Protein-based drugs are highly effective but prone to instability, which can reduce efficacy and trigger immune reactions. Stabilizing excipients are essential to protect proteins during manufacturing, storage, and transport. These excipients prevent degradation by reinforcing hydration shells, maintaining pH, and limiting aggregation or chemical changes. Key stabilizers include:
- Sucrose (CAS No. 57-50-1): A disaccharide that strengthens hydration shells and forms a protective glassy matrix during freeze-drying. Widely used in monoclonal antibody formulations.
- Trehalose (CAS No. 99-20-7): A non-reducing sugar with a high glass transition temperature, offering superior stability in lyophilized formulations.
- Polysorbate 20 (PS20, CAS No. 9005-64-5): A surfactant that prevents protein unfolding and aggregation, especially under mechanical stress.
- Polysorbate 80 (PS80, CAS No. 9005-65-6): Similar to PS20 but tailored for higher protein concentrations and stress conditions.
- Arginine (CAS No. 74-79-3): Reduces viscosity and aggregation in high-concentration protein solutions, especially for subcutaneous injections.
- Histidine (CAS No. 71-00-1): A buffering agent that regulates pH and prevents aggregation in both liquid and lyophilized formulations.
- Kolliphor HS 15 (CAS No. 70142-34-6): A robust surfactant alternative to polysorbates, offering better resistance to enzymatic degradation.
- Kolliphor EL (CAS No. 9004-35-7): Stabilizes proteins by reducing aggregation under oxidative or mechanical stress.
- Phenylalanine (CAS No. 63-91-2): An amino acid that prevents aggregation and strengthens hydration shells, often paired with sugars in lyophilized products.
These excipients are crucial for extending the shelf life of protein drugs and ensuring patient safety. High-purity grades minimize impurities that could accelerate degradation or aggregation. Always consult experts and regulations when selecting excipients for formulations.
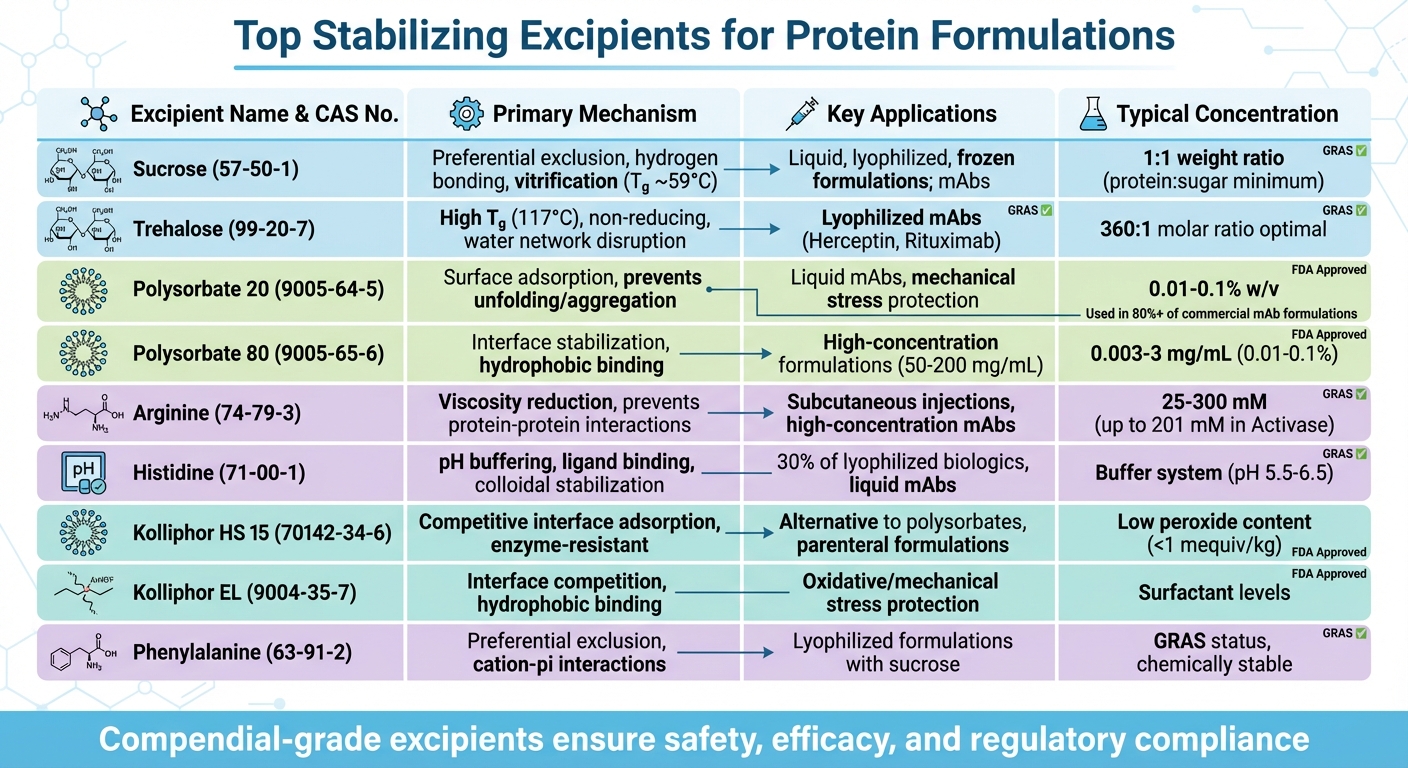
Top 9 Stabilizing Excipients for Protein Formulations: Mechanisms and Applications
Mechanisms of aggregation and stabilisation
1. Sucrose
Sucrose (CAS No. 57-50-1) is a non-reducing disaccharide and one of the most commonly used stabilizing agents in protein formulations. It helps protect therapeutic proteins in both liquid and solid states by strengthening the protein’s hydration shell and favoring the natively folded state of the protein [2].
