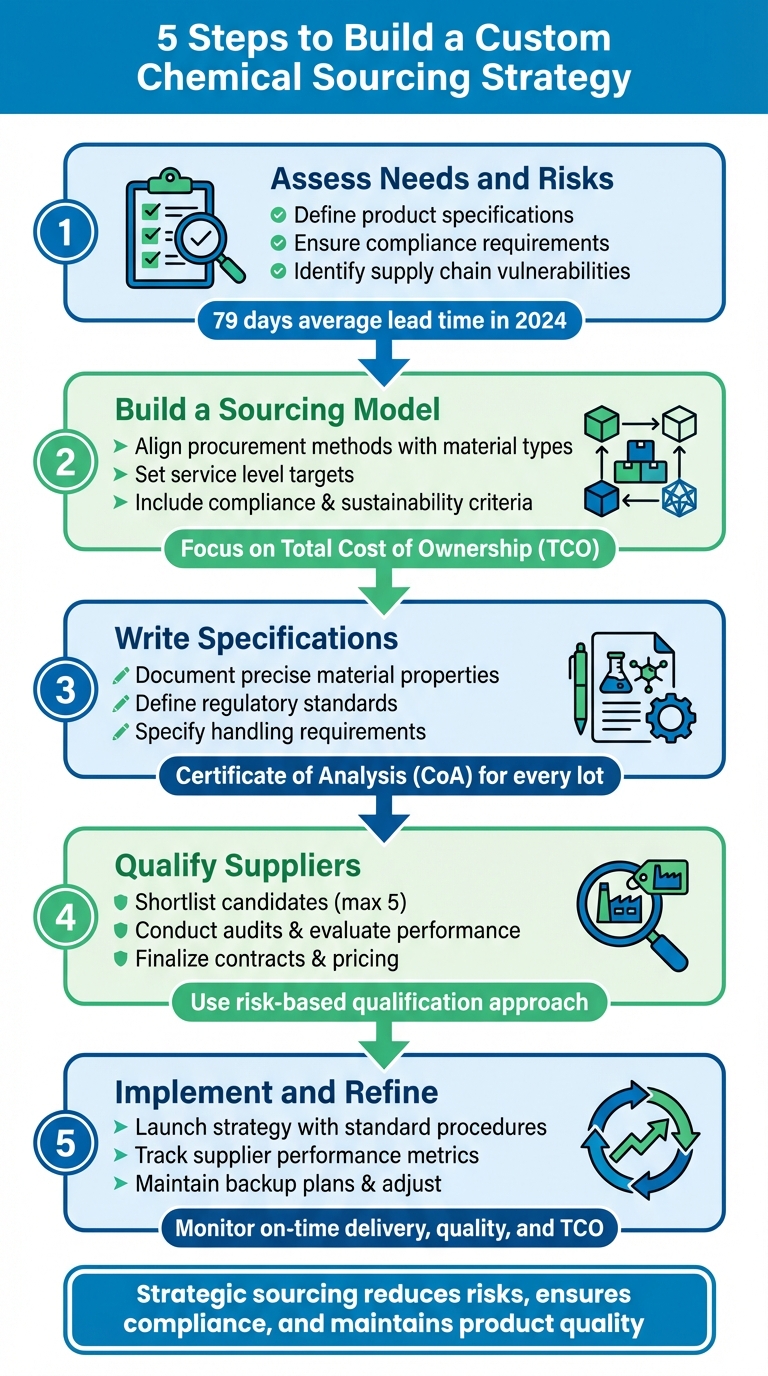
Sourcing chemicals effectively requires a clear, structured approach to navigate today’s complex supply chains. From defining your needs to qualifying suppliers, this guide outlines five steps to create a sourcing strategy tailored to your operation’s specific challenges. Here’s a quick summary:
- Step 1: Assess Needs and Risks – Define product specifications, compliance requirements, and supply chain vulnerabilities.
- Step 2: Build a Sourcing Model – Align procurement methods with material types, set service level targets, and include compliance criteria.
- Step 3: Write Specifications – Document precise material properties, regulatory standards, and handling requirements.
