Artificial sweeteners, like Aspartame (CAS No. 22839-47-0), are low-calorie compounds used to mimic sugar’s sweetness. These additives are highly concentrated – Aspartame is 200 times sweeter than sugar – making them popular in diet sodas and sugar-free desserts. The FDA regulates them as food additives, requiring strict purity, safety, and labeling standards.
Six high-intensity sweeteners are FDA-approved: Saccharin, Aspartame, Acesulfame Potassium (Ace-K), Sucralose, Neotame, and Advantame. Each must meet rigorous thresholds for purity and contaminant levels, as outlined in the Food Chemicals Codex (FCC) and United States Pharmacopeia (USP). For example, Aspartame must include a warning for individuals with Phenylketonuria (PKU) and adhere to usage limits, such as a maximum of 0.5% in baked goods.
Manufacturers must ensure compliance by following Good Manufacturing Practices (GMP), conducting batch tests, and providing detailed documentation like Certificates of Analysis (CoAs). These requirements maintain safety and consistency across food-grade applications.
For businesses, staying compliant means working with suppliers who understand these regulations and provide high-quality, certified products. This ensures consumer safety and aligns with evolving global standards.
Key Regulatory Standards for Artificial Sweeteners
FCC Standards for Artificial Sweeteners
The Food Chemicals Codex (FCC) serves as the cornerstone for U.S. food-grade artificial sweeteners, setting standards for identity, purity, and quality. Published by the U.S. Pharmacopeial Convention, its 14th edition includes 1,282 monographs and 19 appendices, covering over 150 tests and assays[5].
FCC standards hold legal weight, referenced more than 200 times in the U.S. Code of Federal Regulations. The FDA mandates that artificial sweeteners like acesulfame potassium, aspartame, and sucralose meet FCC specifications[4]. Each monograph outlines tests for identification, purity (assay levels), and impurities – such as fluoride or heavy metals – to ensure safety[5].
Since December 2024, the FCC is exclusively available online, with updates released twice a year. These updates are essential for staying current with regulatory changes. Beyond U.S. borders, the FCC is recognized by regulatory authorities in at least nine countries, including Canada, Brazil, Australia, and Israel[5].
While FCC standards focus on food-grade sweeteners, the United States Pharmacopeia (USP) applies primarily to pharmaceutical-grade substances.
USP Monographs and Specifications
The United States Pharmacopeia (USP) specializes in pharmaceutical-grade drug substances and dosage forms[6]. Established in 1820, USP has a long history of developing standards for compounding and bulk substances[9]. Its monographs employ advanced analytical methods designed for the stringent requirements of regulated drug analysis. These standards are mandatory and enforceable by the FDA[9].
For food-related applications, manufacturers should prioritize FCC certification, as FDA food additive regulations (21 CFR Part 172) specifically reference FCC standards[4].
FDA Approval and GRAS Status

FDA oversight of artificial sweeteners builds on the testing and purity benchmarks outlined in FCC and USP guidelines. The agency regulates sweeteners through two primary pathways: food additive approval and Generally Recognized as Safe (GRAS) status. High-intensity sweeteners such as saccharin, aspartame, acesulfame potassium (Ace-K), sucralose, neotame, and advantame must comply with specific sections of the Code of Federal Regulations, detailing their approved uses and maximum levels. Both FCC and FDA guidelines establish similar purity and impurity limits. For example, aspartame must meet the criteria specified in the 7th edition of the FCC (pages 73–74)[2].
For GRAS designation (21 CFR Part 170), detailed data on identity, manufacturing processes, and specifications – aligned with FCC standards – are required[7]. GRAS status can be achieved either through rigorous scientific evaluation or by demonstrating a history of common use before 1958[3].
This content is for informational purposes only. Always consult official regulations and qualified professionals when making decisions about sourcing or formulation.
