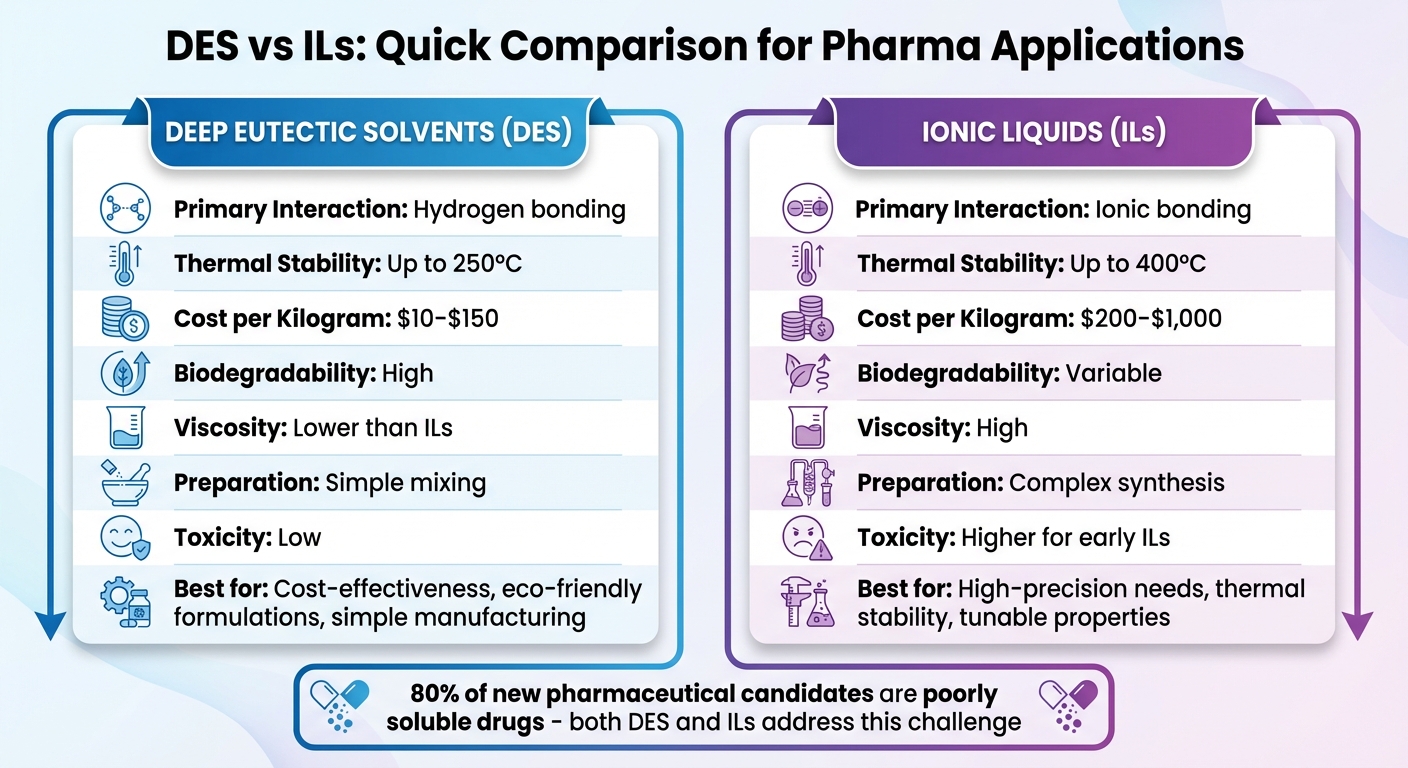
Deep Eutectic Solvents (DES) and Ionic Liquids (ILs) are advanced solvent systems revolutionizing drug formulation. Both improve solubility and stability for poorly soluble drugs, which make up 80% of new pharmaceutical candidates. DES are mixtures of a hydrogen bond donor (HBD) and acceptor (HBA), while ILs are molten salts with ionic bonds. DES are cheaper to produce, more biodegradable, and eco-friendly, while ILs offer higher thermal stability and tunable properties. DES are favored for their simplicity and safety, while ILs excel in precision and versatility, especially for complex drug systems. Each has distinct benefits and limitations, making the choice highly application-specific.
