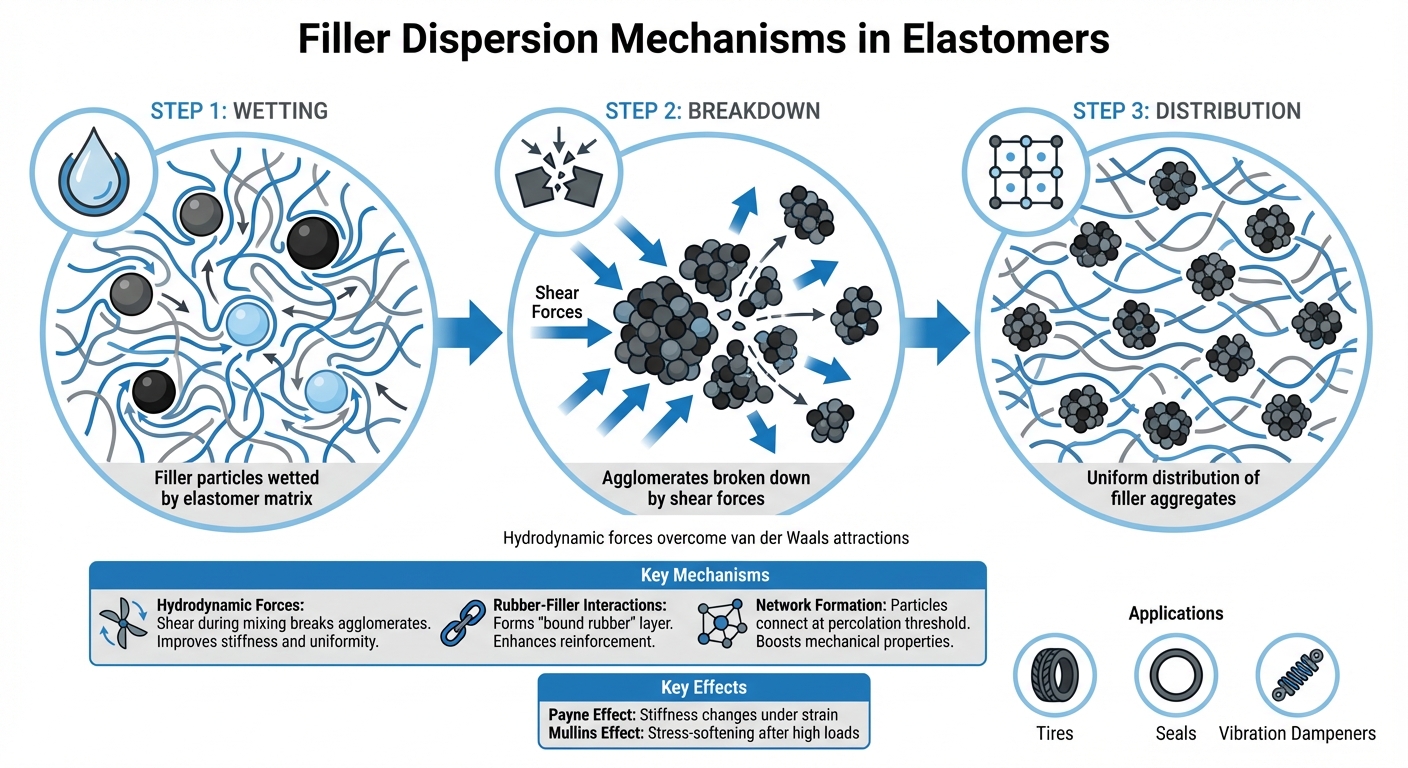
Filler dispersion in elastomers is the process of integrating nano-scale particles, such as Carbon Black or Silica, into a rubber matrix to improve material properties like stiffness, durability, and resistance to wear. This involves three steps: wetting the filler with the elastomer, breaking down filler agglomerates, and distributing filler aggregates evenly. Proper dispersion significantly impacts performance, influencing behaviors like the Payne Effect (stiffness changes under strain) and Mullins Effect (stress-softening after high loads).
Key points include:
- Hydrodynamic Forces: Shear during mixing breaks apart agglomerates, improving stiffness and uniformity.
- Rubber-Filler Interactions: Strong polymer-filler bonds form a rigid "bound rubber" layer, enhancing reinforcement.