- Step 4: Qualify Suppliers – Shortlist, audit, and evaluate suppliers based on quality, reliability, and performance history.
- Step 5: Implement and Refine – Launch your strategy, track supplier performance, and adjust plans to mitigate disruptions.
Key insight: The average lead time in 2024 reached 79 days due to global shipping delays, costing businesses billions annually. Building flexibility and clear documentation into your strategy can reduce risks, ensure compliance, and maintain product quality.
5-Step Chemical Sourcing Strategy Framework
7 Steps for an Effective Sourcing Strategy | Procurement Best Practices
Step 1: Review Your Internal Needs and Risk Factors
Before reaching out to suppliers, it’s crucial to clearly define your operation’s needs. This involves three key steps: understanding your product specifications, ensuring compliance with regulatory requirements, and identifying vulnerabilities in your supply chain. Skipping this preparation can lead to costly mismatches between what you order and what your processes can actually handle. Let’s break this down further.
Identify Product and Process Requirements
Start by creating a detailed list of all the raw materials your operation relies on. Next, determine the chemical grade required for each application. For instance, pharmaceuticals demand USP/NF-grade chemicals, while food processors need materials that meet FCC standards. Industrial operations often use Technical Grade chemicals, but using the wrong grade can lead to regulatory issues or product failures. The table below outlines how different grades align with specific applications:
| Chemical Grade | Purity Level | Primary Use Case |
|---|---|---|
| Technical Grade | Lowest | Industrial uses where high purity is not critical |
| Lab Grade | Intermediate | Educational experiments and general testing; impurities not specified |
| BP/USP Grade | High (Pharmaceutical) | Medicine and cosmetic production; meets British/US Pharmacopoeia standards |
| Food Grade (FCC) | High (Food Safety) | Food additives, preservatives, and household applications |
| Analytical Research | Highest | Specialized lab work, including chromatography and spectroscopy |
Additionally, identify steps in your manufacturing process that could introduce impurities into the final product. In pharmaceutical operations, this means tracking every chemical transformation that could alter the impurity profile or impact critical molecular configurations. It’s also important to distinguish between commercially available chemicals and custom synthesized chemicals, as the latter require stricter oversight and adherence to ICH Q11 principles. These details will guide your supplier evaluations later on.
List Regulatory and Quality Standards
Different industries in the U.S. have varying regulatory requirements, so it’s essential to confirm which standards apply to your materials. For pharmaceuticals, sourcing must comply with FDA Chemistry, Manufacturing, and Controls (CMC) guidelines, which cover everything from drug substance identity to stability data. If you’re sourcing Active Pharmaceutical Ingredients (APIs), you’ll need to follow Q7 Good Manufacturing Practice guidelines, which address raw material handling, quarantine procedures, and lab controls [8].
Make sure to specify the required documentation upfront, such as Certificates of Analysis (CoA), Safety Data Sheets (SDS), and traceability records. As Paresma Patel, Ph.D., Division Director at the FDA, explains, "Pharmaceutical quality is assuring every dose is safe and effective, free of contamination and defects" [7]. The FDA offers a database with over 2,790 guidance documents that can help you filter information based on your product type and compliance needs [6].
Document Operational Limits and Supply Risks
Identify your operational constraints, such as batch sizes, packaging requirements, storage conditions, and lead times. Then, assess potential supply chain risks. Geopolitical risks include tariffs or political instability in supplier regions, while geographic risks involve localized disruptions like floods, port strikes, or shipping delays [3][10].
Avoid relying on a single supplier to prevent production halts in case of disruptions. Borys Schafran of Schafran Associates advises, "Review your key raw material positions, name names, and expose those vulnerabilities" [3]. Use your bill of materials to focus on your top five revenue-generating products. Map out all supplier tiers, their geographic locations, and any alternative sites they operate. Consider building strategic stockpiles of critical materials near your production facilities. Additionally, track 2–3 key metrics for each vendor, such as on-time delivery, quality performance, and responsiveness [10]. Addressing these factors lays the groundwork for a sourcing strategy tailored to your operation.