Quick Comparison:
| Feature | Deep Eutectic Solvents (DES) | Ionic Liquids (ILs) |
|---|---|---|
| Primary Interaction | Hydrogen bonding | Ionic bonding |
| Thermal Stability | Up to 250°C | Up to 400°C |
| Cost per Kilogram | $10–$150 | $200–$1,000 |
| Biodegradability | High | Variable |
| Viscosity | Lower than ILs | High |
| Preparation | Simple mixing | Complex synthesis |
| Toxicity | Low | Higher for early ILs |
Both systems address solubility issues and prevent polymorphism, but DES are often preferred for their affordability and safety, while ILs are used for specialized, high-precision needs.
Deep Eutectic Solvents vs Ionic Liquids: Property Comparison for Pharmaceutical Applications
TMS 13/01/22 ”The Chemistry behind the magic: Deep Eutectic Solvent as a still debated case study”
What Are Deep Eutectic Solvents?
Deep eutectic solvents (DES) are liquid mixtures made by combining a hydrogen bond acceptor (HBA) and a hydrogen bond donor (HBD) in specific molar ratios. This combination significantly lowers the melting point of the resulting mixture. For instance, when choline chloride (melting point: 302°C/572°F) is mixed with urea (melting point: 133°C/271°F) in a 1:2 ratio, the resulting DES has a freezing point as low as 12°C [9].
Unlike ionic liquids (ILs), which are salts made of discrete ions, DES rely on extensive hydrogen bonding networks. These interactions lead to charge delocalization and a reduction in lattice energy, which keeps the mixture in a liquid state at room temperature. Typically, quaternary ammonium salts like choline chloride serve as the HBA, while the HBD can be an amide, carboxylic acid, or alcohol.
DES are relatively simple and inexpensive to produce, requiring only basic processes like heating, grinding, or stirring. Choline chloride, a common ingredient in DES, is widely manufactured as an animal feed additive, making it readily available.
DES are often considered environmentally friendly due to their low vapor pressure, nonflammability, and biodegradability. In recent years, natural deep eutectic solvents (NADES) have gained attention. These are entirely bio-based, composed of primary metabolites, and are especially useful in pharmaceutical applications. Another emerging type is therapeutic deep eutectic solvents (TheDES), where the active pharmaceutical ingredient itself is a component of the mixture.
Properties of Deep Eutectic Solvents
DES are highly adaptable, with properties such as viscosity, density, and polarity that can be fine-tuned by altering their components or molar ratios. This versatility allows formulators to create solvents tailored to specific pharmaceutical requirements. Common types of DES include:
- Type I: Quaternary ammonium salt + metal chloride
- Type II: Quaternary ammonium salt + metal chloride hydrate
- Type III: Quaternary ammonium salt + hydrogen bond donor
- Type IV: Metal chloride hydrate + hydrogen bond donor
- Type V: Mixtures made entirely of non-ionic components
One challenge with DES is their high viscosity, which can make handling and processing difficult. However, adding small amounts of water – typically 30–40% by weight – can significantly reduce viscosity, sometimes by as much as 50-fold, while maintaining the hydrogen bonding network [12]. This adjustment improves flow properties without compromising the solvent’s key characteristics.
DES also exhibit excellent thermal stability across a wide temperature range and tend to biodegrade more readily than traditional ionic liquids, especially those containing imidazolium or pyridinium cations, which are less biodegradable. NADES, in particular, are valued for their low toxicity, as they are made from naturally occurring metabolites.
These customizable properties make DES particularly useful for addressing challenges in drug solubility and stability.
Pharmaceutical Uses of Deep Eutectic Solvents
DES offer several advantages in pharmaceutical applications, particularly in improving drug formulations. One of their primary uses is enhancing the solubility of drugs that are poorly soluble in water. Research by Morrison et al. found that DES can increase drug solubility by factors ranging from 5 to 22,000 compared to water [11]. This is especially important as 40% of marketed oral drugs have solubility below 100 μg/mL, and 80% of new drug candidates fall into low-solubility categories (BCS Class II or IV).
