Epoxy resin wastewater is a byproduct of manufacturing processes that involves hazardous chemicals like Epichlorohydrin (CAS No. 106-89-8) and phenolic compounds. This wastewater is challenging to treat due to its high salinity, toxic organic compounds, and refractory nature, which resists conventional biological treatment. Effective treatment is crucial to meet strict EPA regulations and prevent harm to aquatic ecosystems.
Key Treatment Methods:
- Physical Methods:
- Membrane Filtration: Removes suspended solids and dissolved salts using reverse osmosis or nanofiltration.
- Air Flotation: Uses micro-bubbles to separate oils and lightweight particles.
- Chemical Methods:
- Fenton Oxidation: Breaks down organic pollutants using hydrogen peroxide and iron catalysts.
- Neutralization/Coagulation: Adjusts pH and removes colloidal particles.
- Biological Methods:
- Anaerobic Reactors (EGSB): Degrades organics and generates biogas in oxygen-free conditions.
- Aerobic Treatment (A/O Process): Oxidizes remaining pollutants and removes nitrogen.
- Advanced Systems:
- Thermal Processes: Evaporation and crystallization recover materials like ammonium bromide.
- Resin Adsorption: Captures pollutants for potential reuse.
Challenges include high energy costs for thermal methods, sludge disposal from chemical treatments, and the need for pre-treatment to improve biodegradability. Combining methods – like chemical oxidation followed by biological processes – ensures better pollutant removal while meeting regulatory limits.
For epoxy resin wastewater, a tailored multi-step approach is essential to break down its complex structure and comply with EPA standards.
HUBER Chemical-physical waste water treatment
sbb-itb-aa4586a
Physical Treatment Methods
Physical treatment methods focus on mechanically separating contaminants rather than relying on chemical or biological processes. These techniques are often the first step in wastewater treatment, used to remove settleable solids, suspended particles, oils, and fats before the water moves on to more advanced stages. In epoxy resin production, physical treatment reduces the organic load in the wastewater and prepares it for subsequent chemical or biological treatments. By efficiently removing these materials, physical methods improve the performance of downstream processes.
The choice of a specific physical treatment method depends on factors like particle size, liquid density, concentration of solids, and the required water purity. Larger, more advanced facilities often achieve better efficiency and higher-quality effluent. Membrane separation processes, such as reverse osmosis and nanofiltration, are particularly effective, offering recovery rates between 80% and 98%[2]. These methods are especially valuable for plants aiming to reclaim clean water or minimize discharge volumes.
Vortex-Concave Air Flotation
Vortex-Concave Air Flotation (VCAF) uses a high-speed impeller to create micro-bubbles that attach to suspended solids and emulsified oils. These contaminants are lifted to the surface, forming a froth that can be skimmed off. This technique is highly effective for removing oils, fats, and lightweight suspended materials that don’t easily settle under gravity. Dissolved Air Flotation (DAF) systems, a standard technology in this category, offer capacities ranging from 1 kiloliter per hour (about 264 gallons per hour) to 1,000 kiloliters per hour (about 264,000 gallons per hour)[2], making them suitable for both small and large epoxy resin production facilities.
Industrial wastewater can contain grease and oil concentrations between 0 and 2,000 parts per million (ppm)[2]. Air flotation excels in these conditions by targeting such contaminants. For particles too fine for standard separation, chemical flocculants can be added to increase particle size and improve removal efficiency. Many facilities combine physical and chemical pretreatment to handle the complexities of epoxy resin wastewater effectively.
After flotation, membrane technologies offer further refinement of the effluent.
Membrane Filtration and Distillation
Membrane filtration involves several pressure-driven processes – microfiltration, ultrafiltration, nanofiltration, and reverse osmosis – each designed to remove increasingly smaller contaminants. Ultrafiltration is effective at removing macromolecules and colloidal particles, while reverse osmosis targets dissolved salts, which are often present in epoxy resin wastewater due to neutralization reactions. To ensure efficiency and prevent scaling, membrane systems are customized to meet specific operational needs.