FDA to Change 52 Food Standards
Regulatory Requirements for Common Artificial Sweeteners
FDA-Approved Artificial Sweeteners: Regulatory Standards and Purity Requirements
This section outlines the specific standards and regulations that approved artificial sweeteners must meet, based on guidelines from the FCC and FDA.
Each high-intensity sweetener is subject to strict rules regarding purity, contaminant levels, and permissible uses. FDA-approved sweeteners must comply with the technical requirements detailed in 21 CFR Part 172 to ensure safety and consistency.
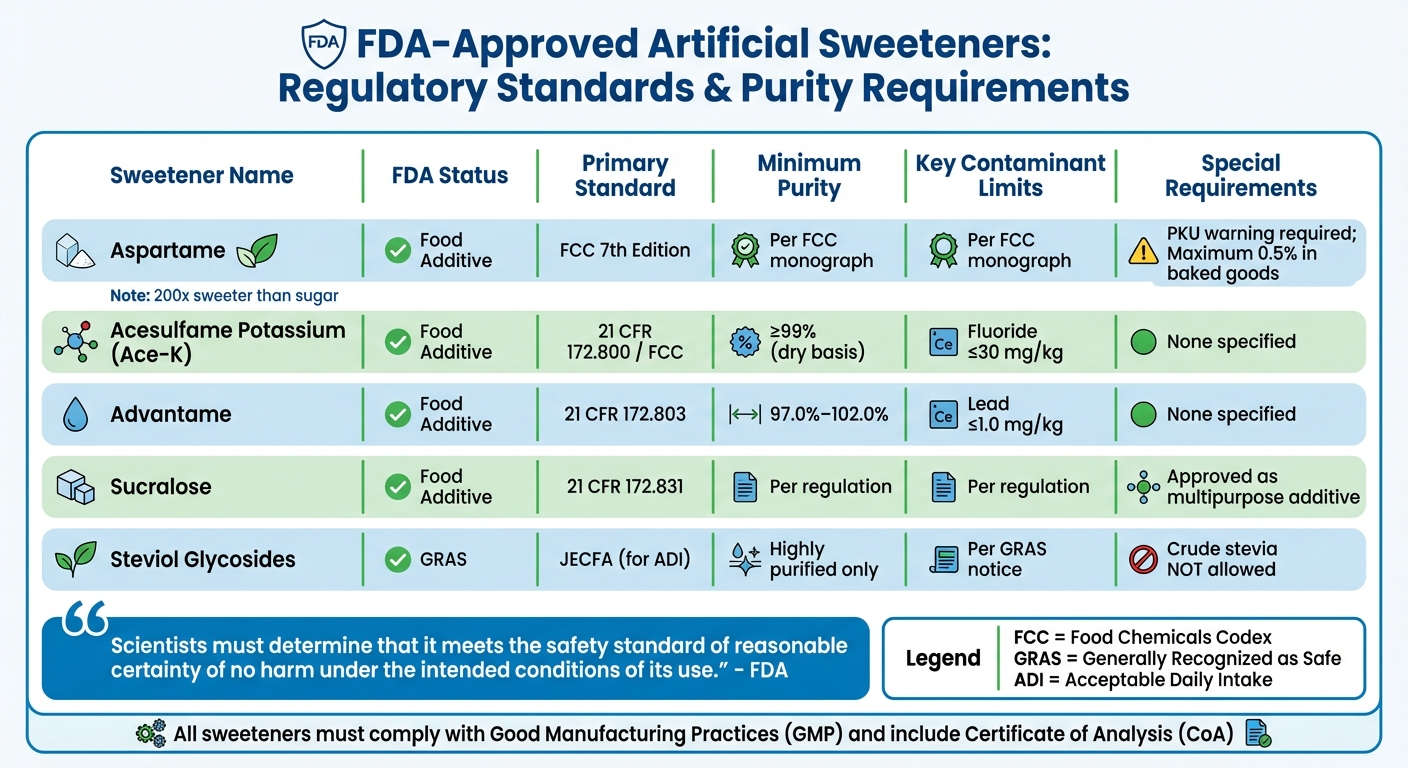
Aspartame, for example, must conform to the FCC 7th Edition monograph, include a warning for individuals with phenylketonuria (PKU), and is restricted to a maximum concentration of 0.5% by weight in baked goods. Additionally, table-top sweeteners containing aspartame typically advise against using it in cooking or baking[2]. Other sweeteners face similarly stringent requirements.
