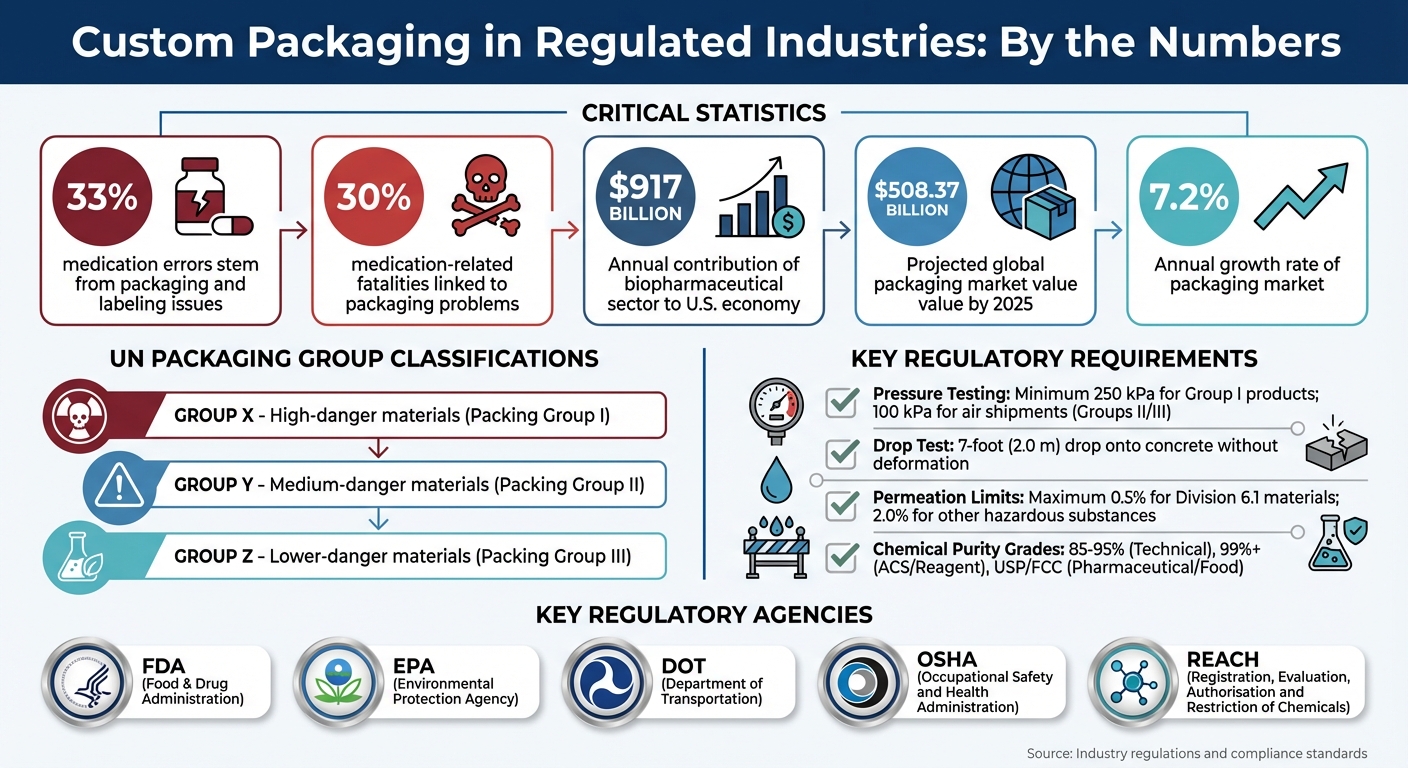
In regulated industries, packaging isn’t just about protecting products – it’s about ensuring safety and meeting legal requirements. Packaging failures can lead to contamination, recalls, or even harm to consumers. For example, 33% of medication errors stem from packaging and labeling issues, contributing to 30% of related fatalities. This makes compliance with standards from agencies like the FDA, EPA, and DOT essential.
Key considerations include:
- Material Compatibility: Packaging must resist chemical reactions, leaching, or contamination.
- Environmental Protection: Packaging should shield products from light, moisture, and extreme temperatures.
- Regulatory Compliance: Adhering to strict labeling, transport, and safety standards is non-negotiable.
