Thermal stability testing helps identify how chemicals react to heat, ensuring safe handling, storage, and transport. It evaluates risks like decomposition, polymerization, and runaway reactions, which can lead to fires, explosions, or toxic gas releases. Key parameters such as Self-Accelerating Decomposition Temperature (SADT) and Time to Maximum Rate (TMR24) are used to define safe temperature limits and prevent accidents.
Quick Highlights:
- Thermal Stability: A chemical’s ability to resist changes when exposed to heat.
- Key Risks: Fires, explosions, and runaway reactions caused by exceeding critical temperatures.
- Testing Methods:
- DSC: Detects reaction onset and heat flow.
- TGA: Measures mass changes during heating.
- ARC: Simulates bulk conditions for runaway potential.
- Regulatory Compliance: SADT and SAPT thresholds guide safe storage and transport, with mandatory temperature controls for sensitive materials.
Thermal stability testing is essential for chemical safety, regulatory compliance, and designing safe industrial processes.
Thermal Decomposition and Runaway Reactions
Physical and Chemical Changes During Heating
When substances are heated, they undergo both physical and chemical changes, which can lead to significant safety hazards. Physical changes include processes like melting, evaporation, sublimation, and recrystallization. For instance, polymorphism – where a material shifts between different crystal forms – or the recrystallization of amorphous substances is commonly observed in pharmaceutical applications [4][5]. While these changes might seem harmless, they can influence a chemical’s stability and reactivity in unexpected ways.
Chemical changes, however, present much greater risks. Thermal instability can result from decomposition (molecular fragmentation), self-reactions, autopolymerization, or oxidation (reaction with oxygen in the air) [1][2]. These processes often generate heat, and when the heat builds up faster than it can dissipate, the system risks entering thermal runaway. This is a dangerous scenario where temperature and reaction rates increase exponentially, feeding into each other [1]. For example, powders with smaller particle sizes have a higher surface-area-to-volume ratio, which can accelerate oxidation. What starts as a slow smoldering or glowing reaction can quickly escalate into flaming combustion, releasing toxic or flammable gases.
Important Thermal Stability Parameters
To predict and prevent runaway reactions, understanding key thermal stability parameters is crucial. These include:
- Onset Temperature (T₀): The point where a material starts to decompose or react at a detectable rate.
- Time to Maximum Rate (TMR): The time required for a reaction to reach its peak rate under adiabatic conditions (no heat loss). The standard TMR24 specifies the temperature at which this takes exactly 24 hours [1].
- Adiabatic Temperature Rise (ΔTₐd): The estimated maximum temperature increase, representing the worst-case scenario for runaway reactions.
One especially critical parameter is the Temperature of No Return (TNR). Once this temperature is exceeded, the heat generated surpasses the system’s ability to cool itself, making runaway reactions inevitable [1]. For example, a study conducted in February 2025 examined methyl methacrylate (MMA) stored in a 55-gallon insulated drum at 68°F (20°C). The substance remained stable until mechanical agitation added 20 watts of frictional heat, pushing it past its TNR of 91.4°F (33.0°C). Based on these findings, the study recommended shutting off agitators when temperatures approach about 5°C (9°F) below the TNR to prevent self-accelerating polymerization [1].
These parameters form the foundation for safe storage practices, which are explored further in the next section.
Long-Term Storage Stability
Ensuring long-term storage stability is vital to avoid delayed runaway reactions, and this requires careful analysis of both isothermal (constant temperature) and non-isothermal (temperature-ramped) behaviors. Bulk containers, in particular, can retain heat long enough for slow reactions to accelerate into dangerous conditions [1][2]. Standard screening methods like Differential Scanning Calorimetry (DSC) may not always detect oxidative self-heating in powders because they rely on small, oxygen-limited samples. For larger quantities, specialized tests such as the Basket Test are more effective. This method considers the volume-to-surface-area ratio to determine safe storage conditions for bulk materials [2].
Kinetic modeling, often using the Arrhenius equation, helps calculate TMR at different storage temperatures, predicting how long a material can remain stable before reaching hazardous thresholds. For chemicals with inhibitors, like MMA, it’s important to account for the depletion of the inhibitor. Once the inhibitor is exhausted, self-heating begins [1]. Regulatory guidelines, such as those from the United Nations Recommendations on the Transport of Dangerous Goods, require temperature-controlled transport when the Self-Accelerating Decomposition/Polymerization Temperature (SADT/SAPT) falls below 122°F (50°C) for intermediate bulk containers or 113°F (45°C) for portable tanks [1].
This content is for informational purposes only. Always consult official regulations and qualified professionals before making decisions related to sourcing or formulation.