Stabilization Mechanism
In liquid formulations, sucrose enhances protein stability through preferential exclusion, where it is excluded from the protein surface, thereby strengthening the hydration shell. During lyophilization, sucrose replaces water by forming protective hydrogen bonds. It also creates a rigid, glass-like matrix through vitrification, which limits molecular mobility. This matrix has a glass transition temperature (Tg) of about 140°F (59°C), requiring storage at least 85°F (30°C) below Tg to maintain stability [2] [8]. These mechanisms make sucrose a reliable stabilizer across different formulation types.
Applications in Protein Formulations
Sucrose is used in liquid, lyophilized, and frozen protein formulations. Its ability to remain amorphous during freezing and thawing is particularly useful in frozen drug substances, where other sugars may crystallize and compromise stability. For example, Bristol-Myers Squibb replaced maltose with sucrose in its Abatacept formulation due to sucrose’s better solubility and amorphous properties. A sugar-to-protein weight ratio of at least 1:1 is common, though studies on rhuMAb HER2 suggest a molar ratio of approximately 360:1 is required for optimal storage stability [8].
Compatibility with Proteins and Other Excipients
Sucrose is compatible with many biologics, including monoclonal antibodies (IgG1, IgG2), fusion proteins, enzymes, and cytokines. It is often combined with other excipients for enhanced performance. For instance, when paired with mannitol, sucrose acts as an amorphous co-stabilizer, preventing crystallization and maintaining protein structure. Many antibody formulations use a combination of a histidine buffer, a disaccharide stabilizer like sucrose or trehalose, and a surfactant such as PS20 or PS80 for optimal stability. Additionally, using high-purity "Low NPI" sucrose reduces the risk of impurity-induced protein aggregation and analytical inaccuracies [2] [8].
Performance in Stability Studies
Research on IgG1 fusion proteins shows that increasing sucrose content improves stability. However, sucrose does not prevent all chemical instabilities, such as aspartic acid isomerization [7] [8]. While sucrose is non-reducing, glycation has been observed in a recombinant monoclonal IgG2 antibody stored at 99°F (37°C) for one month, though no glycation occurred at 39°F (4°C) over 18 months [8]. Emerging studies suggest that at temperatures well below Tg, protein stability is more closely linked to reduced free volume and fast local dynamics rather than global molecular mobility.
This content is for informational purposes only. Always consult official guidelines and qualified professionals before making sourcing or formulation decisions.
2. Trehalose
Trehalose (CAS No. 99-20-7) is a non-reducing disaccharide commonly used as a stabilizer in biopharmaceutical formulations. Its molecular structure, featuring an α-1,1-glycosidic bond connecting two glucose units, provides it with exceptional stability. Recognized as "Generally Recognized as Safe" (GRAS) by the US FDA in 2000 [9], trehalose offers versatile stabilization mechanisms that make it a valuable ingredient in various formulation settings.
Stabilization Mechanism
Trehalose works through multiple stabilization pathways. In liquid formulations, it strengthens the hydration shell around biomolecules through preferential exclusion. During lyophilization (freeze-drying), it replaces water molecules by forming hydrogen bonds and vitrifies into a glassy matrix with a glass transition temperature (Tg) of about 243°F (117°C). This matrix immobilizes proteins, maintaining their structure [9][2]. Moreover, trehalose disrupts water’s tetrahedral network, reducing free water and improving cryoprotection. Its non-reducing nature prevents it from participating in the Maillard reaction with protein amino groups, avoiding common chemical degradation [9].