To extend membrane life and reduce scaling, facilities often add polymeric materials before the pre-filtration stage. Distillation can also play a key role, enabling the recovery and reuse of organic solvents like N,N-dimethylformamide (DMF). This not only cuts down on raw material costs but also reduces the volume of wastewater generated.
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Chemical Treatment Methods
Chemical treatment methods rely on specialized agents to oxidize or neutralize pollutants that can’t be removed through physical processes alone. These techniques are particularly effective against stubborn organic pollutants, transforming them into forms that are easier to handle in subsequent treatment stages. Often, chemical processes act as a bridge between physical pretreatment and biological treatment, converting hard-to-degrade contaminants into substances that microorganisms can process more efficiently.
The success of chemical treatments hinges on several factors, including pH levels, temperature, the amount of reagent used, and the duration of contact. Operators must carefully balance these variables to achieve effective results while managing costs, ensuring worker safety, and addressing sludge disposal challenges. Handling strong oxidizing agents, acids, and solvents requires strict safety measures to prevent accidents and secondary pollution. The use of these chemicals, while effective, can sometimes lead to additional environmental concerns due to the complexity of managing degradation products and potential pollution risks [4].
Iron-Carbon Micro-Electrolysis
This method highlights the role of micro-electrolysis in preparing wastewater for biological treatment. Iron-carbon micro-electrolysis relies on tiny electrochemical cells to break down complex organic compounds in wastewater. It uses zero-valent iron (ZVI) as the anode and carbon as the cathode, generating reactive species that degrade high-molecular-weight organics. Adding chelating agents like EDTA or citric acid can expand the effective pH range, making this technique more versatile. As a pretreatment step, it significantly improves the biochemical oxygen demand to chemical oxygen demand ratio (BOD/COD), making the wastewater more compatible with biological processes [5].
In these systems, iron reacts with hydrogen peroxide or dissolved oxygen to produce ferrous ions (Fe²⁺), which drive oxidation reactions. Facilities typically operate under acidic to neutral conditions, benefiting from the combined effects of electrochemical reduction and chemical oxidation [3].
Fenton Oxidation
Fenton oxidation is a powerful process that combines Fe²⁺ and hydrogen peroxide (H₂O₂) to generate hydroxyl radicals (·OH) – some of the most effective oxidizing agents available. These radicals aggressively break down organic pollutants into carbon dioxide and water. The classic Fenton process works best within a specific pH range of 2.8 to 3.5, where the reaction achieves maximum efficiency [3].
Innovations in Fenton technology include the photo-Fenton process, which uses ultraviolet or visible light to regenerate Fe²⁺, reducing sludge production. Experimental setups with ZVI nanoparticles in photo-Fenton membranes have achieved impressive results, such as degrading 99% of organic dyes in just 90 minutes [3]. Heterogeneous Fenton systems, on the other hand, utilize solid iron-based catalysts like magnetite (Fe₃O₄) or hematite (α-Fe₂O₃), which can be recovered and reused, making them a practical choice for industrial applications [3].
| Feature | Homogeneous Fenton | Heterogeneous Fenton | Photo-Fenton |
|---|---|---|---|
| Catalyst State | Dissolved iron ions (Fe²⁺) | Solid iron materials | Iron catalysts + UV/light |
| pH Range | Narrow (2.8–3.5) | Broader applicability | Improved for various levels |
| Sludge Production | High iron sludge yield | Reduced sludge | Minimal sludge |
| Catalyst Recovery | Difficult | Easier (solid form) | Often immobilized |
Neutralization and Coagulation
Neutralization adjusts wastewater pH to levels that are either safe for discharge or optimal for further treatment. For example, acidic or alkaline streams from epoxy resin production often require balancing with sulfuric acid or sodium hydroxide. Maintaining proper pH is critical because many chemical and biological processes only function within specific ranges. At pH levels below 2.5, oxidation reactions slow due to stable complex formation, while at pH levels between 3 and 7, iron precipitation can hinder efficiency unless chelating agents are used [3].