HEL Thermal screening / adiabatic calorimetery for chemical hazard testing
Common Thermal Stability Testing Methods
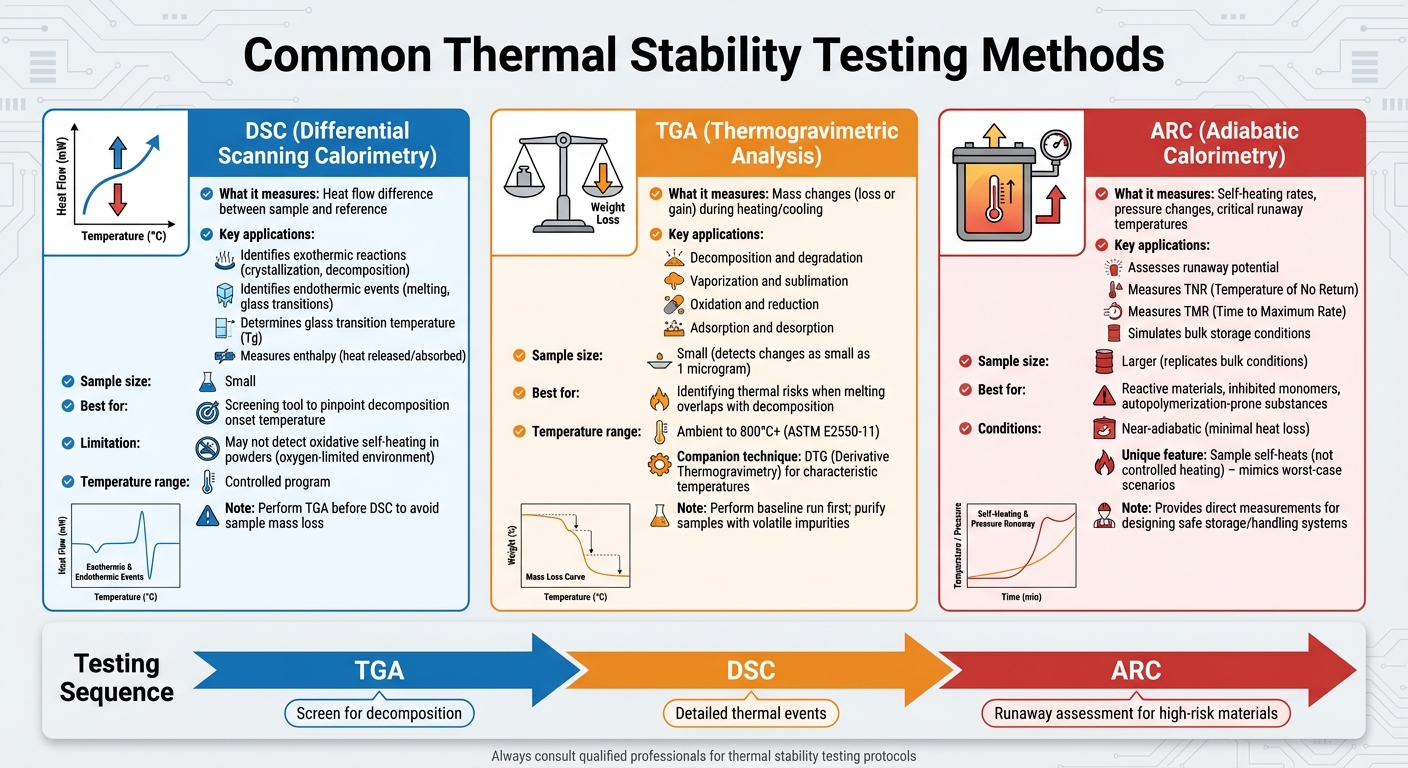
Thermal Stability Testing Methods Comparison: DSC vs TGA vs ARC
Understanding how to test thermal stability is crucial for turning data into practical safety measures. The choice of method depends on the specific heat-related behavior of the material. Techniques like Differential Scanning Calorimetry (DSC), Thermogravimetric Analysis (TGA), and Adiabatic Calorimetry (ARC) each provide unique insights into how materials react to heat. Together, they help establish safer chemical handling practices.
Differential Scanning Calorimetry (DSC)
DSC measures the difference in heat flow between a sample and a reference material as they are heated or cooled through a controlled temperature program [6][7]. This method identifies exothermic reactions (like crystallization or decomposition) and endothermic events (such as melting or glass transitions) [7]. It’s often used as a screening tool to pinpoint the temperature at which decomposition begins and to measure the total heat released or absorbed during a reaction (enthalpy) [6].