Applications in Protein Formulations
Trehalose’s stabilizing properties make it a key ingredient in a wide range of protein formulations. For example, it is used in the lyophilized formulation of Trastuzumab (Herceptin) to leverage its high Tg. Similarly, subcutaneous Rituximab formulations combine α-trehalose dihydrate with L-histidine, L-methionine, and polysorbate 80 to enhance performance [8]. Experimental studies have also shown that trehalose at concentrations of 70 mg/mL can enable immunoglobulin G (IgG) dispersions at 220 mg/mL with a manageable syringeable viscosity of just 36 cP [4].
Compatibility with Proteins and Other Excipients
Trehalose pairs well with histidine buffers (pH 5.5–6.5) and surfactants like polysorbate 20 or 80, creating effective stabilization systems [8][2]. In lyophilized formulations, it is often combined with crystalline bulking agents such as mannitol or sorbitol to improve the appearance of the cake and enhance drying efficiency [8][2]. However, unlike sucrose, trehalose can crystallize during freeze-thaw cycles. This requires careful process control to preserve its protective amorphous state [8].
Performance in Stability Studies
Trehalose’s higher Tg gives it an edge over sucrose in stability studies, particularly at elevated temperatures. For instance, research on chimeric therapeutic antibodies at 140°F (60°C) demonstrates trehalose’s superior performance [8]. Studies suggest that a molar ratio of approximately 360:1 (trehalose to protein) is optimal for maintaining storage stability in lyophilized antibody formulations [8]. Additionally, trehalose can endure temperatures up to 248°F (120°C) for 90 minutes with over 99% recovery, provided relative humidity stays below 90% [9]. Its α-1,1-glycosidic bond is also more acid-resistant than sucrose’s α,β-1,2 bond, contributing to long-term stability [9].
This information is intended for educational purposes only. Always consult official guidelines and qualified experts before making sourcing or formulation decisions.
3. Polysorbate 20 (PS20)
Polysorbate 20 (CAS No. 9005-64-5) is a nonionic surfactant commonly used in monoclonal antibody formulations, with lauric acid making up 40–60% of its fatty acid content [2][10].
Stabilization Mechanism
PS20 plays a key role in stabilizing proteins by adsorbing to hydrophobic surfaces and binding to exposed hydrophobic regions on proteins. This action helps prevent protein unfolding and reduces the risk of aggregation [2]. Additionally, it provides protection during freeze-thaw processes and limits protein loss caused by adsorption to packaging materials like glass vials and prefilled syringes.
Applications in Protein Formulations
PS20 is typically used at concentrations of 0.01%–0.1% across various formulation types. In liquid formulations, it protects proteins from mechanical stress during processes like pumping, agitation during transport, and sterile filtration. For high-concentration formulations (50–200 mg/mL), it helps manage challenges such as increased viscosity, opalescence, and protein particle formation [2][3]. In lyophilized products, PS20 stabilizes proteins during freezing and drying, preventing denaturation and particle formation.
A study conducted by the FDA’s Center for Drug Evaluation and Research in May 2024 examined PS20 stability in two commercial products: Bevacizumab at pH 6.2 with 0.04% w/v PS20 and T-DM1 (Trastuzumab emtansine) at pH 5.0 with 0.02% w/v PS20. Even under enzymatic stress (1 U/mL esterase), the study found no significant impact on the biological activity or monomer purity of the therapeutic proteins after 4 weeks at 98.6°F (37°C) [11].
Compatibility with Proteins and Other Excipients
PS20 is highly compatible with monoclonal antibodies and is often combined with buffers like histidine, stabilizers such as sucrose and trehalose, and amino acids like arginine [2]. However, it may destabilize certain protein complexes, such as virus-like particles (VLPs) [1]. In combination with cyclodextrins, such as hydroxypropyl–β–cyclodextrin (HPβCD), PS20 is better protected from enzymatic degradation [12].
The surfactant primarily degrades through auto-oxidation and ester hydrolysis. Degradation products, including free fatty acids, can form visible or sub-visible particles, which might be misinterpreted as protein aggregates [2][12]. These characteristics make PS20 a reliable option for many formulations, as demonstrated in stability studies.
Performance in Stability Studies
Even trace amounts of residual host cell proteins (HCPs), as low as 1 ppm, can degrade PS20 and produce free fatty acid particles during storage [11]. Its degradation is influenced by pH, with higher pH levels (e.g., pH 7.4) leading to increased fatty acid release compared to lower pH levels (e.g., pH 5.6) [11]. To enhance stability, using high-purity, compendial-grade PS20 is recommended to minimize initial peroxide levels and residual metals that could accelerate oxidation [1][2]. Additionally, storing PS20-containing formulations in environments that limit light and oxygen exposure reduces the risk of auto-oxidation. Advanced analytical techniques like RP-HPLC or fluorescence micelle assays are effective for monitoring PS20 levels throughout a product’s shelf life [3].