Following neutralization, coagulation involves adding chemicals like aluminum sulfate or ferric chloride to destabilize suspended particles and colloids. These coagulants neutralize the charges that keep particles dispersed, allowing them to form larger aggregates, or flocs, which can be removed through settling or filtration. However, the resulting sludge must be managed carefully, as it contains concentrated pollutants that require proper disposal or additional treatment. Together, neutralization and coagulation integrate seamlessly with physical and biological methods, forming a well-rounded wastewater management approach.
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Biological Treatment Methods
Biological treatment relies on microorganisms to break down organic pollutants in epoxy resin wastewater. Unlike chemical methods that use oxidizing agents, biological approaches employ microbial communities to convert complex polymers into simpler compounds through biochemical reactions. While this method is more environmentally friendly – especially for high-strength wastewater – the rigid ether-based polymers in epoxy resin and the sensitive Carbon-to-Nitrogen (C/N) balance (ideally 20–30) present significant challenges for conventional biodegradation systems [6].
For instance, epoxy resin wastewater is particularly difficult to treat due to its polymers’ predominantly ether-based backbone. Unlike other plastics with hydrolyzable ester or amide bonds, these polymers resist standard biological breakdown [7].
"Epoxy polymers, characterized by their strong cross-linking and predominantly ether-based backbone, exhibit a highly rigid and durable structure, placing them within category." – Leon Klose, Institute of Technical Biocatalysis [7]
In one study, researchers isolated Rhodococcus rhodochrous and Ochrobactrum anthropi from soil near an epoxy plant. Together, these microorganisms successfully degraded Bisphenol A and other resin components, but neither could grow independently. Concentrations above 1.2 wt.% proved toxic to the microbes, and using epoxy resin as the sole carbon source resulted in a growth rate that doubled every 48 hours [9]. These limitations highlight the need for pre-treatment steps to break down complex structures, making them more accessible for microbial degradation. Specialized configurations in anaerobic and aerobic systems are often required to address these challenges.
Anaerobic Treatment with EGSB Reactors
To overcome these obstacles, anaerobic and aerobic reactors are frequently combined to enhance biodegradation. Expanded Granular Sludge Bed (EGSB) reactors are a prominent anaerobic technology designed for high-strength wastewater with elevated Chemical Oxygen Demand (COD) levels. These systems operate without oxygen, relying on specialized anaerobic microorganisms to break down organic matter through a multi-stage process, producing biogas as a byproduct. In an EGSB reactor, wastewater flows upward through a bed of granular sludge, ensuring efficient contact between pollutants and microorganisms.
The anaerobic process unfolds in four stages:
- Hydrolysis: Extracellular enzymes break down complex polymers into simpler molecules like glucose, fatty acids, and amino acids [6].
- Acidogenesis: Acidogenic bacteria convert these molecules into volatile fatty acids (VFAs), alcohols, and ketones [6].
- Acetogenesis: Acetogenic bacteria transform VFAs and alcohols into acetate, carbon dioxide, and hydrogen gas. This step depends on syntrophic interactions with methanogens to keep hydrogen partial pressure below 10⁻⁴ atm [6].
- Methanogenesis: Methanogens, which thrive in strictly anaerobic conditions, convert acetate and hydrogen into methane and carbon dioxide [6].
Temperature plays a crucial role in EGSB reactor performance. Mesophilic digestion, operating at 95–104°F (35–40°C), offers stability and lower energy demands but requires longer retention times. Thermophilic digestion, at 131–140°F (55–60°C), accelerates degradation and boosts biogas output but demands more energy and is less stable. Biogas combustion typically achieves electricity production efficiencies of 35% to 42% [6].