In addition to improving solubility, DES can act as drug carriers and help prevent polymorphism, which is the formation of different crystal structures that can impact a drug’s effectiveness. For example, in September 2020, researchers Umerska and Tajber developed "anticrystal" formulations of ketoprofen by combining it with local anesthetics like benzocaine, procaine, and tetracaine. The ketoprofen-procaine system not only prevented crystallization but also enhanced ionization, leading to better drug delivery [1].
NADES have also demonstrated potential in stabilizing sensitive compounds. For instance, they have been shown to stabilize ranitidine·HCl (preventing polymorphism) and chloral hydrate (preventing dissociation) at concentrations up to 250 mg/mL. In February 2025, researchers developed a therapeutic DES called CAGE, made from choline bicarbonate and geranic acid in a 1:2 ratio. This formulation has been used to improve the delivery of antibodies and insulin in the gastrointestinal tract [2].
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
What Are Ionic Liquids?
Ionic liquids (ILs) are salts that stay in a liquid state at temperatures below 100°C (212°F) [4]. They are made entirely of ions, typically a large, irregular organic cation paired with a smaller anion [4]. This unique ionic makeup gives ILs properties that are particularly useful in pharmaceutical formulations.
The irregular shape and distributed charge of IL cations prevent the formation of a crystalline structure, keeping them liquid at lower temperatures [7]. Common cations include imidazolium, pyridinium, and quaternary ammonium groups, while anions range from simple halides to more complex ones like bis(trifluoromethylsulfonyl)imide (NTf₂) and hexafluorophosphate (PF₆). This structural diversity allows ILs to be tailored to improve drug solubility and stability, aligning with modern pharmaceutical needs.
One of the standout features of ionic liquids is their adaptability. By changing the combination of ions, their properties – such as viscosity, density, polarity, and solubility – can be fine-tuned [7]. This has earned them the nickname "designer solvents" [7]. With an estimated 10¹⁸ possible binary combinations, though only about 1,000 have been documented, the potential for creating ILs suited to specific applications is vast [7]. These ionic interactions set the stage for a closer look at their properties.
Another advantage of ILs is their negligible vapor pressure, making them non-volatile and safer than traditional volatile organic solvents [4]. They are also non-flammable and maintain high thermal stability across a broad temperature range of 300–400°C (572–752°F) [7]. These characteristics are especially appealing in pharmaceutical applications, where safety, stability, and precise control over solvent behavior are essential.
Properties of Ionic Liquids
The unique properties of ionic liquids make them especially valuable in pharmaceutical contexts. Thanks to their ionic conductivity, ILs can dissolve a wide range of compounds, including drugs with poor water solubility – a challenge faced by 40% to 70% of drugs currently in development [13]. By adjusting the ion pair, their solvation power can be optimized for specific drug molecules.
Their thermal stability ensures consistent performance during heating or sterilization processes. Additionally, their negligible vapor pressure enhances safety during manufacturing [4].
However, the toxicity and biocompatibility of ILs can vary with the choice of ions. Earlier generations of ILs, often based on imidazolium or pyridinium cations, raised concerns about toxicity. In contrast, newer "Bio-ILs" use naturally derived ions like choline, amino acids, or carboxylic acids to improve biocompatibility [2].
Although ILs generally have higher viscosities than conventional solvents, this can be adjusted by modifying their ion structure or adding small amounts of co-solvents. This flexibility allows them to be optimized for specific manufacturing processes or drug delivery methods.
Pharmaceutical Uses of Ionic Liquids
Ionic liquids address several challenges in pharmaceutical formulations. They improve drug solubility, stabilize sensitive ingredients, and can form Active Pharmaceutical Ingredient-Ionic Liquids (API-ILs), where the drug itself becomes part of the ionic structure. For example, in 2007, ranitidine docusate became the first reported API-IL, solving issues like polymorphism (variations in crystal structure that can affect drug safety and potency) and poor bioavailability [2]. Converting solid drugs into liquid ionic forms can eliminate these risks.