- Network Formation: Filler particles form a connected structure at the percolation threshold, boosting mechanical properties.
- Dynamic Properties: Excessive filler can cause issues like the Payne Effect, where stiffness drops under strain.
- Mixing Techniques: Mechanical shear and chemical surface treatments (e.g., silane coupling agents) ensure even dispersion.
Uniform filler dispersion reduces weak points in elastomers, directly improving performance in applications like tires, seals, and vibration dampeners. Effective dispersion techniques combine precise mechanical mixing with chemical surface treatments, allowing manufacturers to achieve consistent results.
Three-Step Filler Dispersion Process in Elastomers
Filler Dispersion: The Hidden Factor That Makes or Breaks Rubber Performance
How Filler Dispersion Works in Elastomers
Filler dispersion involves breaking apart agglomerates using applied shear, where the combination of residence time and shear rate determines the quality of the dispersion. Higher shear strains fragment large agglomerates into smaller aggregates, which then distribute more uniformly within the elastomer matrix. This process occurs as hydrodynamic forces counteract the cohesive van der Waals attractions between particles. During mixing, manufacturers use mechanical shear to achieve the desired dispersion state[3]. Below, we examine how hydrodynamic forces and rubber–filler interactions influence this process.
Hydrodynamic Effect on Particle Movement
The Guth equation, E = E₀(1 + 0.67fΦ + 1.62f²Φ²), illustrates how rigid filler particles increase the stiffness of a composite. Here, E represents the modulus, f is the particle aspect ratio, and Φ is the filler volume fraction[5]. The shape of the filler plays a crucial role – anisotropic particles, such as rod-shaped carbon nanotubes or layered nanoplatelets, can align during processing to provide directional reinforcement. Additionally, fillers with intricate, branched aggregate structures trap occluded rubber within their voids, effectively increasing the filler volume and, consequently, the stiffness of the composite[4][6].
Rubber-Filler Interactions and Bound Rubber Formation
Strong interactions between filler surfaces and polymer chains result in the formation of a rigid, immobilized layer of rubber at the interface, referred to as bound rubber. This layer, which behaves like glass, cannot be removed via solvent extraction and becomes an integral part of the material’s reinforcing structure. A notable study by Brennan, Jermyn, and Boonstra demonstrated that heat-treating ISAF-grade carbon black at 3,000°C for one hour in styrene-butadiene rubber (SBR) reduced bound rubber content from 34.4% to 5.6%. This reduction also led to a significant drop in stress at 300% strain, from 14.5 MPa to 3.5 MPa[6]. To account for bound rubber in practical applications, engineers often estimate the effective particle diameter to be about 1.5 times larger than the actual primary particle diameter[3].
However, excessive bonding between the filler and polymer can have drawbacks. D.C. Edwards of Polysar Ltd. noted:
Only a minor amount of strong bonding is necessary or desirable, such that polymer/filler slippage can occur, under stress, over most of the interfacial area[2].
This controlled slippage allows stress to distribute more evenly and prevents void formation, ultimately improving the material’s durability under repeated loading. Striking the right balance between bonding and interfacial slippage is essential for optimizing stress distribution and enhancing elastomer performance.
Disclaimer: This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Filler Network Formation and Material Performance
When fillers disperse within an elastomer, they interact to form networks that strengthen the material. These networks arise when individual filler aggregates come close enough to bond through physical forces, creating a continuous framework that reinforces the rubber matrix. The shift from isolated particles to a connected structure happens at a specific concentration called the percolation threshold. Understanding this process is key to enhancing elastomer performance, as the network evolution directly influences the material’s mechanical and dynamic properties.
Percolation Threshold and Strength Enhancement
The percolation threshold is the critical concentration of filler at which individual aggregates start forming an interconnected 3D network within the elastomer matrix [7][5]. Below this threshold, the particles remain isolated or form small clusters, contributing stiffness mainly through hydrodynamic effects. Once the threshold is surpassed, interactions between fillers – such as hydrogen bonding in silica – create a continuous skeleton that significantly boosts reinforcement [5].
This network formation leads to notable improvements in mechanical properties. When the percolation threshold is reached, the material’s elastic modulus increases at low strain levels, as the rigid filler network provides structural support beyond the hydrodynamic contributions of individual particles [5][8]. For conductive fillers like carbon black or carbon nanotubes, the percolation threshold is also the point where the material transitions from being an insulator to a conductor, as a continuous pathway for electron flow is established [9]. For instance, studies on natural rubber reinforced with carbon nanotubes have shown a percolation threshold below 1 wt% [4].