Maintaining the right conditions is essential. Methanogens prefer a neutral pH around 7.0, while hydrolytic and acidogenic bacteria function better in slightly acidic environments (pH 5.5–6.5) [6]. Accumulation of intermediates like ammonia or long-chain fatty acids can inhibit methanogens, potentially causing system failure. Efficient hydrogen transfer between microbial communities is another critical factor for success [6].
| Digester Type | Total Solids (%) | HRT (days) | Advantages | Disadvantages |
|---|---|---|---|---|
| Covered Lagoon | 0.5–2 | 30–40 | Low operating cost | Ineffective in cold climates |
| Complete Mix | 3–10 | 10–25 | Adapts to feedstock changes | High energy demand for mixing |
| Plug Flow | 10–15 | 10–25 | Economical to operate | Solids may settle at the bottom |
| Fixed Film | 1–5 | 2–6 | Compact; low HRT | Risk of media clogging with solids |
Aerobic Treatment with Two-Stage AO Process
Aerobic treatment complements anaerobic methods by further oxidizing residual organics and efficiently removing nitrogen. The Anoxic/Oxic (AO) process alternates between oxygen-free and oxygen-rich environments to achieve thorough degradation. During the anoxic stage, microorganisms convert nitrates into nitrogen gas through denitrification. The subsequent oxic stage introduces aeration to oxidize remaining organic pollutants into carbon dioxide, water, and microbial biomass.
Typically employed as a secondary treatment after anaerobic processes like EGSB reactors, the AO process ensures compliance with regulatory discharge standards. While the oxic stage requires significant energy for aeration, it produces a higher-quality effluent.
Optimized Anaerobic-Anoxic-Oxic (A2/O) systems have shown that a Sludge Retention Time (SRT) of around 15 days effectively reduces the toxicity of complex wastewater [10]. Hydraulic retention times (HRT) of 2.8 hours for the anaerobic/anoxic phase and 6.9 hours for the aerobic phase have also proven effective for treating epoxy resin wastewater [10]. In these systems, aerobes, nitrifiers, and denitrifiers work together to break down various pollutants.
Combining biological processes with pre-treatment methods like Advanced Oxidation Processes (AOPs) or electrochemical techniques can improve performance by converting complex epoxy structures into more biodegradable forms [8]. Additionally, incorporating conductive materials such as carbon or nano-additives may enhance electron transfer between microbial species, addressing some metabolic limitations in both anaerobic and aerobic systems [8].
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Advanced and Combined Treatment Processes
When single-method approaches aren’t enough, advanced and combined treatment processes step in to tackle the challenges of epoxy resin wastewater treatment. By combining multiple technologies, these methods achieve better pollutant removal, recover useful materials, and comply with stricter environmental regulations. Unlike standalone techniques, these systems create a synergy where one process enhances the effectiveness of another.
For example, the debromination of brominated epoxy resin (BER) releases hydrobromic acid (HBr), which naturally lowers the pH and speeds up the degradation of N,N-dimethylformamide (DMF) in the same wastewater stream [1]. This interconnected process allows facilities to handle multiple pollutants at once while cutting costs. Beyond pollutant removal, advanced methods also focus on recovering resources from waste, complementing earlier physical, chemical, and biological treatments.
Evaporation, Concentration, and Crystallization
Thermal separation techniques take single-method approaches to the next level by enhancing resource recovery. These heat-based processes transform complex epoxy wastewater into distinct phases that can be recovered and reused.
- Evaporation removes water by applying heat, leaving behind concentrated dissolved solids and organic compounds.
- Concentration reduces the volume further, making subsequent treatments more efficient.
- Crystallization isolates specific compounds as solid crystals, which can then be repurposed.
These methods break down chemical bonds, producing gases, high-boiling liquids, and solids [4]. For instance, using a subcritical DMF wastewater (SDW) process supported by the National Natural Science Foundation of China, researchers achieved a 98.57% debromination ratio for BER and a 99.59% degradation ratio for DMF under optimal conditions of 572°F (300°C) for 60 minutes [1]. This process also recovered phenolic substances with 86.61% purity and crystallized ammonium bromide (NH₄Br) as a reusable product [1].