One key application of DSC is determining the glass transition temperature (Tg), which marks the change in polymers from a hard, brittle state to a more flexible, rubbery one [6]. To ensure accurate results, calibration with standard references is essential [6].
Experts recommend performing TGA before DSC to identify the decomposition temperature and avoid sample mass loss during DSC analysis [6]. As Nurul Fatahah Asyqin Zainal from Universiti Teknologi MARA explains:
"TGA is also highlighted where the analysis gives important thermal degradation information of a material to avoid sample decomposition during the DSC measurement" [6].
However, DSC’s controlled, oxygen-limited environment might not detect oxidative self-heating in powders [2].
Thermogravimetric Analysis (TGA)
TGA measures changes in a material’s mass – whether loss or gain – while it is heated or cooled in a controlled atmosphere [8][6]. It can identify various chemical and physical processes, including decomposition, degradation, vaporization, sublimation, oxidation, reduction, adsorption, and desorption [8]. This method is especially helpful when a material’s melting point overlaps with its decomposition onset, as melting signals in DSC can obscure decomposition data [4].
Standard TGA protocols, such as ASTM E2550-11, typically cover temperatures from ambient up to 800°C, though some instruments can go beyond this range [4]. Advanced thermobalances can detect mass changes as small as one microgram [6]. Derivative Thermogravimetry (DTG) is often used alongside TGA to pinpoint characteristic temperatures, such as the inflection point indicating the highest rate of weight loss [6].
Before starting, it’s important to perform a baseline run (using an empty furnace or inert sample) under the same conditions to account for buoyancy effects and ensure sensor accuracy [6]. If the sample contains volatile impurities, purification is necessary to avoid interference with decomposition data [4]. TGA serves as a fast and effective method for identifying thermal risks in materials like energetic compounds, pharmaceuticals, and polymers [4].
Adiabatic Calorimetry (ARC)
While DSC and TGA focus on small samples under controlled conditions, ARC replicates bulk conditions where heat can accumulate. Adiabatic Calorimetry assesses runaway potential by measuring self-heating rates, pressure changes, and critical runaway temperatures under near-adiabatic conditions, meaning there is minimal heat loss to the environment. This approach mimics worst-case scenarios, such as when a material generates heat faster than it can dissipate, resembling conditions in bulk storage or processing.
ARC testing provides direct measurements of key parameters like the Temperature of No Return (TNR) and Time to Maximum Rate (TMR), which are crucial for designing safe storage and handling systems. This technique is particularly useful for reactive materials, inhibited monomers, and substances prone to autopolymerization. Unlike DSC and TGA, which follow controlled heating rates, ARC allows the sample to self-heat, offering a realistic view of runaway reaction behavior. These insights are invaluable for creating a comprehensive thermal stability testing strategy.
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
sbb-itb-aa4586a
How to Design a Thermal Stability Testing Plan
Designing a thermal stability testing plan involves a tiered strategy, starting with basic screening methods and progressing to more detailed evaluations as necessary. This approach saves time and resources while ensuring the collection of essential safety data. The goal is to closely replicate actual processing and storage conditions in a laboratory setting.
Step-by-Step Testing Approach
The process begins with quick screening techniques like Thermogravimetric Analysis (TGA) or Differential Scanning Calorimetry (DSC). These methods use small sample sizes to detect mass changes and pinpoint reaction onset temperatures, offering rapid insights into potential hazards. If the margin between the reaction onset temperature and the maximum process temperature is too narrow, more advanced testing, such as Adiabatic Calorimetry (ARC), should be conducted.
For powders, specialized tests like Diffusion Cell or Aerated Cell analysis can measure oxidative self-heating. The Basket Test is another useful method for extrapolating data to assess the safety of bulk storage. These initial findings guide the selection of testing conditions that accurately reflect plant environments, ensuring relevant and actionable results.
Choosing the Right Test Conditions
The test atmosphere must mirror the actual conditions of the plant – whether inert, reactive, or self-generated. For powders, oxygen availability during testing is crucial since smaller particles have a higher surface area-to-volume ratio, which can accelerate self-heating. Always use samples that represent production conditions. If volatile impurities interfere with TGA results, purify the sample or interpret the data cautiously.
After initial temperature-ramped screenings, follow up with isothermal tests to identify optimal process temperatures and safe exposure times. Properly chosen test conditions ensure that the data supports effective safety planning and decision-making.