The specific filler type and structure play a significant role in determining the percolation point. For example, titania particles generated in situ within PDMS form branched networks at loadings as low as 10 phr. In contrast, silica particles at the same loading create smaller, more dispersed domains (around 5 nm), requiring higher concentrations to achieve a similar network state [5]. Nanoscale fillers can reinforce the material at much lower concentrations compared to micron-scale fillers due to their larger polymer-filler interfacial area [5]. However, exceeding the optimal network density can lead to issues – above the percolation threshold, elastomers may exhibit "plastic-like" behavior and significant residual deformation. This happens because mechanical stress disrupts both the filler network and polymer-filler bonds [5].
Although network formation enhances static properties, its breakdown under dynamic conditions results in the Payne Effect.
The Payne Effect on Dynamic Properties
Once a strong filler network is formed, higher filler concentrations can trigger the Payne Effect, characterized by a significant drop in storage modulus (G’) as strain increases [5][7]. This non-linear viscoelastic behavior occurs because the physical bonds between filler particles break under cyclic strain, fragmenting the network into smaller clusters and reducing stiffness [10].
Liliane Bokobza, an independent researcher, explains this phenomenon:
The filler network that results from filler–filler interactions is well known to affect the dynamic viscoelastic behavior of the composite. It is responsible for the typical non-linear viscoelastic behavior of the storage modulus G′ called the ‘Payne effect’ [5].
The extent of the Payne Effect is influenced by the type of filler and its surface treatment. For example, silica tends to exhibit stronger filler-filler interactions due to hydrogen bonding, resulting in a more pronounced Payne Effect unless coupling agents like TESPT are used to improve polymer-filler compatibility [5][4]. Without coupling agents (such as Si69), filler networking dominates at low strains, leading to a higher initial modulus and a more noticeable Payne Effect. In contrast, coupling agents enhance filler-polymer adhesion and dispersion, reducing the Payne Effect [5]. The drop in storage modulus (G’) follows a power law, with the exponent reflecting the fractal structure of filler clusters and the resulting network [7]. This relationship provides engineers with a quantitative method to evaluate network quality and predict dynamic performance.
Disclaimer: This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
sbb-itb-aa4586a
Methods for Achieving Uniform Filler Dispersion
Ensuring fillers are evenly distributed in elastomers is no easy feat. It demands both mechanical precision and chemical finesse. Without these, fillers can clump together, creating weak points that compromise performance. By combining the right techniques, a consistent filler network can be established, which is crucial for boosting elastomer functionality.
Mechanical Mixing and Shear Force Application
The mechanical approach to dispersion hinges on shear strain, which combines the rotor speed (shear rate) and the time materials spend in the mixer. Both batch and continuous mixers apply intense shear forces to break apart filler clumps. However, achieving stable dispersion requires carefully balancing key factors like rotor speed, mixing time, and temperature. The final size of the dispersed filler depends on its type and the size of its primary particles.
Timing and temperature management are critical here. A dual-cycle temperature method works best – first, oil is added after partial dispersion, followed by a heating-cooling cycle to refine the process. Michael C. H. Lee from General Motors Research Laboratories explains:
The heating cycle is for decreasing the microvoid concentration and for enhancing polymer–filler adhesion. The cooling cycle is for improving the degree of mixing of the filler. [12]
Advanced tools like ultra-small angle X-ray scattering (USAXS) have shown that single-stage mixing can achieve excellent nano-dispersion. This is particularly effective for fillers like carbon-coated silica and carbon black in polybutadiene compounds.
Chemical Surface Treatments for Better Compatibility
While mechanical methods physically break down filler clusters, chemical treatments focus on improving how fillers interact with the elastomer. These treatments modify filler surfaces to better match the polarity of the elastomer, enhancing compatibility. For instance, bifunctional coupling agents like bis(3-triethoxysilylpropyl) tetrasulfide (TESPT or Si69) serve as molecular connectors. Their ethoxy groups bond with silica, while their tetrasulfane groups react with hydrocarbon rubber during vulcanization. This process deactivates silica’s silanol groups, reducing clumping and improving dispersion.