Custom packaging solves these challenges by tailoring materials, designs, and features to specific product needs. For instance, temperature-sensitive items may require insulated containers or cooling systems. Allan Chemical Corporation offers expertise in creating compliant packaging solutions, ensuring safety and reliability from production to delivery.
Custom Packaging Compliance: Key Statistics and Requirements for Regulated Industries
Packaging Challenges in Regulated Industries
Navigating packaging challenges is a crucial part of protecting products and meeting strict compliance standards in regulated industries. From choosing the right containers to ensuring safe delivery, every step of chemical handling must align with complex regulatory requirements.
Meeting Regulatory Standards
Regulated industries must adhere to detailed rules set by agencies like the DOT, EPA, FDA, and UN, which govern container selection, labeling, and transport documentation [4][6][8][9]. For example, the DOT enforces hazardous materials regulations under 49 CFR, requiring shippers to address risks like corrosivity, permeability, and embrittlement when selecting packaging [4]. Similarly, the EPA mandates that hazardous waste generators follow DOT standards (49 CFR parts 173, 178, and 179) for packaging before transportation [8].
The United Nations adds another layer of oversight by requiring specific markings on compliant containers. These UN codes indicate the packaging type, material composition, and packing group: X for high-danger Group I materials, Y for medium-danger Group II, and Z for lower-danger Group III [9]. The Hazardous Materials Table in § 172.101 serves as a key reference for identifying approved packaging for specific chemicals [4]. To comply, companies must ensure the UN markings align with the product’s specific gravity and vapor pressure requirements [9].
These regulations emphasize the importance of selecting materials that maintain product integrity and stability.
Material Compatibility and Product Stability
Using incompatible packaging can lead to chemical reactions, container breakdown, and product contamination [4]. Federal regulations hold shippers accountable for ensuring that containers resist corrosivity, aging, and embrittlement over time [4].
To prevent safety risks, strict limits on permeation rates are in place for plastic containers used in chemical applications [4]. In the biopharmaceutical sector – which contributes over $917 billion annually to the U.S. economy – assessments for extractables and leachables are critical. These evaluations help avoid contamination that could compromise the safety, identity, strength, quality, or purity of products [10][11]. Packaging for higher-purity chemical grades, such as USP or FCC, must also be free from harmful contaminants to ensure safety for human use [3].
Once the right materials are chosen, packaging must still withstand the challenges of transportation and storage.
Transportation and Storage Requirements
Transporting chemicals exposes packaging to a range of physical stresses, including shocks, vibrations, temperature swings, and pressure changes, all of which can lead to leaks or damage [4][9]. For instance, liquids expand when heated, and without sufficient headspace (known as outage or ullage), this expansion can cause leaks or permanently distort containers [4].
Pressure management is another critical concern, especially for materials that release gases or have high vapor pressures. Packaging Group I products must withstand a minimum test pressure of 250 kPa, while air shipments of Group II or III materials require at least 100 kPa ratings [9]. For self-reactive Division 4.1 materials, exceeding control or emergency temperatures during transit can trigger dangerous decomposition. To address this, temperature-controlled packaging with coolants like dry ice or liquid nitrogen is often necessary [12]. Additionally, cylinder valve protection must endure a 7-foot (2.0 m) drop onto concrete without deforming or breaking [7].
These challenges highlight the importance of creating tailored packaging solutions that meet both regulatory requirements and practical demands.
Disclaimer: This content is for informational purposes only. Always consult official regulations and qualified professionals when making sourcing or formulation decisions.
Custom Packaging Solutions for Compliance and Safety
Custom packaging transforms regulatory hurdles into precise, tailored solutions by aligning every aspect of the container with the specific hazards and regulatory requirements of the chemical it holds. Instead of relying on one-size-fits-all containers, this approach ensures that materials, closures, and protective features are carefully matched to the chemical’s hazard profile. This not only safeguards the product’s integrity but also ensures compliance from the moment the product is filled until it reaches its final destination.
Application-Specific Packaging
The choice of containers is guided by the chemical’s hazard classification and physical properties. Packaging adjustments, such as pressure ratings and UN markings (“X” for high hazard, “Y” for medium hazard, and “Z” for lower hazard), ensure the container aligns with the chemical’s risk level[9]. High-hazard applications often require specialized closures, including taper-threaded connections, non-perforated packless valves, or solid plugs with inert gasketing, to eliminate the risk of leaks[7].
