Importing chemicals into the U.S. requires compliance with the Toxic Substances Control Act (TSCA), which regulates chemical substances to protect public health and the environment. Under TSCA Section 13, importers must certify shipments as either compliant with TSCA (positive certification) or exempt (negative certification). Failure to comply can result in fines up to $50,000 per day per violation and shipment delays.
Key Steps for TSCA Compliance:
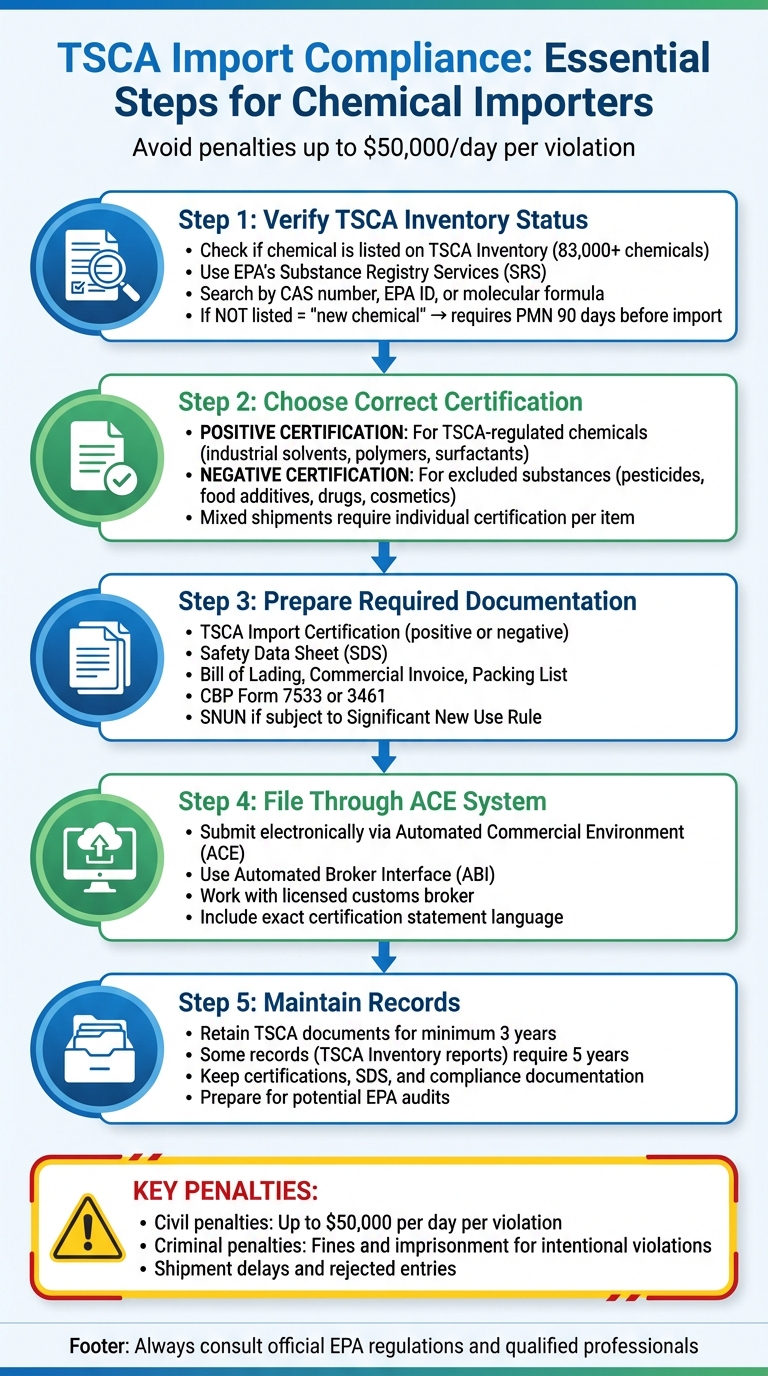
- Verify TSCA Inventory Status: Check if the chemical is listed as "existing" on the TSCA Inventory. Unlisted chemicals require a Premanufacture Notification (PMN) to be filed 90 days before import.