A study by Tay and Porter demonstrated this approach with lumefantrine, an antimalarial drug. By pairing lumefantrine with docusate to create an IL and incorporating it into Type IIIB lipid-based formulations, the system maintained drug supersaturation in tests and increased plasma exposure by 35 times in animal models compared to standard formulations [1].
Ionic liquids also enhance drug delivery through the skin and mucosal tissues. IL-in-oil microemulsions have been shown to improve transdermal delivery by reducing the skin’s barrier function [1]. This method is particularly beneficial for patients who struggle to swallow pills or require localized treatment.
In addition, ILs are being explored as antimicrobial agents. For instance, researchers Santos and Branco developed ciprofloxacin- and norfloxacin-based ILs that increased antimicrobial activity against Staphylococcus aureus by 11-fold and Klebsiella pneumoniae by 20-fold [1]. These examples highlight how ILs can not only improve solubility but also enhance therapeutic effects.
In drug synthesis, ionic liquids offer a greener alternative to traditional organic solvents. In 2000, researchers used [C₄C₁im][PF₆] to achieve a high-yield (90–94%) synthesis of pravadoline, a nonsteroidal anti-inflammatory drug (NSAID), replacing hazardous dimethylformamide (DMF) [13]. This substitution significantly reduces the environmental impact of pharmaceutical manufacturing, which often generates 25 to 100 kilograms of waste for every kilogram of product.
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
Property Comparison: DES vs. ILs
When examining Deep Eutectic Solvents (DES) and Ionic Liquids (ILs) for pharmaceutical applications, several key differences emerge. The most fundamental distinction lies in their molecular interactions: ILs are stabilized by ionic bonds, while DES rely on extensive hydrogen bonding to maintain their structure [15][2].
Both solvents exhibit higher viscosity compared to water, but DES have a notable advantage with their lower room-temperature viscosity. This makes them easier to pump, filter, and process, which can simplify manufacturing operations [14][15].
Another area of contrast is thermal stability. ILs are generally stable at temperatures ranging from 300–400°C (572–752°F), whereas DES typically remain stable only up to 150–250°C (302–482°F) [14]. This difference becomes critical for processes requiring high-temperature sterilization or synthesis. That said, both ILs and DES share two important safety features: negligible vapor pressure and nonflammability, reducing risks during production [15].
Cost is another significant factor. ILs, especially those of high purity, are priced between $200 and $1,000 per kilogram. In comparison, DES are far more economical, with costs ranging from $10 to $150 per kilogram [14]. This steep price gap is largely due to their preparation methods: ILs require complex, multi-step synthesis, while DES are produced through a simpler mixing process, making them more cost-effective for large-scale use.
For a quick overview, the table below highlights the key differences:
Property Comparison Table
| Property | Ionic Liquids (ILs) | Deep Eutectic Solvents (DES) |
|---|---|---|
| Primary Interaction | Ionic bonding [15] | Hydrogen bonding [15] |
| Thermal Stability | High (up to 300–400°C / 572–752°F) [14] | Moderate (up to 150–250°C / 302–482°F) [14] |
| Viscosity at Room Temp | Very high [14] | High, but lower than ILs [14] |
| Preparation Method | Complex multi-step synthesis [14] | Simple mixing of components [14] |
| Cost per Kilogram | $200–$1,000 [14] | $10–$150 [14] |
| Biodegradability | Often poor (1st/2nd generation) [15] | Generally excellent [14] |
| Vapor Pressure | Negligible [15] | Negligible [15] |
| Polarity Range | Broad (highly polar to non-polar) [15] | Typically hydrophilic and polar [15] |
Environmental considerations further set DES apart from ILs. Life cycle assessments reveal that DES formulations can reduce greenhouse gas emissions by 40–60% compared to ILs [14]. Moreover, certain imidazolium-based ILs are toxic to aquatic organisms at concentrations as low as 1–10 mg/L. In contrast, DES formulations require concentrations 10–100 times higher to produce similar toxic effects, highlighting their superior biodegradability [14]. This environmentally friendly profile positions DES as a promising choice for more sustainable pharmaceutical manufacturing.