"The SDW process proposed in this study is an efficient technology for the synergistic treatment of the BER waste and DMF wastewater." – Fu-Rong Xiu, Professor/Researcher [1]
However, these processes require substantial energy. Operating temperatures between 572°F and 1,112°F (300°C to 600°C) lead to high electricity or fuel costs, making them cost-effective mainly for facilities with on-site energy recovery systems or access to affordable heat sources [1][4]. Adding catalysts like molten zinc chloride (ZnCl₂) can speed up thermal degradation while preserving the quality of recovered fibers. For example, carbon fibers retained 95% of their original tensile strength after treatment [4].
Resin Adsorption
Resin adsorption uses materials like activated carbon, zeolites, or metal-organic frameworks (MOFs) to capture pollutants from wastewater. Unlike chemical oxidation, which destroys contaminants, adsorption binds them to a solid surface, allowing for potential recovery and reuse. This method is highly effective for removing substances like benzene, phenol, amines, carboxylic acids, and sulfonic acids from epoxy wastewater streams [11].
"Adsorption resin… can realize the resource of wastewater. For benzene, phenol, amine, carboxylic acid and sulfonic acid… the biggest feature of this technology is also in wastewater treatment, also realizes the recovery of organic toxicants." – Lanshen New Material Technology Co. Ltd. [11]
Adsorption systems often achieve removal rates over 99% for biological, organic, and inorganic pollutants [12]. MOFs, in particular, offer excellent selectivity and capacity for targeting heavy metals and persistent organic compounds in complex wastewater [12]. The adsorbent materials can be regenerated through thermal or chemical methods, extending their lifespan and lowering disposal costs. To enhance performance and avoid premature saturation, facilities should use pre-treatment steps like physical separation or coagulation to remove bulk hydrocarbons and suspended solids before adsorption [12]. Combined with earlier treatment stages, these advanced methods make wastewater management more efficient while recovering valuable resources.