- Choose the Right Certification: Submit a positive certification for TSCA-regulated chemicals or a negative certification for excluded items like pesticides or drugs.
- File Through ACE System: Use the Automated Commercial Environment (ACE) system for electronic certification submission.
- Maintain Records: Retain TSCA-related documents for at least three years.
Avoid Common Pitfalls:
- Misclassifying substances under TSCA vs. other federal regulations.
- Overlooking regulatory flags like Significant New Use Rules (SNURs).
- Failing to provide accurate certifications for mixed shipments.
Proper preparation, accurate documentation, and staying informed about regulatory updates are essential for smooth customs clearance and avoiding penalties.
TSCA Import Compliance Process: 5 Essential Steps for Chemical Importers
Import and Export Certification, Allegations/Substantial Risk
TSCA Import Certification Basics
After confirming a chemical’s status on the TSCA Inventory, the next step is ensuring proper certification for regulatory compliance. Every chemical shipment entering the U.S. must include a TSCA certification filed with U.S. Customs and Border Protection (CBP). This certification confirms that the shipment either complies with TSCA regulations or is exempt from TSCA’s jurisdiction. Filing is done electronically through the ACE system at the time of entry.
There are two types of certifications: positive and negative. A positive certification confirms that the shipment adheres to TSCA requirements, including compliance with Section 5 (Premanufacture Notification and Significant New Use Rules), Section 6 (restrictions on specific chemicals like PCBs and asbestos), and Section 7 (judicial actions). The declaration reads:
"I certify that all chemical substances in this shipment comply with all applicable rules or orders under TSCA and that I am not offering a chemical substance for entry in violation of TSCA or any applicable rule or order thereunder" [3].
This certification applies to substances covered under TSCA, such as industrial solvents, polymers, and surfactants listed on the TSCA Inventory.
A negative certification, on the other hand, is used when the shipment includes substances regulated by other federal agencies. The declaration states:
"I certify that all chemicals in this shipment are not subject to TSCA" [3].
This applies to items such as pesticides (regulated under FIFRA), food additives, drugs, cosmetics, medical devices (regulated by the FDA), and other excluded products like tobacco, nuclear materials, firearms, and ammunition.
Key Terms and Definitions
TSCA-regulated substances include a broad range of industrial chemicals, such as solvents, polymers, plastics, inorganic chemicals (metals and minerals), petrochemicals, surfactants, and chemical additives. These substances must appear on the TSCA Inventory, which lists over 83,000 chemicals [1]. Any chemical not on the Inventory is classified as "new" and requires a Premanufacture Notification (PMN) to be submitted at least 90 days before import [5].
Excluded materials are governed by other federal regulations. For example, finished products (articles) that contain chemicals but do not release them during normal use typically do not require TSCA certification unless a specific TSCA rule applies.
Positive vs. Negative Certification
The choice between positive and negative certification depends on the substance’s regulatory classification. A positive certification is required for chemicals subject to TSCA rules, including those listed on the TSCA Inventory, items imported under an R&D exemption, and new chemicals with approved PMNs. Industrial chemicals like solvents, polymers, and surfactants commonly fall under this category.
Negative certification is used when the substance is regulated by another federal agency. For instance, vitamin C for food use is regulated by the FDA under the FFDCA, while pesticides are managed by the EPA under FIFRA. Misclassification can result in shipment delays and penalties of up to $50,000 per day [3].
| Certification Type | When to Use | Typical Examples | Regulating Agency |
|---|---|---|---|
| Positive | Chemical complies with TSCA rules | Industrial solvents, polymers, surfactants | EPA (under TSCA) |
| Negative | Chemical is excluded from TSCA | Pesticides, food additives, drugs, cosmetics, nuclear materials | FDA, EPA (FIFRA), NRC, ATF |
Mixed Shipments
For shipments containing both TSCA-regulated and excluded substances, each item must be certified individually based on its regulatory status. For example, if a shipment includes a TSCA-covered industrial solvent and an FDA-regulated food-grade preservative, the solvent requires a positive certification, while the preservative needs a negative certification.