Allan Chemical Corporation offers compendial-grade PS20 that adheres to stringent quality standards.
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
4. Polysorbate 80 (PS80)
When it comes to surfactants, Polysorbate 80 (PS80) provides essential stabilization support. Known chemically as polyoxyethylene sorbitan monooleate (CAS No. 9005-65-6), this surfactant is a cornerstone in protein therapeutics, appearing in over 80% of commercial monoclonal antibody formulations [15]. To comply with European Pharmacopoeia standards, at least 58% of its total fatty acid content must come from the monooleate fraction [3][10].
Stabilization Mechanism
PS80 stabilizes proteins by working at interfaces to prevent unfolding and binding to hydrophobic areas of proteins, acting almost like a molecular chaperone. This helps reduce aggregation caused by surface-induced or mechanical stress [2][3].
It also shields proteins from mechanical stress during manufacturing and handling. By minimizing aggregation caused by agitation – such as shaking during transport or shear stress during pumping – PS80 helps preserve protein structure. Additionally, it protects formulations during freeze–thaw cycles and lyophilization by reducing surface-induced denaturation. This dual role is key to extending the shelf life of protein-based drugs.
Applications in Protein Formulations
PS80 is typically used in concentrations ranging from 0.003 to 3 mg/mL (approximately 0.01% to 0.1%), tailored to the specific formulation and stress conditions [3][2].
- Liquid formulations: It prevents aggregation during processes like pumping, filtration, and filling. In high-concentration formulations (50–200 mg/mL), it plays a critical role in maintaining protein solubility and preventing both reversible and irreversible aggregation [3].
- Lyophilized products: PS80 protects against freezing and dehydration stresses, reducing subvisible particle formation upon reconstitution. It also minimizes protein loss by limiting adsorption to hydrophobic surfaces such as glass vials, tubing, and sterilization filters.
Compatibility with Proteins and Other Excipients
The performance of PS80 is influenced by the formulation environment, particularly the buffer system. For instance, PS80 degrades rapidly in histidine buffers, likely due to pro-oxidant complexes formed with metal ions [14]. In contrast, citrate, phosphate, or tris buffers can stabilize PS80 by chelating metal ions and suppressing oxidation [14]. Opting for these buffer systems can greatly enhance the surfactant’s long-term stability, especially in the presence of trace metals.
Trace metals like iron and copper can catalyze PS80 oxidation, with even 20 ppb levels of iron posing a risk [15]. Adding chelating agents, such as EDTA or citric acid, can help sequester these metals and reduce oxidation [1][14]. Additionally, residual host cell proteins (HCPs), like lipases and esterases, can hydrolyze PS80, releasing free fatty acids that may lead to particle formation [3].
"Common surfactants such as polysorbates are well known to contain trace amounts of peroxides that can become significant during storage and consequently compromise protein stability through oxidation of susceptible amino acids."
– Jay Kang, Director of Analytical and Formulation Development, Patheon [1]
These precautions are essential for maximizing PS80’s effectiveness in diverse formulations.
Performance in Stability Studies
PS80 can degrade through hydrolysis of its ester bond or auto-oxidation of its polyoxyethylene segments [3][10]. Hydrolysis may release free fatty acids that precipitate and potentially trigger protein aggregation [3][2]. Degradation products like hydroperoxides and free radicals can oxidize sensitive protein residues, such as methionine and tryptophan, leading to structural changes and loss of bioactivity [3][1].
Using high-purity grades of PS80 can significantly reduce peroxides and residual metals, improving protein stability [2][1]. To further protect PS80, it should be stored in tightly sealed containers and shielded from light to prevent auto-oxidation and hydroperoxide formation [13][3]. The industry increasingly favors super-refined, high-purity PS80 to minimize impurities that could compromise protein stability or lead to visible particle formation.
Allan Chemical Corporation provides compendial-grade PS80 that meets the stringent quality requirements of biopharmaceutical applications.
This information is intended for educational purposes only. Always consult official guidelines and qualified experts before making formulation or sourcing decisions.
5. Arginine
Arginine plays a key role in high-concentration protein formulations by reducing viscosity and preventing protein aggregation. L-Arginine (CAS No. 74-79-3) has become a preferred excipient for formulations, especially those designed for subcutaneous injections. As of 2023, arginine is included in 20 approved protein injectable products, 17 of which are monoclonal antibody formulations[18]. Its ability to address two major challenges – lowering viscosity and preventing aggregation – makes it particularly effective in formulations with protein concentrations of 100 mg/mL or higher.
Stabilization Mechanism
Arginine stabilizes proteins through a dual mechanism involving its peptide backbone and guanidinium side chain[16]. At concentrations below 100 mM, it interacts with charged regions like glycine, while at higher concentrations (above 100 mM), its guanidinium group binds to exposed hydrophobic patches, reducing protein-protein interactions[16][18].