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
Step 2: Create Your Sourcing Model
After identifying your internal needs and potential risks, the next step is to craft a sourcing model tailored to your operation’s priorities. This involves determining how you’ll procure each material, setting clear expectations for supplier performance, and incorporating criteria like environmental and safety standards. A well-thought-out model strikes a balance between cost, efficiency, and compliance, while maintaining the flexibility to adapt as circumstances change. This framework lays the groundwork for evaluating suppliers effectively in later stages.
Choose Sourcing Methods by Material Type
Start by categorizing your chemicals into two main groups: raw materials (ingredients directly used in production) and MROs (maintenance, repair, and operating supplies like safety equipment or filters). Raw materials often require procurement based on production forecasts, while MROs are better managed with automatic reorder systems tied to inventory thresholds.
For bulk raw materials, consider direct contracts with manufacturers to maximize logistics efficiency, such as full truckload shipments. Specialty or research-grade chemicals – like electronic-grade solvents or custom APIs – are better sourced from providers that offer access to unique compounds and specialized services, such as reweighing or dissolving samples into specific formats. Temperature-sensitive APIs, on the other hand, demand logistics partners that adhere to Good Distribution Practices (GDP), especially for materials requiring dry ice or cold-chain handling.
For materials with short shelf lives, Just-in-Time (JIT) sourcing can help reduce waste and limit excess inventory. When sourcing critical production materials, prioritize suppliers that meet GMP or IPEC standards and provide all necessary documentation upfront. Below is a summary of recommended sourcing methods by material type:
| Material Type | Recommended Sourcing Method | Key Consideration |
|---|---|---|
| High-Volume Raw Materials | Direct Contracts with Manufacturers | Optimize logistics with full truckloads [12] |
| Specialty/Research Chemicals | Specialty Providers or Marketplaces | Access to unique compounds and reformatting [13] |
| MRO Supplies | Automatic Reorder Systems | Ensure uninterrupted daily operations [12] |
| Temperature-Sensitive APIs | GDP-Certified Logistics Partners | Maintain product integrity during transit [1] |
By aligning sourcing methods with material needs, you can enhance both efficiency and compliance throughout your supply chain.
Set Service Level Targets
Building on your earlier assessments, establish service level targets that address operational risks. Define specifics like lead times, delivery reliability, minimum order quantities (MOQs), and pricing expectations. For instance, global shipping delays in 2024 pushed average lead times to 79 days [1]. While local sourcing within the U.S. offers shorter lead times and fewer geopolitical risks, it often comes with higher labor and tax costs. On the other hand, global sourcing requires more robust forecasting.
Instead of focusing solely on unit price, assess the Total Cost of Ownership (TCO), which includes acquisition, usage, logistics, compliance, and disposal costs [1]. Lower upfront costs may hide additional expenses, such as regulatory delays or waste management. For example, implementing a digital tracking platform has been shown to reduce procurement lead times by 10 days and cut administrative work by 20% [4]. For perishable or high-turnover materials, ensure suppliers can consistently meet tight delivery windows.
"Strategic sourcing is not just about reducing costs – it’s about driving innovation, ensuring quality, and securing a competitive edge in an ever-evolving market." – James Kennemer, Cosmo Sourcing [17]
Track a few key metrics for each vendor, such as on-time delivery rates, quality performance, and technical support responsiveness. These metrics will help ensure your suppliers meet your service level expectations consistently.
Add Sustainability and Compliance Criteria
Incorporate environmental, safety, and regulatory standards into your sourcing model. For example, align material selection with Green Chemistry principles, such as using renewable feedstocks, reducing hazardous substances, and designing for easier degradation [14]. Replace harmful solvents like Toluene or Xylene with safer alternatives such as d-Limonene (from citrus) or Ethyl Acetate. However, confirm that these replacements still meet safety and flammability requirements as outlined in their Safety Data Sheets (SDS) [14].
Ensure suppliers comply with OSHA’s Hazard Communication Standard, which mandates proper labeling, chemical classification, and SDS availability [9]. Noncompliance can result in significant fines, as demonstrated by OSHA’s $50 million in penalties for safety violations in 2018 [9]. For pharmaceutical-grade materials, verify adherence to cGMP regulations (21 CFR Parts 210 and 211) and ICH M7 guidelines for mutagenic impurities [15][11]. Analytical-grade chemicals should meet purity thresholds of over 98%, so specify these standards in your sourcing criteria [16].