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Comparison of Treatment Methods
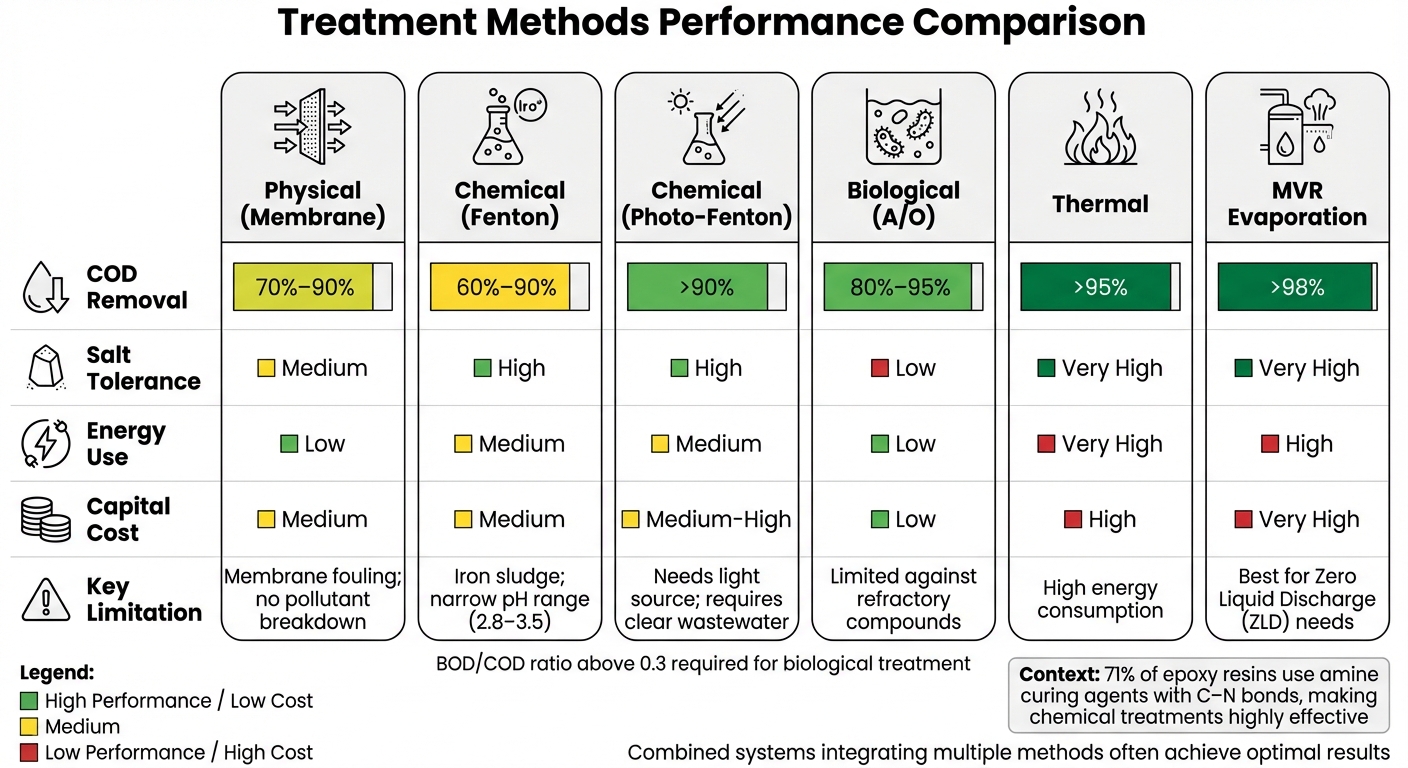
Epoxy Resin Wastewater Treatment Methods Comparison: Efficiency, Costs, and Limitations
Evaluation Criteria
Selecting the right treatment method involves evaluating several key metrics. These metrics address the challenges associated with physical, chemical, and biological treatment methods. One critical measure is COD (Chemical Oxygen Demand) removal efficiency, which reflects how well a method reduces organic pollutants – a key requirement for meeting NPDES permit standards under the Clean Water Act. Another consideration is energy consumption, typically measured in kWh per gallon treated, as it directly impacts long-term operational costs. Sludge production is also significant, as it affects disposal costs and compliance with RCRA regulations, particularly for chemical treatments that generate hazardous waste. Additionally, salt tolerance becomes crucial when chloride levels exceed 1%, potentially inhibiting biological processes. Finally, capital and operating costs must be carefully considered to ensure accurate budgeting.
"The homogeneous Fenton process has three main challenges: the narrow optimal operating pH range of 2.8–3.5, the generation of large amounts of iron sludge and the difficulty in catalyst recovery." – MDPI Membranes [3]
Performance Comparison Table
Each treatment method offers its own set of strengths and weaknesses. Physical methods, such as membrane filtration, excel at separating contaminants with low energy consumption but cannot break down pollutants and are prone to fouling. Chemical methods like Fenton oxidation achieve 60%–90% COD removal but produce iron sludge and require a specific pH range of 2.8–3.5 [3]. Biological treatments, such as two-stage anaerobic/oxic (A/O) processes, are cost-effective but may struggle with refractory compounds often found in epoxy resin wastewater [3]. Meanwhile, thermal degradation achieves over 95% removal efficiency but comes with high energy demands, requiring operating temperatures between 572°F and 1,112°F (300°C to 600°C) [4]. The table below summarizes these metrics for an easier comparison.