"Arginine’s effect on the protein unfolding behaves like the sum of its constituent parts, glycine and the guanidinium ion."
– Robert J. Falconer, University of Sheffield[16]
In freeze-dried formulations, maintaining arginine in an amorphous state is critical to ensure effective cryoprotection[17].
Applications in Protein Formulations
In practical use, arginine is typically added at concentrations ranging from 25 to 300 mM, where it effectively reduces viscosity and mitigates aggregation[18]. Its inclusion in high-concentration antibody formulations, which make up 65% of arginine-containing products, reflects the growing preference for subcutaneous delivery due to its convenience for patients.
Examples of notable formulations include Hemlibra (150 mg/mL, 150 mM), Enspryng (120 mg/mL, 150 mM), and Xolair (150 mg/mL, 200 mM), all of which demonstrate arginine’s dual role in stabilizing proteins and adjusting tonicity[18]. For intravenous formulations, Activase (alteplase) contains the highest dose of arginine among approved products – 3,500 mg per 100 mg vial (approximately 201 mM) – to maintain protein solubility after reconstitution[18].
In one study, the addition of 200 mM arginine HCl to a 250 mg/mL bovine gamma globulin solution reduced viscosity from 59.6 cP to 40.0 cP, showcasing its effectiveness in high-concentration protein solutions[18].
Compatibility with Proteins and Other Excipients
Arginine is essential in extending the shelf life and maintaining the efficacy of protein formulations. It pairs well with sugars like sucrose and trehalose in stabilization strategies. While sugars prevent initial protein unfolding through preferential exclusion, arginine suppresses aggregation of partially unfolded proteins[18].
"The use of Arg as a viscosity reducer and protein stabilizer in high concentration formulations will be the inevitable future trend of the biopharmaceutical industry for subcutaneous administration."
– Steven Ren, CMC Management, WuXi Biologics[18]
The type of arginine salt used can also impact its performance. While arginine hydrochloride (Arg.HCl) is the most common form, arginine glutamate (Arg.Glu) is often more effective at reducing protein-protein interactions on a per-mole basis[18]. Arg.Glu is particularly advantageous in frozen formulations, as mannitol can induce crystallization of Arg.HCl, compromising its stabilizing effect. Counterions like glutamate, acetate, or aspartate help prevent this issue[17][18].
Although arginine may lower a protein’s thermal unfolding temperature (Tₘ) by about 3.3°C due to the mild chaotropic nature of its guanidinium group, this minor reduction in thermal stability is generally outweighed by its ability to prevent aggregation during storage and handling.
Performance in Stability Studies
Arginine performs well under temperature stress, preventing both soluble and insoluble aggregate formation[18]. It also reduces opalescence in high-concentration IgG solutions by mitigating attractive electrostatic interactions, ensuring clarity during long-term storage[18].
In freeze-dried formulations, arginine HCl poses challenges due to its low glass transition temperature (Tg′) of approximately –44 to –42°F (–42 to –44°C)[17]. This low Tg′ can lead to collapse during primary drying unless careful drying cycles or bulking agents are used. However, when arginine remains amorphous throughout freeze-drying, it provides effective protection against desiccation and osmotic stress[17].
Arginine also minimizes protein adsorption to surfaces like glass vials, polystyrene containers, and chromatography columns during manufacturing, helping maintain protein concentration and activity[18]. For subcutaneous formulations, maintaining osmolality between 240 and 600 mOsm/kg is crucial to balance injection site comfort and arginine’s functional performance[18].
Allan Chemical Corporation supplies compendial-grade L-arginine, ensuring the high purity and consistency required for biopharmaceutical applications.
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
sbb-itb-aa4586a
6. Histidine
Histidine (L-Histidine, CAS No. 71-00-1) stands as one of the four most commonly used buffers in approved protein therapeutics, alongside acetate, phosphate, and tris [6]. Its main role is to regulate pH, a critical factor influencing the chemical and physical stability of protein formulations. Around 30% of biologics currently on the market are sold as lyophilized products, where histidine often plays a key role in maintaining solid-state pH [19][6]. These characteristics make it an essential component in both liquid and lyophilized formulations.
Stabilization Mechanism
Histidine stabilizes proteins through several pathways. Its ability to bind ligands is considered the primary mechanism, enhancing both thermodynamic and conformational stability [6]. Additionally, histidine can be excluded from the protein surface, which helps favor the protein’s folded state [21]. For colloidal stabilization, histidine adjusts protein-protein interactions by influencing the balance of attractive and repulsive forces between molecules, effectively reducing the risk of aggregation [6][21].