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
sbb-itb-aa4586a
Pros and Cons in Drug Delivery
Deep Eutectic Solvents (DES) and Ionic Liquids (ILs) both bring unique benefits and challenges to drug delivery systems, offering solutions for solubility and stability issues. Selecting between these two often depends on balancing performance needs with practical concerns like cost, safety, and manufacturing complexity.
Solubility and Stability
One of the standout advantages of both ILs and DES is their ability to enhance solubility. For instance, ILs have shown the capability to increase plasma exposure by as much as 35 times in vivo [1]. DES formulations, on the other hand, can improve the solubility of poorly water-soluble drugs by up to 12 times [5]. Both approaches also help prevent polymorphic transitions, keeping Active Pharmaceutical Ingredients (APIs) stable in a liquid state [1][16][2]. While these benefits are significant, they lead to further considerations around toxicity and compatibility.
Biocompatibility and Safety
Biocompatibility is where ILs and DES diverge sharply. Traditional ILs, particularly early generations, are known for their high toxicity and limited biodegradability, which restricts their application in sensitive drug formulations [16][2]. As Professor Masahiro Goto from Kyushu University explained:
"Many ILs are inherently toxic, which is the main challenge toward developing IL-based drug formulations and drug delivery systems" [16].
However, newer Bio-ILs that use biocompatible ions, such as cholinium, have improved safety profiles, though they still require extensive testing [2][16]. In contrast, Natural Deep Eutectic Solvents (NaDES) – made from natural metabolites like sugars and amino acids – are generally safer for pharmaceutical use [2][6].
Practical Considerations: Cost and Manufacturing
When it comes to production, DES have a clear edge. Their preparation is straightforward, involving simple mixing or heating of components to produce a pure product without waste [5]. Alexandre Paiva from Universidade Nova de Lisboa highlighted this advantage:
"The preparation of the THEDES systems yields a 100% pure product, with no losses during production and no need for subsequent purification steps" [5].
In contrast, ILs require more complex synthesis and purification processes, which increase costs and lengthen development timelines [15]. However, DES are not without their challenges – high viscosity can make handling more difficult, particularly for oral delivery systems [2].
Regulatory Hurdles
Neither ILs nor DES currently have regulatory approval for slow-release drug delivery systems [18]. The absence of long-term toxicity data and comprehensive safety evaluations in humans remains a significant barrier for pharmaceutical developers. While third-generation ILs (Bio-ILs) have addressed some safety concerns, widespread adoption is still contingent on further clinical trials [2][16].
Drug Delivery Comparison Table
| Aspect | Ionic Liquids (ILs) | Deep Eutectic Solvents (DES) |
|---|---|---|
| Solubility Enhancement | Up to 35-fold plasma exposure increase [1] | Up to 12-fold solubility improvement [5] |
| Polymorphism Control | Maintains APIs in liquid state [1][2] | Maintains APIs in a supercooled state [2] |
| Biocompatibility | Poor (1st/2nd gen); improved in Bio-ILs [16][2] | High, especially with NaDES [2] |
| Toxicity Profile | High for traditional ILs; lower for Bio-ILs [16] | Low, based on natural metabolites [2][6] |
| Production Cost | Higher due to complex processes [15] | Lower with simple mixing [15][17] |
| Scalability | Challenging; requires purification [15] | Straightforward; benefits from 100% atom economy [5] |
| Viscosity Challenges | Variable; can be tailored via ion structure [2] | High viscosity may impede oral delivery [2] |
| Regulatory Status | No approved slow-release systems [18] | No approved slow-release systems [18] |
This content is for informational purposes only. Always consult official regulations and qualified professionals before making decisions about sourcing or formulations.
Biocompatibility and Toxicity
Ionic Liquids (ILs) are stabilized through ionic interactions, while Deep Eutectic Solvents (DES) rely on hydrogen bonding [2]. This fundamental structural difference often makes DES a safer choice, particularly for sensitive pharmaceutical applications, as conventional ILs tend to exhibit higher toxicity [2].