Acesulfame potassium must achieve a purity of at least 99% on a dry basis, with fluoride levels capped at 30 mg/kg[4]. Advantame must fall within an assay range of 97.0%–102.0% and have lead content no greater than 1.0 mg/kg[10]. Sucralose, regulated under 21 CFR Part 172.831, is approved as a multipurpose additive, allowing its use across a wide range of food products[10].
"Regardless of whether a substance is approved for use as a food additive or its use is determined to be GRAS, scientists must determine that it meets the safety standard of reasonable certainty of no harm under the intended conditions of its use." – FDA[1]
Table: FCC vs. USP Standards Comparison
The table below highlights the differences between FCC and USP standards for several sweeteners.
| Sweetener | FDA Status | Primary Standard | Minimum Purity | Key Contaminant Limits | Special Requirements |
|---|---|---|---|---|---|
| Aspartame | Food Additive | FCC 7th Ed.[2] | Per FCC monograph | Per FCC monograph | PKU warning; max 0.5% in baked goods |
| Acesulfame Potassium | Food Additive | 21 CFR 172.800 / FCC[4] | ≥ 99% (dry basis) | Fluoride ≤ 30 mg/kg | None specified |
| Advantame | Food Additive | 21 CFR 172.803[10] | 97.0%–102.0% | Lead ≤ 1.0 mg/kg | None specified |
| Sucralose | Food Additive | 21 CFR 172.831[10] | Per regulation | Per regulation | Approved as a multipurpose additive |
| Steviol Glycosides | GRAS | JECFA (for ADI)[1] | Highly purified only | Per GRAS notice | Crude stevia not allowed |
The FDA also determines an Acceptable Daily Intake (ADI) for approved additives during its premarket review. This ADI represents the amount considered safe for daily consumption over a lifetime[1]. Manufacturers are responsible for ensuring that their raw materials meet these specifications, which must be verified through certificates of analysis and batch-specific documentation.
This information is intended for educational purposes only. Always consult official regulations and qualified experts before making sourcing or formulation decisions.
sbb-itb-aa4586a
Compliance Requirements for Food-Grade Applications
Ensuring artificial sweeteners meet regulatory standards involves adhering to certified manufacturing practices and maintaining thorough documentation throughout the supply chain.
Manufacturing and Sourcing Certifications
Artificial sweeteners, like all food additives, must be produced following Good Manufacturing Practice (GMP) guidelines[11][2]. The FDA mandates that production facilities be registered and inspected, with operations complying with current Good Manufacturing Practices (cGMP) as outlined in 21 CFR part 211[8].
To ensure consistency, manufacturers are required to conduct batch analysis, typically testing at least five batches to confirm uniform identity and purity. These analyses must also verify that contaminants such as lead, arsenic, cadmium, and mercury remain within safe limits[11].
Controlling impurities is critical for consumer safety, and this principle applies directly to food-grade sweeteners. For added assurance, many producers rely on quality management systems like ISO 9001:2015, which help maintain consistent production standards. For example, Allan Chemical Corporation offers USP and FCC-grade sweeteners that meet stringent quality benchmarks, ensuring batch-to-batch reliability. Such rigorous processes not only enhance manufacturing but also simplify the subsequent documentation and technical review phases.
Required Documentation and Technical Support
Thorough documentation is essential to demonstrate regulatory compliance. Each shipment must include a Certificate of Analysis (CoA) and a Safety Data Sheet (SDS). These documents provide critical details such as the chemical name, CAS number, empirical formula, purity, test methods, and contaminant limits[8][11].
