample COAs to assess the supplier’s testing rigor and documentation standards [17]. Opt for suppliers whose test results demonstrate comfortable margins within specification limits, as opposed to those operating near the edge of compliance [17].
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
Conclusion: Complete Checklist for Industrial Surfactant Selection
Key Takeaways
Bringing together the steps outlined earlier, this checklist is designed to simplify the process of choosing industrial surfactants. A structured method ensures you address performance needs, safety regulations, and supply dependability. Begin by identifying the specific application and understanding the substrate material to avoid issues like etching or swelling [6]. Then, select the appropriate surfactant chemistry by aligning the charge type and HLB (Hydrophilic-Lipophilic Balance) values with your phase requirements. For example, water-in-oil emulsions typically need HLB values between 3 and 6, while oil-in-water systems require values between 8 and 18 [1].
Lab testing is crucial to confirm compatibility and stability, as theoretical models alone cannot guarantee performance in practical settings [1]. Ensure compliance with biodegradability and toxicity standards by reviewing the Safety Data Sheet for proper handling protocols [2][6]. Finally, choose a supplier who offers consistent product quality and reliable technical support.
Final Consolidated Checklist
Here’s a streamlined checklist to help ensure no detail is missed:
Application & Performance:
- Clearly define the intended application and establish performance goals.
- Specify targets for detergency, foaming, and wetting capabilities.
- Document operational conditions, including temperature ranges, pH levels, and cleaning methods [6].
Chemistry Selection:
- Select surfactant type based on charge (anionic, cationic, nonionic, or amphoteric) and functional requirements.
- Match HLB values to emulsion needs – lower for water-in-oil, higher for oil-in-water.
- Opt for surfactant chemistries that align with formulation objectives.
Testing & Compatibility:
- Perform small-scale tests to confirm compatibility with your system.
- Assess stability across varying temperature and pH conditions.
- Ensure resistance to hard water, focusing on nonionic options like fatty alcohol ethoxylates [1][6].
Regulatory & Safety:
- Verify that biodegradability meets EPA guidelines.
- Check for acute aquatic toxicity thresholds (≤1 ppm) and ensure a 10-day biodegradation window.
- Review the Safety Data Sheet for required protective equipment and handling instructions [2][6].
Quality & Supply:
- Request Certificates of Analysis with batch-specific test results and confirm lot traceability.
- Confirm lead times and availability of ready-stock options.
- Evaluate supplier support, including technical assistance and documentation, to ensure smooth industrial operations.
This content is for informational purposes only. Always consult official regulations and qualified professionals when making sourcing or formulation decisions.
FAQs
What should I consider when choosing the right type of surfactant for my application?
When choosing a surfactant, the molecular charge plays a crucial role. Anionic surfactants, which carry a negative charge, excel in cleaning and foaming applications. On the other hand, cationic surfactants, with their positive charge, often provide antimicrobial benefits but don’t mix well with anionic agents. Nonionic surfactants, which are neutral, perform well in hard water and are ideal for gentle formulations. Amphoteric surfactants, featuring both positive and negative charges, can adjust to different pH levels, making them suitable for mild or pH-sensitive applications.
In addition to charge, think about specific performance requirements such as detergency, emulsification, or foam generation. Also, evaluate compatibility with other ingredients, pH stability, and resistance to temperature changes. If you’re working in a regulated industry, ensure that the surfactant complies with standards like USP or FCC to streamline compliance and maintain quality. Carefully balancing these factors will help you find the most effective and economical surfactant for your application.
How can I make sure the surfactant I choose meets U.S. environmental and safety standards?
To ensure your surfactant aligns with U.S. safety and environmental standards, begin by checking if it complies with EPA Safer Choice criteria. Surfactants carrying this label are assessed for low aquatic toxicity and quick biodegradability, which helps minimize their impact on waterways. Another helpful resource is the Safer Chemical Ingredients List (SCIL), which categorizes approved surfactants based on their function and environmental safety.
For products intended for regulated industries such as pharmaceuticals, food, or cosmetics, it’s crucial to ensure the surfactant meets specific standards like USP, FCC, ACS, or NF. These benchmarks are often mandatory for compendial-grade chemicals and provide confidence in their quality and compliance.
Allan Chemical Corporation can assist in sourcing surfactants that meet these stringent standards. Their selection includes options with Safer Choice certification or SCIL approval, as well as technical-grade and compendial-grade products. They also provide the necessary documentation to help you navigate regulatory requirements effectively.
What are the best steps for testing surfactant compatibility and stability in formulations?
To assess surfactant compatibility and stability, it’s best to begin with small-scale testing before moving to larger batches. Combine the surfactant with each ingredient in your formulation – such as solvents, polymers, salts, and active ingredients – at the intended concentration. Observe the mixture over a 24-hour period at room temperature, watching for signs like separation, cloudiness, or precipitation. Additionally, monitor key properties such as pH, viscosity, and interfacial tension, as any shifts in these values may point to incompatibility.
For more detailed evaluation, accelerated stability tests can provide valuable insights. Store samples at elevated temperatures (e.g., 104°F ± 4°F) with high humidity (75% RH) for 2–4 weeks, and then recheck their physical properties. Thermal cycling, such as alternating between -4°F and 140°F, can help identify temperature-sensitive issues. Stress tests, including the addition of salt (e.g., 0.1 M NaCl) or pH adjustments, simulate harsher conditions to further evaluate stability. Advanced analytical methods like high-performance liquid chromatography (HPLC) can confirm the surfactant’s integrity and identify any potential degradation.
Allan Chemical Corporation provides technical- and compendial-grade surfactants that comply with USP, FCC, ACS, and NF standards. Their ISO-certified laboratory also offers tailored compatibility and stability testing to ensure your formulation maintains reliable performance throughout its shelf life.





Comments are closed