Early-generation ILs present several challenges, including high aquatic toxicity, limited biodegradability, and sensitivity to water and air [2]. These issues are further influenced by structural variations. For instance, ILs with longer alkyl chains on their cations are more toxic to biological membranes. However, incorporating ester groups into the side chains can improve their biodegradability [2].
To address these concerns, the pharmaceutical industry has developed third-generation ILs, often referred to as Bio-ILs. These formulations use biocompatible cations like cholinium, betainium, and carnitine, paired with biologically active anions [2][16]. This approach reduces toxicity while maintaining the functional benefits of ILs, highlighting the importance of solvent biocompatibility in tackling formulation challenges.
For poorly soluble drugs, enhanced biocompatibility is a game-changer. Natural Deep Eutectic Solvents (NaDES), made from primary metabolites such as sugars, amino acids, and organic acids naturally found in cells, are considered the most biocompatible solvents available [2][8]. Their components are generally recognized as safe. This is particularly relevant given that approximately 80% of new drug candidates (classified as BCS Class II/IV) struggle with poor solubility [2]. Solvent selection is critical for improving solubility without introducing additional toxicity risks.
Emma L. Smith from Nottingham Trent University highlights a key challenge in this area:
"While the individual components of DESs tend to be individually well toxicologically characterized, there is very little information about the toxicological properties of the eutectic solvents themselves" [10].
This gap in toxicological data emphasizes the need for further research, even though the natural origin of NaDES components provides some assurance regarding their safety.
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Future Developments in Pharmaceutical Use
Pharmaceutical research is making strides with third-generation Bio-ILs and Natural Deep Eutectic Solvents (NaDES), guided by principles of green chemistry. These systems prioritize biocompatible cations like choline and amino acids, which improve biodegradability and reduce toxicity. As Professor Rosa María Martín‐Aranda from UNED highlights:
"Environmental issues will drive the implementation of technologies based on ionic liquids in the pharmaceutical industry and the production of fine chemicals" [19].
A particularly promising development is Therapeutic Deep Eutectic Solvents (TheDES), which incorporate active pharmaceutical ingredients (APIs) directly into the solvent matrix. This approach eliminates polymorphism and can enhance API solubility by as much as 12 times [5]. Considering that nearly 80% of new drug candidates fall into the poorly water-soluble categories of BCS Class II and IV, these systems address a pressing need in drug formulation [2].
On the manufacturing side, techniques like Hot Melt Extrusion (HME), microwave-assisted synthesis, and ultrasonication are gaining traction. These methods enable continuous, solvent-free production processes that are more environmentally friendly [2]. With regulatory bodies increasingly pushing for the elimination of volatile organic compounds in pharmaceutical production, these green methods are aligning with industry and environmental goals. Such advancements also open the door to innovative drug delivery systems.
Surface Active Ionic Liquids (SAILs) further enhance drug delivery by acting as both solvents and surfactants. They form micelles and vesicles that improve the solubility and bioavailability of poorly soluble drugs. For instance, in September 2020, researchers Islam and Goto developed choline-based IL-in-oil microemulsions that achieved acyclovir drug loadings of up to 7.7 mg/mL, significantly improving skin permeation in pig models [1].
As biocompatibility improves, regulatory frameworks are evolving to favor formulations derived from natural sources. Many innovations now use GRAS (Generally Recognized as Safe) components, simplifying safety evaluations. Additionally, ionic liquids (ILs) and deep eutectic solvents (DES) are being reclassified as excipients or active ingredients, streamlining their regulatory approval process. With the ability to create approximately 1 million binary ILs and an astronomical 10^18 ternary ILs through different ion combinations, the possibilities for designing compliant formulations are nearly limitless [2].
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Conclusion
The discussion above underscores important differences in the performance and practicality of DES and ILs, particularly in addressing the challenge that nearly 80% of new drug candidates struggle with poor water solubility [2]. Each solvent system offers unique benefits tailored to specific formulation needs. ILs stand out for their precise tunability and high thermal stability, while DES are favored for their cost-effectiveness, eco-friendly nature, and straightforward manufacturing.