To further reduce supply chain risks, enforce lot traceability and change control protocols. Require suppliers to provide comprehensive documentation, including Certificates of Analysis (COA) and SDS, for every lot [16]. For custom-synthesized chemicals, request justification for the chosen "starting material" to ensure it aligns with FDA/ICH Q11 Guidance on impurity profiles [11]. By embedding these criteria into your sourcing model, you not only manage risk but also support operational efficiency and long-term sustainability.
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
Step 3: Write Clear Technical and Regulatory Specifications
Once your sourcing model is established, the next step is to convert your internal requirements into well-documented, precise specifications. These specifications act as a bridge, ensuring suppliers clearly understand your needs and helping to avoid miscommunication, quality issues, or compliance lapses. They should address three core areas: the material’s properties, the regulatory standards it must meet, and the proper handling procedures from storage to transportation.
Create Detailed Material Specifications
Begin with the basics: chemical identifiers such as CAS numbers, INCI names (for cosmetics), and exact formulations. Include acceptable ranges for critical properties like pH levels, as minor deviations can disrupt your manufacturing processes [18]. Clearly state purity requirements, for example, “≥98% for analytical-grade solvents,” and specify any impurities or trace metal limits that could affect your product quality. Require a Certificate of Analysis (COA) for each lot to confirm quality and a Certificate of Conformity to ensure safety [18][5].
Packaging and delivery formats are equally important. Specify whether materials will arrive in bulk shipments, bags, or totes, and ensure your facilities can handle these configurations [19]. For temperature-sensitive materials, outline storage conditions like “15–25°C” and include shelf-life limits to prevent degradation [18][19]. These detailed specifications not only guide suppliers but also reinforce consistency from planning to delivery.
Define Industry-Specific Compliance Requirements
Tailor your documentation and testing standards to meet the needs of your industry. For pharmaceuticals, reference current Good Manufacturing Practices (cGMP) under 21 CFR Parts 210 and 211, and require materials to comply with USP or NF monographs [21][24]. If sourcing for food applications, ensure compliance with the Food Safety Modernization Act (FSMA) and request migration testing results for food contact materials, as well as heavy metal analysis [20][21].
For electronics, include restrictions under RoHS for substances like lead, mercury, and cadmium [20][22]. Use the 16-section Safety Data Sheet (SDS) format, aligned with the Globally Harmonized System (GHS), to provide clear information on chemical hazards, composition, and handling [20][23]. For materials governed by the Toxic Substances Control Act (TSCA), require Pre-manufacture Notifications (PMN) for new substances and Chemical Data Reporting (CDR) for production volumes exceeding 25,000 lbs at a single site [23]. California-based operations should also include Proposition 65 warning labels for any of the over 900 chemicals linked to cancer or reproductive harm [20].
"Understanding how new obligations apply to your business and what action to take as a result is crucial." – Steven Lewis, UK Materials, Technology & Innovation Business Developer, RINA [22]
Thorough compliance documentation ensures suppliers meet your regulatory needs without exception.
Specify Logistics and Handling Needs
To maintain material integrity and safety, clearly define packaging, storage, and transport requirements. Specify container types such as IBCs, drums, or ISO tanks, and materials like HDPE or stainless steel, along with seal specifications [26]. For hazardous chemicals, require labeling that complies with DOT dangerous goods regulations and OSHA’s Hazard Communication Standard, including GHS signal words and pictograms [23][25].
Identify incompatible substances that must not be stored or transported near your materials to avoid dangerous reactions. For temperature-sensitive or shock-sensitive items, mandate the use of data loggers or GPS tracking with temperature sensors to maintain a verifiable chain of custody [26][27]. Environmental controls like temperature ranges, humidity limits (e.g., “<50% RH”), and ventilation requirements should also be included to prevent degradation during transit or storage. Additionally, specify Personal Protective Equipment (PPE), spill cleanup protocols, and any specialized equipment needed for unloading [25][26].