| Method | COD Removal | Salt Tolerance | Energy Use | Capital Cost | Key Limitation |
|---|---|---|---|---|---|
| Physical (Membrane) | 70%–90% | Medium | Low | Medium | Membrane fouling; no pollutant breakdown [3] |
| Chemical (Fenton) | 60%–90% | High | Medium | Medium | Iron sludge; narrow pH range (2.8–3.5) [3] |
| Chemical (Photo-Fenton) | >90% | High | Medium | Medium-High | Needs light source; requires clear wastewater [3] |
| Biological (A/O) | 80%–95% | Low | Low | Low | Limited against refractory compounds [3] |
| Thermal | >95% | Very High | Very High | High | High energy consumption [4] |
| MVR Evaporation | >98% | Very High | High | Very High | Best for Zero Liquid Discharge (ZLD) needs |
For municipal dischargers, maintaining a BOD/COD ratio above 0.3 is essential to avoid the need for additional chemical pretreatment. If the ratio drops below 0.3, chemical pretreatment becomes necessary to improve the biodegradability of the wastewater before biological treatment. Interestingly, about 71% of epoxy resins use amine curing agents with C–N bonds, which are particularly responsive to oxidizing acids – making chemical treatments highly effective for such formulations [4].
The best treatment approach depends on the specific wastewater composition, regulatory requirements, and available resources. Many facilities opt for combined systems, which integrate multiple methods to capitalize on their individual strengths.
This content is for informational purposes only. Always consult official regulations and qualified professionals before making decisions related to wastewater treatment.
Combined Treatment Systems
Many facilities achieve optimal results by combining several treatment stages. This usually starts with physically removing suspended solids and oils, followed by chemical oxidation to break down complex resin structures, and ends with biological degradation to handle the remaining organic matter [2]. This step-by-step approach is necessary to tackle the unique challenge of epoxy wastewater – its stable, three-dimensional cross-linked structure makes these resins both infusible and insoluble [4]. To address this, precise chemical pretreatment is crucial for breaking down these tough bonds before biological processes can effectively take over.
"As noted by ScienceDirect, the curing reaction creates a robust, irreversible structure." – ScienceDirect [4]
Chemical pretreatment plays a key role in these integrated systems, targeting the C–N and C–O bonds within epoxy’s cross-linked framework. Since about 71% of epoxy resins are cured using amine agents [4], breaking these bonds transforms infusible materials into biodegradable fragments. This chemical step acts as a bridge between the initial physical separation and the final biological degradation, making the overall treatment more efficient. For instance, research by Wu et al. showed that molten ZnCl₂ at 680°F (360°C) could achieve complete degradation in just 80 minutes, while preserving 95% of carbon fiber tensile strength [4].
Advanced oxidation techniques further enhance these systems by integrating multiple treatment stages. One example is photo-Fenton–membrane technology, which combines oxidation with physical separation. In this method, the photo-Fenton reaction produces hydroxyl radicals (•OH) to break down complex organic chains, while a membrane removes the resulting fragments and suspended solids. Lee et al. developed a photo-Fenton–active membrane by embedding polypyrrole (PPy) and zero-valent iron (ZVI) nanoparticles onto a polyvinylidene fluoride (PVDF) substrate. Using a 10 mmol/L H₂O₂ concentration, this system degraded 99% of methylene blue in 90 minutes. Additionally, it featured a self-cleaning effect that degraded foulants on the membrane surface, extending its lifespan and maintaining high filtration efficiency [3].
Physical pretreatment methods, such as Dissolved Air Flotation (DAF), protect downstream biological processes from sudden surges in contaminants and clogging. Many advanced systems also incorporate membrane bioreactors (MBRs), which combine biological treatment with ultrafiltration. This setup effectively removes fine particles and dissolved contaminants that traditional systems might overlook [2].
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Conclusion
Treating epoxy resin wastewater requires a carefully designed, multi-step approach. As outlined earlier, combining physical, chemical, and biological methods is essential to address the complex nature of these contaminants. Physical processes, like membrane filtration, effectively remove suspended solids and oils. Chemical treatments, such as Fenton oxidation, target the breakdown of tough cross-linked structures. Once chemical pretreatment improves biodegradability, biological systems like EGSB reactors and two-stage AO processes handle the remaining organic matter.