For instance, if your work involves sensitive APIs requiring specific physicochemical properties or exceptional stability, ILs – especially third-generation Bio-ILs – could be the ideal choice. On the other hand, if you’re looking for a greener, less toxic option that scales easily through methods like Hot Melt Extrusion, DES (particularly Natural Deep Eutectic Solvents) might be more practical. As Professor François Jérôme from Université de Poitiers aptly states:
"DESs exhibit similar physico-chemical properties to the traditionally used ionic liquids, while being much cheaper and environmentally friendlier" [3].
Both systems help mitigate polymorphism and significantly enhance bioavailability. For example, Therapeutic DES can improve API solubility by as much as 12-fold [5]. However, they differ in cost and complexity. DES are simpler to prepare, often requiring just physical mixing, but their high viscosity can make handling and oral administration challenging. ILs, while requiring more intricate synthesis, offer nearly unlimited design possibilities, with an estimated 1 million binary and 10^18 ternary IL combinations [2].
Emerging trends in sustainability and biocompatibility also play a critical role in solvent selection. The pharmaceutical industry increasingly favors formulations that are both biocompatible and sustainable, aligning with regulatory standards. Whether you choose DES or ILs, it’s essential to prioritize components derived from natural metabolites like choline, amino acids, or sugars to ensure safety and biodegradability. Keep water content in DES formulations below 30–40%, and always conduct long-term stability studies for reliability [2].
This content is for informational purposes only. Always consult official regulations and qualified professionals when making sourcing or formulation decisions.
FAQs
What makes Deep Eutectic Solvents more environmentally friendly than Ionic Liquids in pharmaceuticals?
Deep eutectic solvents (DES) stand out as a greener alternative to ionic liquids in pharmaceutical applications. They are usually biodegradable, non-volatile, and crafted from renewable, low-toxicity materials. This makes their environmental impact much smaller compared to ionic liquids, which can often linger in the environment and pose toxicity concerns.
Another advantage of DES is their ability to generate fewer volatile organic compounds (VOCs), which helps improve air quality. These eco-friendly qualities make DES particularly appealing for delicate drug formulations and other tightly regulated pharmaceutical processes.
How do deep eutectic solvents improve drug solubility and stability?
Deep eutectic solvents (DESs) improve drug solubility by creating flexible hydrogen-bonding networks. These solvents are formed by combining a hydrogen-bond donor – such as urea, sugars, or organic acids – with a hydrogen-bond acceptor like choline chloride. This combination disrupts the crystalline structure of drugs, allowing them to dissolve more effectively. DESs can even be customized to align with the properties of a specific active pharmaceutical ingredient (API), achieving high drug concentrations, such as 250 mg/mL for certain compounds.
In addition to enhancing solubility, DESs help maintain drug stability. Their strong hydrogen-bonding capabilities and high viscosity create a protective microenvironment, limiting exposure to oxygen and moisture that can lead to degradation. Another advantage is their ability to stay liquid at relatively low temperatures (below 212°F), making them ideal for handling sensitive formulations. With these properties, DESs offer an eco-friendly and efficient solution for improving drug solubility and ensuring stability during storage and use.
What challenges limit the use of ionic liquids in pharmaceuticals?
The use of ionic liquids (ILs) in pharmaceuticals comes with its own set of hurdles, primarily centered around biocompatibility and toxicity. To ensure ILs are safe for biological applications, they must be carefully engineered to minimize harmful effects on tissues. This has sparked extensive research aimed at developing safer, biocompatible ILs for drug formulations.
Another significant challenge lies in navigating regulatory approval. Since current drug-approval frameworks are tailored to conventional solvents, introducing ILs often demands thorough safety evaluations and comprehensive data to satisfy regulatory requirements. On top of that, the physicochemical properties of ILs – such as their high viscosity and strong ionic interactions – can complicate formulation stability, manufacturing processes, and scaling up for larger production. These factors make the integration of ILs into pharmaceuticals a complex but intriguing endeavor.





Comments are closed