"When it comes to shipping hazardous chemicals, off-the-shelf logistics solutions just won’t do." – Mike Challman, VP of Managed Services, CLX Logistics [28]
Document these requirements in Standard Operating Procedures (SOPs) and conduct regular audits to ensure suppliers consistently meet your expectations [23][24]. This level of precision helps prevent costly errors, speeds up supplier onboarding, and safeguards your operations from compliance or quality setbacks.
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
sbb-itb-aa4586a
Step 4: Find and Qualify Suppliers
Selecting the right suppliers is crucial for ensuring quality and compliance in your operations. Start by identifying potential candidates, thoroughly evaluating their capabilities, and securing clear agreements to establish a reliable partnership.
Create a Supplier Shortlist
Begin by narrowing your search to a maximum of five suppliers who have relevant industry experience and hold essential certifications, such as cGMP or FSMA compliance. Assess their manufacturing capabilities, production tracking systems, and the availability of technical support. For example, Allan Chemical Corporation offers over 40 years of experience in regulated industries, providing just-in-time delivery and technical support across sectors like pharmaceuticals, food, and electronics.
Collaborate with experts from manufacturing, quality assurance, and logistics to develop a clear set of requirements. Request detailed documentation from each supplier to verify their compliance and capabilities. Using a numeric scoring system, evaluate candidates based on factors like manufacturing capacity, regulatory adherence, and delivery timelines.
Run a Qualification Process
Adopt a risk-based approach to supplier evaluation. Classify suppliers into two categories: quality-critical (e.g., providers of Active Pharmaceutical Ingredients) and non-critical. Quality-critical suppliers should undergo on-site audits to examine workflows, equipment maintenance, hygiene standards, and material traceability. Non-critical suppliers may be assessed through off-site reviews [29][31].
During audits, verify key records such as business licenses, regulatory approvals (e.g., FDA or EMA), and compliance with 21 CFR Parts 210 and 211 for cGMP operations [29][15]. Test product samples alongside their Certificates of Analysis (CoA) to ensure they meet your specifications before proceeding with any agreements [30][31]. Use clear pass/fail criteria for these audits and document all requirements and evaluations in your internal change-control system [31].
"Qualifying a supplier… should be thought of as a kind of quality system within a quality system." – The FDA Group [30]
Suppliers should be reassessed periodically based on their criticality and performance history. Those with recurring quality issues may require closer scrutiny through ongoing performance reviews and re-audits [29][30].
Once you’ve qualified your suppliers, the next step is finalizing contracts and pricing.
Finalize Contracts and Pricing
Use objective data, such as market benchmarks and historical spending, to negotiate Total Cost of Ownership (TCO), which includes base price, shipping, storage, and handling costs. Ensure all pricing is clearly defined in USD terms [35][5]. A helpful negotiation strategy is the 70/30 rule: spend 70% of the time listening and 30% talking to better understand the supplier’s position [32].
Define your negotiation goals, including your Most Desired Outcome (MDO), Least Acceptable Agreement (LAA), and Best Alternative to a Negotiated Agreement (BATNA) [34]. Multiyear contracts can lock in pricing and secure supply. For instance, one company negotiated a 10% discount and capped annual price fluctuations by committing to a multiyear agreement for titanium dioxide feedstock [33].
"Negotiation isn’t just a checkpoint in the sourcing process – it’s a strategic capability. When done right, it becomes a lever for long-term value creation." – Philip Ideson, Procurement Consultant [32]
Draft legally binding Quality Agreements to outline quality expectations, regulatory responsibilities, Corrective and Preventive Action (CAPA) procedures, and audit schedules [29]. To avoid delays, introduce standard contract terms during the RFx process rather than waiting until price negotiations are complete [32].
This information is intended for general guidance. Always consult official regulations and qualified professionals when making sourcing or formulation decisions.
Step 5: Launch, Track, and Refine Your Strategy
Once your supplier contracts are finalized, it’s time to put your sourcing strategy into action. This step brings together supplier onboarding, performance tracking, and contingency planning to ensure your approach delivers consistent results. By building on earlier evaluations, this phase focuses on implementing standardized processes, monitoring supplier performance, and staying flexible in response to market changes.