"Buffers comprise an integral component of protein formulations. Not only do they function to regulate shifts in pH, they also can stabilize proteins by a variety of mechanisms."
– Mark Cornell Manning, Journal of Pharmaceutical Sciences [6]
By maintaining a stable pH, histidine prevents chemical degradation pathways such as deamidation, oxidation, or hydrolysis [6][1]. It also interacts with proteins through hydrogen bonding and electrostatic interactions, improving solubility and minimizing self-association in the native state [21].
Applications in Protein Formulations
Histidine plays a critical role in various protein formulations:
- Liquid formulations: Histidine primarily regulates pH, which is vital for the stability of monoclonal antibody (mAb) products, especially at high concentrations (50–200 mg/mL) [20][21]. It addresses challenges like solubility, aggregation, and viscosity in these dense solutions.
- Lyophilized formulations: In freeze-dried products, histidine helps establish and maintain the microenvironmental pH, ensuring the protein remains stable and active after water removal [6].
- High-concentration formulations: By managing solubility and viscosity, histidine reduces the formation of aggregation-prone species in dense solutions [6][20].
- Frozen formulations: Histidine offers cryoprotection by minimizing pH shifts during freezing, outperforming inorganic buffers like phosphate [6].
| Formulation Type | Role of Histidine | Benefit |
|---|---|---|
| Liquid | pH Regulation | Maintains conformational and colloidal stability [6] |
| Lyophilized | Microenvironmental pH Control | Protects proteins during freezing and sublimation [6] |
| High-Concentration | Solubility & Viscosity Management | Reduces aggregation in dense solutions [6][20] |
| Frozen | Cryoprotection | Minimizes pH shifts during freezing [6] |
Compatibility with Proteins and Other Excipients
Histidine, as an amine-based buffer, has a high temperature coefficient, meaning pH may shift if storage conditions differ from preparation temperatures [22][5]. These shifts can indirectly affect protein stability and lead to pH-sensitive degradation processes like deamidation and isomerization [22][5].
"Amine buffers (e.g., Tris and histidine) have high temperature coefficients, storage at temperatures that are different from the temperature of preparation could shift formulation pH."
– Jalpa Patel et al., Bioprocess International [5]
Histidine residues within a protein sequence, or the excipient itself, are vulnerable to oxidation, especially at the imidazole ring [5]. This ring readily binds metal ions, which can bring oxidizing agents close to the protein and catalyze oxidation of nearby residues like methionine [5]. Trace metal ions (e.g., Fe³⁺, Cu²⁺) from impurities or packaging can accelerate this process via the Fenton reaction [1]. However, when combined with sugars and surfactants, histidine enhances overall formulation stability [1][3].
Performance in Stability Studies
Histidine is an organic buffer prone to photodegradation, so formulations should be protected from light to avoid adduct formation [6]. Compared to inorganic buffers, histidine offers better pH control and reduces freezing-induced pH shifts [6]. While polysorbates may degrade enzymatically or oxidize, histidine is generally more chemically stable, though it remains sensitive to photo-oxidation [20][21].
This stability makes histidine a cornerstone of modern protein therapeutics. For optimal results, high-purity grades (e.g., Emprove Expert) are recommended to minimize endotoxin levels and microbial contamination – critical for injectable applications [21]. It’s worth noting that histidine can interfere with some protein content assays, such as the Lowry method [6]. Allan Chemical Corporation supplies compendial-grade L-histidine, ensuring the purity and consistency required for biopharmaceutical use.
This content is for informational purposes only. Consult official guidelines and qualified professionals before making sourcing or formulation decisions.
7. Kolliphor HS 15
Kolliphor HS 15 (polyoxyl 15 hydroxystearate, CAS No. 70142-34-6) is a nonionic surfactant approved by the FDA for use in parenteral, oral, and ophthalmic applications [12]. It is made up of mono- and diesters of polyethylene glycol (PEG) with 12-hydroxy stearic acid, along with free, unmodified PEG [23]. Unlike polysorbates – commonly found in about 90% of approved monoclonal antibody formulations – HS15 is gaining attention as a strong alternative due to its resistance to enzymatic degradation [2][3].
Stabilization Mechanism
HS15 stabilizes proteins by competitively adsorbing at hydrophobic interfaces, including liquid/gas (air), liquid/liquid (silicone oil), and liquid/solid (glass or PDMS) [23]. By effectively "blocking" these interfaces, it prevents proteins from unfolding and forming aggregates [23]. While both HS15 and polysorbates work through competitive adsorption, HS15 stands out for its greater resistance to enzymatic hydrolysis by host cell proteins (HCPs) such as lipases and esterases [23].
"HS15 can compete with the widely used polysorbates in terms of interfacial activity and protein stabilization, while offering higher robustness against degradation by a lipase and an esterase."