Silica particles created through pyrogenic methods typically measure between 5 and 50 nanometers, with aggregates forming rigid structures around 100 nanometers or larger. These aggregates resist mechanical breakdown, making chemical surface treatments indispensable. Liliane Bokobza, an independent researcher, highlights their importance:
The use of silane coupling agents in combination with silica in nonpolar polymers decreases the filler–filler interactions and simultaneously increases the polymer–filler interactions. [11]
Another effective technique is polymer grafting, where polymer chains are bonded to filler surfaces through radical or photocatalytic polymerization. This enhances the "wetting" of the filler by the elastomer matrix and provides greater control over the compound’s microstructure. Additional treatments, like carbon-coating silica, help counteract surface charge issues caused by silanols, further improving the filler network. These modifications can be done during mixing or by using pre-treated fillers, simplifying the process.
Allan Chemical Corporation supplies a variety of technical-grade chemicals and offers sourcing expertise to help achieve optimal filler dispersion.
Disclaimer: This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Conclusion
The way fillers are dispersed plays a crucial role in determining the performance of elastomers. When fillers are distributed evenly, they help prevent cracks from forming and extend the material’s service life [5]. On the other hand, uneven dispersion leads to weak points in the material, which can cause it to fail prematurely.
These performance benefits are directly tied to improved processing methods and material interactions. Take tire manufacturing as an example: better filler dispersion minimizes viscoelastic losses, reduces rolling resistance, and enhances fuel efficiency [1]. The same principle applies across various applications, whether it’s in tires, industrial seals, or vibration dampeners.
Achieving uniform filler dispersion depends on a combination of particle physics and chemical interactions. Techniques like mechanical mixing and chemical surface treatments are essential to create consistent dispersion. Industries that excel in these areas can fine-tune their mixing processes, choose effective coupling agents, and monitor critical parameters to produce elastomers with reliable and predictable properties.
For nanoscale fillers, precise control becomes even more critical. These materials provide excellent reinforcement at lower loadings but demand careful management of processing conditions and surface chemistry [5]. By addressing these factors, manufacturers can ensure their compounds deliver consistent performance – proving that uniform dispersion is key to better results.
Allan Chemical Corporation offers technical-grade chemicals and sourcing expertise to support manufacturers in achieving optimal filler dispersion for their elastomer formulations.
Disclaimer: This content is for informational purposes only. Always consult official regulations and qualified professionals when making sourcing or formulation decisions.
FAQs
What are the main advantages of achieving uniform filler dispersion in elastomers?
Achieving even filler dispersion in elastomers brings several important advantages. It boosts the material’s elastic modulus and strength, enhancing durability and enabling the elastomer to handle different types of stress more effectively. This also results in better toughness, fatigue resistance, and abrasion resistance, ensuring the material maintains its performance over time.
Additionally, uniform dispersion ensures more predictable dynamic behavior, such as minimizing energy loss (lower Payne effect). This leads to greater efficiency and dependability in various applications, making consistent filler dispersion a critical factor in improving the performance and lifespan of elastomer-based products.
What’s the difference between mechanical mixing and chemical surface treatments for dispersing fillers in elastomers?
Mechanical mixing uses techniques like fluidized beds or ultrasonic methods to physically break apart clusters of filler particles and spread them evenly throughout the elastomer matrix. This process depends on mechanical energy to achieve proper dispersion.
On the other hand, chemical surface treatments involve applying coupling agents or dispersing aids to alter the surface of filler particles. These treatments enhance the compatibility between the filler and the elastomer, minimizing particle clumping and improving dispersion without relying solely on mechanical force. Depending on the application and the material properties needed, these methods can be applied individually or in combination.
What is the percolation threshold, and how does it affect elastomer reinforcement?
The percolation threshold marks the critical concentration point where filler particles within an elastomer form a continuous network throughout the material. Once this network is established, the elastomer experiences a noticeable boost in mechanical properties, such as increased stiffness and strength, which is reflected in a sharp rise in its modulus.
This effect happens because the interconnected filler particles enhance load transfer and limit the movement of the polymer matrix, providing stronger reinforcement. Grasping the concept of the percolation threshold is key to fine-tuning filler content, ensuring the right mix of performance and ease of processing for elastomer-based applications.





Comments are closed