Roll Out Standard Procedures
Start by formalizing your vendor onboarding process. Gather all necessary documentation, such as business licenses, tax forms, insurance details, and payment information, to properly register suppliers [36]. Use vendor management tools integrated with ERP systems to centralize supplier data and streamline operations [36].
Set clear protocols for handling chemical orders. Ensure deliveries come with accurate labeling, compound IDs, and supplier numbers. Request machine-readable files to integrate seamlessly into your systems, reducing manual errors [13]. For regulated chemicals, establish Standard Operating Procedures (SOPs) for tasks like lot sequestration, change control, batch traceability, and pedigree verification to meet cGMP compliance standards [38]. Additionally, create specific handling guidelines for hazardous or temperature-sensitive materials – like those requiring dry ice – to maintain product integrity during transport and storage [13].
Make Certificates of Analysis (CoAs) easily accessible [37]. For example, Spectrum Chemical’s quality team conducts over 15,000 lab tests annually to ensure chemicals meet strict safety, purity, and efficacy standards [38]. Use electronic systems, such as EDI (Electronic Data Interchange) or web-based portals, to speed up ordering processes and minimize errors [38].
With these procedures in place, the next focus is on evaluating supplier performance.
Monitor Performance Metrics
Evaluate key metrics like on-time delivery, order accuracy, and quality issues to gauge supplier reliability. Financial metrics are equally important – track total cost savings, landed costs (including freight, insurance, duties, and taxes), and pricing changes [39][12]. Pay attention to working capital ratios like Days Payable Outstanding (DPO) and Days Inventory Outstanding (DIO) to measure financial efficiency [36].
"While traditional operational metrics such as on-time delivery, quality and cost savings are fundamental, you must also consider suppliers’ innovation capabilities, their ability to support new product introductions, the quality of the relationship and the responsiveness of the supplier’s service team." – Gartner [39]
Automated supplier scorecards can help standardize performance evaluations. Conduct regular reviews against established KPIs to maintain accountability [36][39]. For raw materials with short shelf lives, monitor just-in-time inventory metrics to avoid overstocking or running out [12]. Go beyond surface-level financial checks to ensure suppliers remain financially stable and avoid unexpected risks [39].
After analyzing performance data, it’s essential to prepare for potential disruptions.
Maintain Backup Plans and Make Improvements
Strengthen your supply chain by working with suppliers that have multiple manufacturing sites and shipping methods [19]. Prequalify alternate sources through detailed risk assessments to understand the potential impact of disruptions [19][1]. Keep local safety stocks near the point of use to minimize the risk of production delays [3].
"Price competitiveness cannot be the sole driver for evaluating supply options; companies need to diversify the way they buy and consume chemicals." – GEP [19]
Refine your strategy by regularly reviewing Total Cost of Ownership (TCO), which includes costs like shipping, testing, rework, waste, and delays [1]. Update supply and demand forecasts frequently and shift inventory between plant locations to manage regional fluctuations [19]. Leverage B2B digital platforms to run parallel RFQs, quickly qualify alternate suppliers, and consolidate documentation for improved traceability [1][2]. The chemical sourcing platform market is expected to grow significantly, reaching $3.08 billion by 2032, with a CAGR of 12.5% [1].
Move beyond transactional relationships by building collaborative partnerships with key suppliers. This approach can secure priority production schedules and better support during market changes [19][17]. Work with distributors that have dedicated market intelligence teams to develop custom solutions and create redundant sourcing options [3]. Incorporate AI and machine learning to forecast demand and identify potential shortages before they arise [1].
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Conclusion
This guide has outlined essential steps to turn complex chemical sourcing challenges into a streamlined and effective strategy. Crafting a tailored sourcing approach requires balancing technical, regulatory, and operational considerations. By following five key steps – assessing internal needs, creating a sourcing model, defining precise specifications, qualifying suppliers, and continuously improving the process – you can build a sourcing strategy that stays compliant, minimizes risks, and safeguards product quality throughout the supply chain.
The world of chemical sourcing is changing quickly. Predictive tools like AI, sustainability benchmarks, and digital B2B platforms are replacing outdated manual processes and traditional brokers. Companies that prioritize supplier qualification, integrate documentation requirements early on, and maintain dual sourcing options are far better equipped to handle market fluctuations and meet regulatory demands [1].