Using Test Data for Safety Planning
The data collected during testing plays a critical role in process design, storage protocols, and emergency response planning. Parameters like the Self-Accelerating Decomposition Temperature (SADT) and Time to Maximum Rate (TMR) are essential for determining safe storage conditions for bulk materials. However, screening techniques like TGA should always be paired with more comprehensive hazard analyses to define safe operating limits. As highlighted in ASTM E2550-11:
"This test method [TGA] shall not be used by itself to establish a safe operating or storage temperature. It may be used in conjunction with other test methods… as part of a hazard analysis of a material."
Given the complexity of factors such as heating rates and material construction, it’s important to have an expert review your thermal stability data. Expert evaluation ensures that laboratory results translate effectively into safe plant operations by accounting for scale-up, environmental conditions, and potential accelerators of degradation. These steps align with industry standards, supporting the safe handling and storage of chemicals throughout the facility.
This information is provided for educational purposes only. Always consult official regulations and qualified professionals when making decisions about sourcing or formulations.
Using Thermal Stability Data for Chemical Safety
Translating Test Data into Safety Measures
Thermal stability test data plays a crucial role in creating safety protocols that protect both workers and facilities. Key parameters like Time to Maximum Rate (TMR24) and Temperature of No Return (TNR) are the backbone of these safety measures. TMR24 indicates the temperature at which a material takes 24 hours to reach its maximum decomposition rate under adiabatic conditions, aligning with typical industrial operating cycles [1]. On the other hand, TNR marks the point where heat generation surpasses the system’s cooling capacity – beyond this threshold, thermal runaway becomes inevitable without extraordinary intervention [1].
To prevent such scenarios, safety margins are established. For example, programming agitators to automatically shut off at temperatures just below the TNR can stop runaway reactions. A practical protocol might involve shutting off agitators when materials reach 5°F below the TNR, allowing them to cool safely back to ambient levels [1]. In bulk storage, thermal stability data helps determine packaging that balances external heat protection with internal heat dissipation [1]. These measures seamlessly integrate into broader plant safety strategies, ensuring operations remain controlled and secure.
Meeting Regulatory Requirements
Thermal stability data is also critical for meeting regulatory standards across industries. For instance, the FDA mandates stability protocols for biological products to confirm they retain safety, identity, purity, and potency throughout their shelf life [9]. Similarly, assessing the physical stability of pharmaceutical raw materials helps avoid costly recalls due to unexpected transformations [5].
In transportation, thermal stability parameters are essential for compliance. The UN Recommendations on the Transport of Dangerous Goods (UNRTDG) classify self-heating solids and monomers as Class 4, Division 4.1 substances if their Self-Accelerating Decomposition Temperature (SADT) or Self-Accelerating Polymerization Temperature (SAPT) is 167°F or lower and the heat of reaction exceeds 300 J/g [1]. Temperature controls become mandatory during transport if the SADT or SAPT falls below 122°F for intermediate bulk containers or 113°F for portable tanks [1]. These thresholds ensure materials remain stable during shipping and storage.
Testing standards like ASTM E2550-11 provide a baseline for identifying thermal hazards, though they should be used alongside other methods. Swati Umbrajkar, Ph.D., Manager of the Chemical Process Evaluation Group at Chilworth Technology, highlights the importance of this data:
"Understanding the thermal stability of all processed products (raw materials, intermediates, final products, by‑products, and waste streams)… is a pre‑requisite for safe processing." [2]
Working with Trusted Chemical Suppliers
To implement effective safety measures, partnering with reliable chemical suppliers is essential. Trusted suppliers provide comprehensive thermal stability data, which is critical for on-site safety protocols. This information is typically detailed in Safety Data Sheets (SDSs), especially in Section 10 (Stability and Reactivity) and Section 14 (Transport Information), covering stabilizers and storage conditions necessary to maintain chemical stability [10].
Suppliers also manage chemical inhibitors that prevent unwanted heating and exothermic reactions in unstable materials [1]. Since inhibitors are consumed over time – at a faster rate as temperatures rise – supplier-provided storage guidelines are vital for maintaining safety [1]. With global plastics production increasing by 12% between 2017 and 2021, the volume of unstable initiators and monomers in the supply chain has grown [1]. This makes it even more critical to work with suppliers who conduct stability testing under real-world conditions, ensuring accurate heat transfer and stability data [3].
For example, Allan Chemical Corporation, with over 40 years of experience, sets a high standard by providing complete thermal stability documentation. Their technical-grade and compendial-grade solutions (USP, FCC, ACS, NF) come with verified safety data. By maintaining direct relationships with manufacturers, they ensure customers receive materials with reliable thermal stability information. When sourcing thermally sensitive materials, facilities should request specific kinetic data, such as TMR24 and SADT values, to establish safety measures before products even arrive on-site.