– Nadine Löw et al., Pharma Solutions, BASF SE [23]
HS15 also has a lower peroxide content (<1 mequiv/kg) compared to Polysorbate 80 (2 mequiv/kg), reducing the risk of oxidative damage to proteins [23]. Stability studies show that HS15 effectively minimizes the formation of visible and subvisible particles in antibody formulations exposed to mechanical stress [23]. These features make it a reliable choice for stabilizing proteins in pharmaceutical formulations.
Applications in Protein Formulations
In liquid protein formulations, HS15 works by competing for adsorption at hydrophobic interfaces like liquid/air and liquid/solid surfaces [23]. It has been shown to reduce the release of silicone oil droplets from siliconized vials and prefilled syringes, which helps limit protein-silicone interactions that can lead to aggregation [23]. HS15 has been successfully tested with a variety of proteins, including polyclonal bovine IgG, monoclonal antibodies (e.g., panitumumab), fusion proteins (e.g., abatacept), and enzymes such as amylase [23][12].
Compatibility with Proteins and Other Excipients
HS15 is also highly compatible with common formulation components. It integrates well with biopharmaceutical buffer systems and excipients like histidine, sodium acetate, sodium chloride, maltose, and phosphate buffers [23]. It offers a distinct advantage in formulations containing residual HCPs, as it is less prone to enzymatic degradation by lipases and esterases compared to polysorbates [23]. While HS15 does contain ester bonds, it remains more stable under pharmaceutical conditions than polysorbates [23]. However, like other PEGylated surfactants, it is susceptible to oxidation of its polyoxyethylene (POE) groups [3]. Additionally, HS15 is noted for having a better toxicity profile than Polysorbate 80 [12].
Allan Chemical Corporation provides Kolliphor HS 15 in compendial grades, ensuring the high purity and consistency needed for biopharmaceutical applications.
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
8. Kolliphor EL
Kolliphor EL (Polyoxyl 35 castor oil, CAS No. 9004-35-7) is a nonionic surfactant approved by the FDA for use in parenteral, oral, and ophthalmic formulations [12]. It serves as a reliable alternative to polysorbates, particularly for addressing challenges like visible or subvisible particle formation caused by polysorbate degradation [12]. Its primary component is polyoxyethylene glycerol triricinoleate, with smaller amounts of polyethylene glycol ricinoleate and free PEG [3].
Stabilization Mechanism
Kolliphor EL works by stabilizing proteins through two key mechanisms: it competes for adsorption at hydrophobic interfaces and binds directly to exposed hydrophobic regions of proteins. This dual action helps reduce protein unfolding and aggregation [2][3][12]. These characteristics make it especially useful in formulations exposed to oxidative or mechanical stress.
Applications in Protein Formulations
Kolliphor EL has demonstrated its ability to protect proteins under challenging conditions. For example, it has been shown to stabilize amylase in oxidative environments and bovine serum albumin (BSA) during agitation [12]. This highlights its effectiveness in formulations prone to oxidative degradation or agitation-induced stress.
Compatibility with Proteins and Other Excipients
Kolliphor EL is compatible with a range of proteins, such as amylase and BSA. However, like polysorbates, it is susceptible to ester hydrolysis and polyoxyethylene (POE) oxidation [12][3]. To mitigate these risks, it’s important to screen for residual host cell proteins that possess lipase or esterase activity, which can accelerate enzymatic hydrolysis. Additionally, selecting the highest purity grades can help minimize degradation byproducts like peroxides [2].
Allan Chemical Corporation offers compendial-grade Kolliphor EL, ensuring the purity and consistency required for biopharmaceutical applications.
This information is provided for reference only. Always consult regulatory guidelines and qualified experts when making sourcing or formulation decisions.
9. Phenylalanine
Phenylalanine (CAS No. 63-91-2) is an aromatic amino acid widely used as a stabilizing excipient in protein formulations. It holds GRAS status and is FDA-approved for both injectable and oral applications [12]. Unlike polysorbates, which can degrade due to chemical and enzymatic hydrolysis, phenylalanine remains chemically stable under standard storage conditions [20][12].
Stabilization Mechanism
Phenylalanine enhances protein stability through several mechanisms. It employs preferential exclusion, which strengthens the hydration shell around proteins and helps maintain their native folded state [21][2]. Additionally, it binds to partially unfolded proteins, preventing aggregation [21][2]. As an aromatic amino acid, phenylalanine also contributes stabilization via cation-pi interactions and dispersive forces [24].
Applications in Protein Formulations
Thanks to its diverse stabilization pathways, phenylalanine has shown promising results in combination with sucrose for lyophilized protein formulations. This pairing improves protein stability during drying and storage, making it particularly effective for formulations subject to temperature changes, pH variations, or freeze-thaw cycles [19][1][21].