Material compatibility is another critical factor. Containers, valves, plugs, and gaskets must resist degradation, discoloration, or brittleness when exposed to the chemical[7]. For added protection, opaque or amber-colored materials and resins like HDPE or LDPE are used to shield contents from light, moisture, or reactive gases like oxygen[1]. In cases where self-reactive materials pose a risk of decomposition during transit, packaging solutions may include interior supports for coolants such as dry ice or liquid nitrogen to maintain safe temperatures, even if primary refrigeration systems fail[12].
Additional physical protections and controlled environments further enhance the reliability of these packaging solutions.
Controlled Packaging Environments
Contamination is a concern even before the chemical enters its container. Cleanrooms and controlled environments play a vital role in packaging operations, particularly for pharmaceuticals, high-purity chemicals, or substances intended for food-contact applications[16]. These facilities adhere to strict cleanliness standards, requiring comprehensive employee training and rigorous hygiene protocols at every stage – from blending to filling and final packaging[15][16].
To ensure safety during transportation, containers are equipped with protective caps, valve guards, and reinforced overpacks. For Hazard Zone A or B materials, these safeguards must endure drops of at least 7 feet (2.0 meters) onto concrete or steel without compromising the container’s integrity[7]. Additionally, cylinders with wall thicknesses under 0.08 inch (2.03 mm) must be overpacked in protective boxes to meet safety requirements[7].
Labeling and Documentation Requirements
Accurate labeling and thorough documentation are the final layers of protection in ensuring compliance. Clear labels prevent mix-ups and contamination, while regulations demand separation of packaging operations and unambiguous container identification[18][19]. Labels must include lot or control numbers to enable full traceability of manufacturing history and batch control[18][19]. For chemicals, labels must comply with the Globally Harmonized System (GHS), using standardized red-bordered pictograms to communicate hazards clearly[17].
Documentation responsibilities extend well beyond the label itself. Under the Federal Food, Drug, and Cosmetic Act, a product is considered misbranded if its packaging lacks required information or fails to alert consumers to tamper-evident features[18]. Custom packaging services ensure detailed batch production records are maintained, documenting the suitability of labeling materials before operations begin. For drug products, when cut labeling is used, every label must undergo a 100-percent inspection – either electronically or through manual visual checks verified by a second person[18]. Strict control over labeling issuance also requires the destruction of any unused labels bearing lot or control numbers[18]. These meticulous processes ensure safe, compliant, and traceable chemical management.
Disclaimer: This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
Benefits of Custom Packaging for Regulated Industries
Custom packaging offers unique advantages that go beyond standard options, addressing critical needs like product safety, operational efficiency, and brand trust. These tailored solutions ensure product integrity, reduce costs, and enhance the way customers perceive your brand.
Product Safety and Stability
The use of non-reactive materials – such as HDPE for corrosive substances or stainless steel for reactive chemicals – plays a key role in maintaining product safety and effectiveness. Experts agree that proper packaging not only ensures compliance but also enhances overall safety and operational reliability[21].
Custom packaging designs often include features like amber or opaque containers to shield products from UV light, moisture, and reactive gases[22][1]. For temperature-sensitive items, insulated containers or specialized cooling systems provide stability even during refrigeration failures[20][22]. Additional safeguards, such as double seals, pressure relief valves, and secondary containment, offer robust protection against leaks and contamination[20][22]. Some suppliers are even integrating IoT sensors and RFID tags to monitor conditions like temperature and pressure in real time, enabling early detection of potential issues before they escalate into serious problems[20][22].
Beyond enhancing product safety, custom packaging contributes to operational efficiency.
Improved Efficiency
Custom packaging equipped with RFID tags and IoT sensors simplifies tracking and provides real-time alerts, reducing the likelihood of manual errors[22]. Ergonomically designed dispensing tools help minimize spills and handling risks, making day-to-day operations safer and more efficient[22].
Digital tools like QR codes on custom labels grant instant access to Material Safety Data Sheets (MSDS), eliminating the need for physical paperwork and streamlining compliance workflows[22]. Automated filling and sealing systems integrated with custom-designed containers ensure consistent quality and safety standards across production batches[20]. Additionally, packaging tailored for specific storage conditions – such as moisture-resistant barriers or temperature-controlled environments – preserves product quality even through complex logistics networks.