Integrated treatment systems take this a step further by combining these methods for maximum efficiency. The best systems are tailored to the specific characteristics of the wastewater, ensuring all contaminants are addressed. Key considerations include wastewater composition, pH requirements, and sludge management. For instance, homogeneous Fenton processes demand a narrow pH range and generate significant sludge, whereas heterogeneous photo-Fenton methods offer broader pH flexibility and produce less waste [3]. Choosing low-sludge methods can significantly reduce disposal costs over time.
Another critical factor is the use of high-quality, technical-grade chemicals to ensure treatment effectiveness and regulatory compliance. These chemicals must meet stringent standards. With over 40 years of experience, Allan Chemical Corporation supplies technical-grade and compendial-grade chemicals (USP, FCC, ACS, NF) designed for wastewater treatment. Their reliable delivery, competitive pricing, and strong supplier network support facilities in maintaining efficient operations while managing costs.
Ultimately, achieving efficient and compliant wastewater treatment depends on using premium chemicals and designing a customized, integrated system. Assess your wastewater composition and operational needs, then consult experts to develop a solution that meets EPA requirements and ensures long-term success.
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
FAQs
What challenges are involved in treating wastewater from epoxy resin production?
Treating wastewater from epoxy resin production presents unique challenges due to the presence of high-strength organic polymers, uncured monomers, solvents, and volatile organic compounds (VOCs). These substances are tough to break down using conventional biological treatments, often resulting in elevated chemical oxygen demand (COD) levels and toxicity. To address this, pretreatment steps such as neutralization, oxidation, and equalization are typically necessary before moving on to further processing stages.
Another hurdle is the variability in wastewater flow rates and contaminant concentrations, which can overwhelm fixed-capacity treatment systems. A successful approach often requires multiple treatment steps. These can include physical separation methods like oil-water separation, chemical oxidation, and advanced processes such as ion exchange or photocatalysis. However, each technique comes with its own set of challenges – adsorbents can become saturated, and oxidation may produce unwanted by-products – making it essential to strike a balance between efficiency and cost management.
Allan Chemical Corporation plays a key role in these efforts by offering a range of specialty treatment chemicals. This includes advanced oxidants, flocculants, and adsorption media, all designed to align with U.S. regulatory standards while improving the performance of multi-stage treatment systems.
Why is combining treatment methods effective for managing epoxy resin wastewater?
Combining different treatment methods, often referred to as a treatment train, works well because each process focuses on specific contaminants while addressing the gaps left by others. For example, physical methods like filtration or oil-water separation are excellent for removing solids and non-aqueous liquids. Chemical processes, such as oxidation or ion exchange, target dissolved organics and heavy metals. Finally, biological treatments handle leftover compounds, breaking them down into harmless byproducts. Together, these methods tackle a broader range of pollutants and deliver higher overall efficiency.
This combination also boosts performance. For instance, using a biological reactor alongside an electrochemical system can improve contaminant removal and lower energy consumption. The biological stage creates stable conditions, which makes the chemical step more effective. Allan Chemical Corporation plays a key role in supporting these systems by supplying specialty reagents like oxidants, ion-exchange resins, and biocatalyst nutrients. These products help manufacturers meet U.S. regulatory standards while keeping costs and operations efficient.
Why is chemical pretreatment important for treating epoxy resin wastewater?
Chemical pretreatment is essential for managing epoxy resin wastewater, as it effectively breaks down the high levels of organic resin compounds present. This step helps to lower chemical oxygen demand (COD), total nitrogen, and ammonia concentrations, reducing the toxicity of the wastewater.
By tackling these pollutants early in the process, chemical pretreatment prepares the wastewater for subsequent biological or physical treatment methods, boosting the efficiency of these treatments and promoting safer environmental practices.





Comments are closed