Specialized partners play a vital role in making these strategies work. Providers with strong market knowledge, multiple sourcing options, and local inventory can help procurement teams reduce risks without overextending resources [3]. For industries like pharmaceuticals, food, and cosmetics, partnering with experienced suppliers ensures access to high-quality, well-documented materials. For example, Allan Chemical Corporation, with over 40 years of experience, demonstrates this approach through just-in-time delivery, competitive pricing, and reliable supplier relationships tailored to meet custom sourcing requirements.
Investing in a well-thought-out sourcing strategy delivers measurable benefits – fewer delays, lower overall costs, and consistent product quality. By adopting these practices, you can build a supply chain that is both flexible and compliant.
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
FAQs
What are the main risks to watch for when sourcing chemicals internationally?
Sourcing chemicals from international suppliers comes with a host of challenges that can directly affect quality, costs, and delivery schedules. One major hurdle is regulatory and compliance issues. Each country has its own set of safety, import-export, and environmental regulations, which can shift unexpectedly, causing delays or adding unforeseen expenses. On top of that, supply chain disruptions – like raw material shortages, customs bottlenecks, or transportation hiccups – can lead to inventory shortfalls and production downtime.
Another key concern is price instability and currency fluctuations, which can drive up costs, especially when dealing with suppliers in regions with unpredictable exchange rates. Then there’s the issue of quality control. Without proper vetting, suppliers may provide subpar or mislabeled products, jeopardizing production processes. Finally, geopolitical risks, such as trade sanctions or restrictions, can suddenly block shipments or cut off access to critical materials, forcing companies to scramble for costly alternatives.
Allan Chemical Corporation addresses these risks head-on. By maintaining a strong supplier network and rigorous supplier verification processes, they ensure consistent quality and compliance. Their ISO-certified quality systems and just-in-time delivery model further enhance reliability. Plus, with a diverse portfolio of technical- and compendial-grade products and transparent pricing in U.S. dollars, AllanChem helps procurement teams navigate global sourcing challenges with confidence and ease.
How can I make sure the chemicals I source meet regulatory standards?
To meet industry regulations, start by pinpointing the specific requirements for each chemical. These might include grade, purity, packaging, and adherence to standards such as USP, FCC, or FDA cGMP. Confirm that your suppliers can provide essential documentation, including Certificates of Analysis (COAs), batch records, and safety data sheets, to ensure compliance.
When selecting suppliers, it’s crucial to evaluate their quality systems, inspect their manufacturing and storage practices, and verify that they maintain proper documentation for every shipment. Keeping comprehensive records, such as test results and supplier audit reports, will ensure you’re always prepared for regulatory inspections.
For a more efficient approach, you might consider working with a trusted specialty chemical provider. With decades of experience, companies like Allan Chemical Corporation offer reliable delivery schedules, detailed documentation, and expertise to help you stay compliant while concentrating on your core operations.
How can I reduce supply chain disruptions when sourcing chemicals?
Reducing supply chain disruptions in chemical sourcing calls for a well-thought-out and flexible strategy. Begin by conducting regular assessments to identify potential risks with your suppliers. Pair this with contingency plans that allow you to pivot quickly to alternative sources when necessary. Expanding your supplier network, both geographically and across various vendors, can help minimize reliance on a single source, reducing the fallout from regional or vendor-specific disruptions.
Keeping inventory levels balanced is another critical step. Using demand forecasting and inventory management tools ensures you’re ready for unexpected delays without tying up resources in excessive stock. At the same time, maintaining strict quality controls and compliance checks is vital to meet regulatory requirements, even when sourcing alternatives on short notice.
Allan Chemical Corporation brings over 40 years of expertise to help navigate these challenges. They offer just-in-time delivery, an extensive inventory of technical- and compendial-grade chemicals, and specialized knowledge across regulated industries such as pharmaceuticals, food, and cosmetics. Their strong supplier relationships and certified processes provide a dependable and compliant supply chain to keep your operations running seamlessly.





Comments are closed