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Conclusion
Thermal stability testing plays a crucial role in ensuring the safe handling, storage, and transport of chemicals. By identifying risks such as runaway reactions, fires, explosions, and toxic gas releases, it establishes critical safety thresholds like Self-Accelerating Decomposition Temperature (SADT), Self-Accelerating Polymerization Temperature (SAPT), and Time to Maximum Rate (TMR24). These parameters directly influence storage guidelines, transport classifications, and emergency response frameworks [1]. A comprehensive testing approach is essential to address the diverse challenges posed by different materials.
Relying on a single testing method isn’t enough. Screening techniques like Differential Scanning Calorimetry (DSC) and Thermogravimetric Analysis (TGA) provide quick insights into potential hazards. For powders, specialized methods such as the Diffusion Cell or Basket Test are better suited to detect oxidative self-heating in low-oxygen environments, which standard tests might miss [2].
Accurate thermal stability data is also a cornerstone of regulatory compliance. For example, the FDA’s Section 211.166 mandates written stability programs to determine appropriate storage conditions and expiration dates [3]. Similarly, the United Nations Recommendations on the Transport of Dangerous Goods (UNRTDG) requires temperature controls if a material’s SADT or SAPT is below 122°F (50°C) for intermediate bulk containers or 113°F (45°C) for portable tanks [1].
Working with experienced suppliers, like Allan Chemical Corporation, ensures access to reliable thermal stability documentation. Their technical- and compendial-grade products (USP, FCC, ACS, NF) are supported by verified stability data, reducing uncertainty and risk. When sourcing thermally sensitive chemicals, always request detailed kinetic data, such as TMR24 and SADT values, to ensure safety from the outset.
The importance of thermal stability testing cannot be overstated. As Swati Umbrajkar, Ph.D., Manager of the Chemical Process Evaluation Group at Chilworth Technology, aptly states:
"The assessment and understanding of thermal instability associated with process materials is a prerequisite for safe manufacturing" [2].
By combining robust testing protocols with dependable supplier partnerships, companies can safeguard their workforce, meet regulatory requirements, and maintain operational efficiency. These practices are the foundation of a proactive safety strategy that supports every aspect of chemical management.
This content is for informational purposes only. Always consult official regulations and qualified professionals when making sourcing or formulation decisions.
FAQs
How do DSC, TGA, and ARC differ in thermal stability testing?
Differential Scanning Calorimetry (DSC), Thermogravimetric Analysis (TGA), and Accelerating Rate Calorimetry (ARC) are essential tools for assessing thermal stability, each serving a unique role in understanding material behavior under heat.
DSC examines heat flow in or out of a sample during heating or cooling. This technique highlights key thermal events such as melting, crystallization, and decomposition. However, it can face challenges in pinpointing decomposition temperatures if they overlap with melting processes.
TGA measures weight changes in a sample as it is heated, making it particularly effective for identifying mass losses caused by volatilization, decomposition, or oxidation. This method is invaluable when tracking how a material’s composition changes with temperature.
ARC takes a safety-focused approach by studying self-heating behavior in a sealed, pressurized environment under near-adiabatic conditions. It delivers vital insights into ignition temperatures, self-heating rates, and pressure-related effects, mimicking conditions that DSC and TGA can’t replicate.
By combining these techniques, researchers gain a well-rounded view of a material’s thermal properties and safety characteristics, offering critical insights for both development and risk assessment.
What is the Temperature of No Return (TNR) and why is it critical for chemical safety?
The Temperature of No Return (TNR), often called the critical temperature, marks the point where a chemical reaction spirals out of control. At this stage, external cooling is no longer effective, and the reaction speeds up dramatically. This can lead to hazardous outcomes such as fires, explosions, or dangerous pressure buildup.
To avoid these risks, strict safety measures are implemented to ensure operating, storage, and transport temperatures stay well below the TNR. These precautions include controlling batch sizes, setting strict temperature limits, continuously monitoring temperature levels, and equipping systems with effective heat dissipation and pressure-relief mechanisms. For customers working with Allan Chemical Corporation, TNR values are clearly specified for each chemical, helping ensure safe handling and storage within these critical thresholds.
Why is it essential to work with trusted chemical suppliers for thermal stability testing?
Partnering with reliable chemical suppliers is essential for obtaining precise and dependable thermal stability data. This information plays a key role in ensuring the safe handling, storage, and transportation of chemicals. By having access to accurate data, you can reduce the likelihood of accidents, adhere to regulatory requirements, and mitigate risks tied to hazardous materials.
Trusted suppliers also offer timely insights and customized technical-grade solutions to address your unique needs. Their support helps maintain smooth and secure operations while aligning with industry standards.





Comments are closed