Compatibility with Proteins and Other Excipients
Phenylalanine is generally compatible with other formulation components and primary packaging materials [3]. It stabilizes proteins through cation-pi interactions, dispersive forces, and hydrogen bonding, leveraging its aromatic side chain [24]. Moreover, phenylalanine can promote protein solubilization and assist in refolding by modifying protein-solvent interactions and acting as a molecular crowder [3]. However, its residues are prone to photo-oxidation under visible or near-UV light, which can lead to structural changes and potential loss of bioactivity [20].
This information is intended for reference purposes only. Always consult regulatory guidelines and qualified professionals when making sourcing or formulation decisions.
Conclusion
When working with protein-based therapeutics, the fragility of proteins cannot be overstated. Proteins possess minimal stabilization energy, making them highly susceptible to chemical degradation (such as oxidation, deamidation, and hydrolysis) and physical instability (like denaturation, aggregation, and precipitation) throughout manufacturing, transport, and storage [19][20]. This sensitivity highlights the importance of using excipients that not only stabilize but also protect proteins under stringent quality standards.
Compendial-grade excipients play a key role in ensuring both safety and efficacy. These high-purity excipients address common degradation pathways by minimizing impurities and reducing the risk of aggregation. In contrast, lower-grade excipients can introduce contaminants that accelerate protein degradation and aggregation. Protein aggregates not only lose their biological function but may also provoke adverse immune responses, which can be life-threatening for patients [21][2].
"A good practice is to use compendial excipients in the development study and control the quality/storage of the excipients from the very beginning, so no surprises are encountered in GMP manufacturing and subsequent long-term storage." – Jay Kang, Director of Analytical and Formulation Development, Patheon [1]
In addition to stabilizing formulations, high-purity excipients simplify regulatory compliance. Regulatory agencies require injectable formulations to have minimal visible and sub-visible particles [21]. Compendial-grade excipients that meet standards such as USP, Ph. Eur., or JP come with thorough documentation, streamlining the qualification and risk assessment process [25]. For biopharmaceutical manufacturers, collaborating with experienced specialty chemical providers ensures access to reliable stabilizers backed by robust quality systems and regulatory expertise.
Allan Chemical Corporation brings over four decades of experience in supplying compendial-grade excipients. From sucrose to polysorbates, their offerings support the stringent demands of biopharmaceutical formulations. With direct manufacturer relationships, just-in-time delivery capabilities, and a dedicated technical support team, Allan Chemical ensures access to high-purity stabilizers – including USP, FCC, ACS, and NF grades – needed to meet stringent quality and regulatory requirements. Their expertise helps formulation scientists achieve consistent, high-quality results for protein-based therapeutics.
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
FAQs
What advantages does sucrose offer compared to trehalose in stabilizing protein formulations?
Sucrose offers distinct advantages over trehalose in protein formulations. It plays a vital role in improving storage stability by minimizing protein aggregation. Furthermore, sucrose helps preserve protein structure by creating a fully amorphous state. Research also indicates that increasing sucrose concentrations consistently enhances protein stability, making it a dependable option for safeguarding delicate proteins during both formulation and storage.
What are the differences between polysorbate 20 and polysorbate 80 in protein formulation stability and use?
Polysorbate 20 (PS20) and Polysorbate 80 (PS80) are widely used excipients in protein formulations, appreciated for their role in stabilizing proteins by reducing aggregation and shielding them from interfacial stress. Despite their shared purpose, they exhibit distinct stability characteristics and are suited to different applications.
PS20 tends to degrade more readily through hydrolysis and oxidation. This degradation can release free fatty acids, potentially leading to the formation of subvisible particles. As a result, PS20 is more vulnerable to elevated temperatures or enzymatic activity, which can compromise long-term stability. On the other hand, PS80 is less prone to enzymatic hydrolysis but has its own challenges. Its fatty acid composition makes it susceptible to autooxidation, which can also impact the stability of formulations over time.
Selecting between PS20 and PS80 requires careful consideration of factors like oxidative stress, the presence of host cell proteins, and storage conditions. While both excipients are effective at stabilizing proteins, their performance is closely tied to the specific demands and conditions of the formulation.
Why is arginine commonly used in high-concentration protein formulations for subcutaneous injections?
Arginine is commonly incorporated into high-concentration protein formulations for subcutaneous injections because it helps lower viscosity, making the solution easier to inject. Beyond this, arginine supports protein solubility and refolding, which are crucial for preserving the formulation’s stability.
It also plays a vital role in reducing protein aggregation and interactions, ensuring that proteins stay functional and effective, even at high concentrations. These characteristics make arginine an important component in creating stable, injectable protein-based biopharmaceuticals.





Comments are closed