These operational improvements not only save time and reduce errors but also contribute to a stronger brand presence in the market.
Branding and Market Differentiation
In highly regulated industries, where trust is a deciding factor, the quality of your packaging can make or break a customer’s confidence in your product. Generic or poorly designed packaging may suggest a lack of attention to detail, while professional custom packaging communicates reliability and care[23].
Custom solutions allow businesses to integrate regulatory elements like GHS labels, hazard symbols, and safety instructions into their designs seamlessly, rather than as clunky add-ons[22][20]. This approach not only meets compliance requirements but also reinforces brand identity. Logos, color schemes, and thoughtfully designed packaging boost brand recognition, encouraging repeat purchases and making product information easy to find[23][24]. Companies that adopt advanced features like smart packaging or environmentally friendly materials stand out by demonstrating a commitment to safety and sustainability, positioning themselves as leaders in their field[22][20].
By addressing both regulatory and operational needs, custom packaging doesn’t just protect your product – it strengthens your brand and competitive edge.
Disclaimer: This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
sbb-itb-aa4586a
Allan Chemical Corporation‘s Custom Packaging Capabilities
Allan Chemical Corporation stands out in the field of custom packaging, offering tailored solutions for industries with strict regulatory requirements. With over 40 years of experience, the company combines a deep understanding of compliance with adaptable service models to meet the needs of sectors like pharmaceuticals, food, and cosmetics. This expertise ensures packaging solutions that balance regulatory demands with operational efficiency, supported by stringent quality controls.
Certified Processes for Quality and Compliance
Allan Chemical follows ISO 9001:2015 standards, ensuring that every step of the packaging process adheres to strict quality procedures and minimizes contamination risks[26][27]. This commitment to quality is complemented by adherence to ISO 14000 standards, which focus on environmentally responsible practices. Together, these certifications highlight the company’s dedication to both product integrity and sustainable operations.
Documentation is a cornerstone of Allan Chemical’s compliance framework. Customers have access to Certificates of Analysis (COA), Safety Data Sheets (SDS), and Technical Data Sheets (TDS) upon request. This ensures full traceability and audit readiness, providing peace of mind for businesses operating in regulated environments[25].
Flexible Packaging and Delivery Options
Allan Chemical’s flexible service model is designed to accommodate varying production requirements. Whether you need small batch sizes or larger orders, the company ensures timely delivery. Orders placed by 2:00 PM CT are shipped the same day, and next-day shipping options are available for urgent needs[25]. This efficiency helps businesses maintain lean inventories without disrupting production schedules.
For hazardous materials, Allan Chemical uses DOT-certified and UN-rated packaging to ensure safe transport and compliance with labeling standards. Controlled environments during repackaging prevent contamination, even under tight deadlines[26]. Every shipment is backed by detailed documentation, reinforcing the company’s commitment to safety and compliance.
Expert Technical Support
Allan Chemical provides more than just packaging services – it offers expert technical support to guide customers through regulatory challenges and product specifications. The technical support team works closely with clients, offering immediate access to essential documentation and compliance assistance. This ongoing support reflects Allan Chemical’s focus on building long-term partnerships rather than one-off transactions.
By combining certified processes, adaptable service models, and dedicated technical support, Allan Chemical positions itself as a trusted partner for businesses in regulated industries. Its family-owned structure emphasizes personalized service and a commitment to quality, ensuring that packaging solutions meet both regulatory standards and operational needs.
Disclaimer: This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
How to Implement Custom Packaging Solutions
Introducing custom packaging in regulated industries requires a careful balance of compliance, safety, and operational efficiency. This process starts with understanding the relevant regulations and ends with seamlessly integrating the packaging into production. Each step is crucial – any misstep could lead to costly product recalls or compliance violations.
Assessing Regulatory and Material Requirements
The first step is identifying the federal regulations that apply to your industry. For example, pharmaceutical companies must adhere to FDA Current Good Manufacturing Practice (CGMP) regulations outlined in 21 CFR Parts 210 and 211 [18][1]. If you’re dealing with hazardous materials, the Department of Transportation (DOT) standards under 49 CFR Parts 173, 178, and 179 dictate packaging requirements [8][5]. Knowing these rules early on helps avoid expensive redesigns down the road.