Technical documentation should align with established standards like the Food Chemicals Codex (FCC) or United States Pharmacopeia (USP). For instance, aspartame regulations outlined in 21 CFR 172.804 explicitly reference the FCC 7th edition, making adherence to FCC specifications a legal obligation[2].
Stability testing is another key requirement, verifying that the sweetener maintains its performance over its shelf life. Recovery rates for analyte concentrations above 0.1 mg/kg must fall between 80% and 110%[11]. Additionally, analytical methods must deliver a relative standard deviation below 10% for measurements exceeding 0.1 mg/kg[11].
This content is intended for informational purposes only. Always consult official regulations and qualified professionals when making sourcing or formulation decisions.
Conclusion
Meeting regulatory standards is not just a recommendation for artificial sweeteners – it’s a legal requirement. The FDA has approved six sweeteners for use as food additives: aspartame, acesulfame potassium, sucralose, neotame, advantame, and saccharin. Each of these must meet strict purity standards and usage limits to ensure safety and compliance.
Navigating these regulations demands working with suppliers who are well-versed in FCC and USP specifications. The Food Chemicals Codex (FCC), frequently cited in the U.S. Code of Federal Regulations, offers detailed guidance. Suppliers adhering to current Good Manufacturing Practices (cGMP) and providing extensive technical documentation – such as batch analyses, stability data, and impurity limits – can significantly simplify the compliance process for manufacturers [11][13].
Allan Chemical Corporation brings over 40 years of expertise in sourcing FCC- and USP-grade artificial sweeteners. Their offerings include Certificates of Analysis, Safety Data Sheets, and technical support, all designed to meet regulatory demands. By collaborating with thoroughly vetted manufacturers, Allan Chemical ensures timely delivery of compliant solutions, helping businesses stay ahead of regulatory changes.
Choosing experienced suppliers is key to maintaining compliance and fostering growth in regulated markets. As global standards evolve and regulatory frameworks modernize, reliable partners help safeguard brand integrity, prioritize consumer safety, and support long-term success [12].
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
FAQs
What regulations oversee the safety and use of artificial sweeteners in the U.S.?
In the United States, artificial sweeteners are carefully regulated to ensure both their safety and proper use. The Food and Drug Administration (FDA) mandates that high-intensity sweeteners undergo thorough safety evaluations before being approved as food additives. These evaluations are designed to confirm that the sweeteners meet strict safety criteria for human consumption.
In addition to these evaluations, artificial sweeteners must adhere to federal regulations, including 21 CFR Part 172 and Part 170. These sections provide detailed guidelines for food additives and ingredients, ensuring that sweeteners meet quality standards and are safe for their intended applications in food products.
What is the difference between FCC and USP standards for artificial sweeteners?
The FCC standards are designed to guarantee the safety, purity, and identity of food-grade ingredients, including items like artificial sweeteners. These guidelines are widely acknowledged in the food industry and cover key aspects such as solubility and purity to ensure ingredient safety.
In contrast, USP standards are tailored primarily for pharmaceuticals, dietary supplements, and certain food ingredients. They focus on medical-grade safety and quality, setting rigorous benchmarks to ensure ingredients meet the high standards required in regulated industries.
While both sets of standards are essential, their application depends on the intended use – whether ensuring food-grade or pharmaceutical-grade quality.
What documentation is needed to meet FDA requirements for artificial sweeteners?
To meet FDA regulations for artificial sweeteners, you’ll need to prepare essential documentation. This includes safety assessments, a thorough explanation of the manufacturing process, and proof of compliance with standards such as GRAS (Generally Recognized as Safe) or food additive petitions.
Additionally, the FDA mandates the submission of chemical and technological data. This data should cover the sweetener’s composition, its intended applications, and results from safety testing. Adhering to these requirements ensures your product aligns with regulatory standards and prioritizes consumer safety.





Comments are closed