When evaluating packaging, focus on four key areas: protection, compatibility, safety, and performance [1]. Protection ensures the packaging shields the product from light, oxygen, water vapor, and microbial contamination. Compatibility means the packaging materials shouldn’t interact with the product in ways that could reduce potency or cause discoloration. Safety is especially critical for high-risk applications like injectables or inhalation products, where toxicological evaluations and extraction studies are required [1][2]. Performance testing verifies that the packaging functions properly – whether it’s delivering an exact dose from an inhaler or maintaining sterility in prefilled syringes.
Material selection depends on the purity needs of your application. For general industrial use, technical grade chemicals (85–95% purity) might suffice. However, analytical work often requires ACS/Reagent grade chemicals (99% or higher purity) [3]. For food and pharmaceutical applications, USP/FCC grades are mandatory. The United States Pharmacopeia – National Formulary provides standards for identity, strength, and purity, which are essential when selecting materials [28]. For oral and topical products, safety can often be established by referencing Food Additive Regulations (FAR) under 21 CFR 172–186, while injectables typically require USP Biological Reactivity Tests [2].
"A packaging system found acceptable for one drug product is not automatically assumed to be appropriate for another." – FDA Guidance for Industry [1]
Stability testing is critical for determining expiration dates and ensuring the packaging maintains product quality over time [18][1]. For over-the-counter (OTC) drug products, tamper-evident features are also required to visibly indicate any tampering [18]. To maintain quality control, establish written procedures for receiving, identifying, sampling, and testing all packaging materials before use [18].
Once these requirements are clear, the next step is finding a packaging partner with a proven track record in regulatory compliance.
Choosing Experienced Suppliers
The right packaging supplier is essential for ensuring compliance and product safety. Look for partners who have in-depth knowledge of regulations from agencies like the FDA (food and drugs), EPA (disinfectants and pesticides), and DOT (hazardous materials) [15][13][29]. Their expertise should go beyond surface-level understanding and include hands-on experience with audits and inspections.
Choose suppliers with strong quality assurance systems, full traceability, and certifications like UN ratings and documented (C)GMP compliance [15][29][30]. Reliable co-packing facilities can maintain high efficiency – up to 90% – while meeting strict regulatory standards [15]. Conduct audits of the supplier’s material sources to ensure compliance with federal and state safety standards, such as California’s Proposition 65 [13].
"Packaging compliance is not about avoiding penalties; it’s about making safe, trustworthy products that your customers will buy." – Maker’s Row [13]
Suppliers should also perform rigorous testing, including drop, compression, and environmental stress tests, to identify potential issues before the product hits the market [14][30]. Evaluate their ability to handle lead times and navigate challenges like international holidays or supply chain disruptions [30]. A trustworthy partner will provide realistic timelines for compliant packaging approval rather than making unrealistic promises.
Once you’ve secured a reliable supplier, the final step is integrating the custom packaging into your production process.
Integrating Custom Packaging into Production
After defining regulatory needs and securing a capable supplier, focus on streamlining the integration process. Standardize artwork approval workflows by involving legal, marketing, and regulatory teams to create error-free packaging designs [31]. Use digital tools and specialized software to automate label checks, reducing the risk of mistakes in ingredient lists or hazard warnings [32]. These proactive measures can prevent costly recalls.
Ensure that all packaging is designed to withstand vibrations, temperature changes, and shocks during transportation [30][4]. For hazardous materials, plastic packaging must meet strict permeation rate limits – no more than 0.5% for Division 6.1 materials or 2.0% for other hazardous substances over specified timeframes [4]. Functional barriers, such as metal foil, can be added to prevent migration of inks or adhesives into the product [2]. Regularly train staff on storage requirements and the proper handling of child-resistant or sterile packaging [30].
Detailed record-keeping is essential throughout the process. Maintain thorough documentation of material sourcing, regulatory correspondence, and testing results to simplify audits and demonstrate compliance [14]. Develop multi-language labeling templates to adapt easily for different markets without compromising brand consistency [32]. Review your supply chain to verify that every material and process complies with safety standards, including FDA Food Additive Regulations (FAR) [2][13].
Continuous improvement is vital. Update processes regularly and train teams to keep up with evolving regulations and supply chain changes [14][30]. The global packaging market is expected to reach $508.37 billion by 2025, with a growth rate of 7.2% annually [31]. Many companies are using AI-driven tools to track regulatory updates in real time from agencies like the FDA and EMA [31]. Consider adopting similar systems to stay ahead of compliance changes.
Disclaimer: This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
Conclusion
Custom packaging plays a critical role in ensuring regulatory compliance, safeguarding safety, and supporting operational efficiency. By meeting the strict standards set by agencies like OSHA, EPA, DOT, and REACH, proper packaging not only protects workers and the environment from potential hazards but also prevents dangerous chemical reactions through careful material selection and advanced containment features. These measures collectively reduce risks across the entire supply chain.
Beyond safety, custom packaging enhances operational reliability. Technologies designed to minimize errors and streamline processes allow businesses to maintain lean inventories while consistently delivering high-quality products. Additionally, adopting sustainable practices – such as biodegradable materials or reusable containers – not only aligns with environmental goals but also supports compliance with regulatory requirements. These advantages highlight the importance of collaborating with a knowledgeable packaging supplier.
Partnering with experienced professionals ensures that manufacturers can meet regulatory demands through precise material choices, thoughtful designs, and accurate labeling.
Allan Chemical Corporation exemplifies this expertise with over 40 years of experience in providing tailored packaging solutions. Their flexible batch sizes, just-in-time delivery, and deep knowledge of compendial-grade standards (USP, FCC, ACS, NF) cater to industries such as pharmaceuticals, food, cosmetics, and industrial applications. With robust technical support and a commitment to exact specifications, Allan Chemical Corporation offers reliable solutions for navigating the challenges of regulated markets.
Achieving success with custom packaging requires careful planning, collaboration with trusted suppliers, and a commitment to continuous improvement. By investing in tailored packaging strategies, businesses can protect their products, ensure workforce safety, and strengthen their reputation – ultimately gaining a competitive edge in regulated industries.
Disclaimer: This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
FAQs
Which U.S. agencies regulate packaging for chemicals and other products in regulated industries?
In the U.S., packaging for chemicals, pharmaceuticals, and other regulated products is closely monitored by three primary agencies:
- U.S. Department of Transportation (DOT): The DOT enforces packaging standards through its Hazardous Materials Regulations (HMR). These regulations address general packaging requirements, chemical compatibility, and testing to ensure safe transportation.
- Environmental Protection Agency (EPA): The EPA focuses on hazardous waste packaging, requiring that it meets strict safety guidelines before it can be transported or disposed of properly.
- Food and Drug Administration (FDA): The FDA oversees packaging for pharmaceuticals and biologics, ensuring compliance with safety and efficacy standards outlined in the Federal Food, Drug, and Cosmetic Act.
Together, these agencies ensure that packaging in regulated industries adheres to rigorous safety, environmental, and quality standards across the United States.
How does custom packaging improve the safety and stability of chemical products?
Custom packaging is essential for ensuring the safety and stability of chemical products, as it addresses their unique requirements. By incorporating compatible materials, leak-resistant seals, and temperature-controlled designs, it minimizes risks like leaks, unwanted chemical reactions, or product degradation during handling, transport, and storage.
Moreover, features such as secondary containment and customized designs help meet regulatory standards, safeguarding both the product and the people handling it. This level of precision is critical in industries where safety and accuracy cannot be compromised.
Why is material compatibility important in custom packaging for regulated chemicals?
Material compatibility plays a key role in custom packaging, ensuring that both the container and its contents remain stable and safe. Choosing the wrong material can trigger chemical reactions, leading to degradation, corrosion, or even the release of harmful by-products. These issues can jeopardize safety, compromise product quality, and violate regulatory standards.
Custom packaging solutions are designed with the specific properties of the stored chemical in mind. For instance, acids can corrode metals, while certain plastics may break down when exposed to solvents. By carefully selecting materials like HDPE (High-Density Polyethylene), PP (Polypropylene), or specific metals based on the product’s needs, packaging can retain its integrity throughout storage, transport, and handling. This approach not only safeguards the product but also ensures compliance with strict regulations set by agencies such as the EPA, OSHA, and DOT.
At Allan Chemical Corporation, matching regulated products with the appropriate packaging materials is a priority. Their expertise ensures safety, stability, and adherence to compliance standards across industries including pharmaceuticals, food, cosmetics, ceramics, and electronics.





Comments are closed