Within the ACE system, you must provide the appropriate certification code for each line item. A single blanket certification for the entire shipment is not allowed. CBP officially discontinued blanket certifications on March 21, 2017 [6]. To ensure compliance, work closely with your customs broker to confirm that each substance is properly identified and certified before the shipment reaches the port of entry.
At Allan Chemical Corporation, we recognize the importance of TSCA certification in upholding compliance with U.S. chemical import regulations. Up next, learn how to prepare your documentation before importing.
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
Pre-Import Compliance Steps
To avoid delays, penalties, or rejected entries, it’s important to complete these steps before importing. These tasks help confirm your substance’s regulatory status and ensure you have the necessary documentation in place.
TSCA Inventory Check
Start by determining whether your chemical is classified as "existing" (listed) or "new" (not listed) under the Toxic Substances Control Act (TSCA). A chemical is considered existing if it appears on the TSCA Inventory, while anything not listed is classified as new [2]. To check the most up-to-date list of active chemicals, refer to the TSCA Inventory, which the EPA updates twice a year [7][8].
Use the EPA’s Substance Registry Services (SRS) to search for your chemical by its CAS number, EPA ID, molecular formula, or synonyms [10]. The SRS provides details like regulatory flags (e.g., SNURs or Section 4 test rules) and specifies whether a chemical is marked as "active" or "inactive" [8][9]. If your chemical’s identity is considered confidential business information (CBI), it may not appear in the public Inventory by name. In such cases, you’ll need to contact the EPA directly for clarification [4].
"For purposes of regulation under TSCA, if a chemical is on the TSCA Inventory, the substance is considered an ‘existing’ chemical substance in U.S. commerce. Any chemical that is not on the Inventory is considered a ‘new chemical substance.’" – US EPA [2]
If your chemical is not listed on the Inventory, you must submit a Pre-manufacture Notification (PMN) at least 90 days before importing [4]. Before filing, check if your substance qualifies for exemptions such as the Low Volume Exemption (LVE), Research and Development (R&D) Exemption, or Polymer Exemption [4].
Required Documentation
Once you’ve confirmed your chemical’s Inventory status, gather the necessary documentation to ensure compliance. For every shipment, you’ll need one of the following:
- A positive TSCA Import Certification (confirming compliance)
- A negative certification (indicating the substance is excluded) [5]
Additionally, prepare a Safety Data Sheet (SDS) that outlines the chemical’s properties, hazards, and handling instructions [5]. Other standard customs documents include the Bill of Lading (BoL), Commercial Invoice, and Packing List [5]. For customs clearance, file CBP Form 7533 or 3461 with U.S. Customs and Border Protection [5]. If your chemical is subject to a Significant New Use Rule (SNUR), you must also file a Significant New Use Notice (SNUN) with the EPA at least 90 days before import [5].
Be sure to retain all TSCA-related records for at least three years, as required by law [5].
Identify Applicable Exclusions
Next, determine if your substance qualifies for any statutory exclusions. Certain products – like food, drugs, cosmetics, and pesticides – are excluded from TSCA oversight and follow different regulatory pathways [1][5].
If your chemical is manufactured exclusively for export, you may be exempt from PMN requirements if it is labeled "for export" and the recipient intends to export or process it solely for export [9]. Export notification is typically not required for articles (finished products that don’t release chemicals during normal use) or for substances present in de minimis concentrations [9]. Check 40 CFR 707 Subpart D to confirm if article or de minimis exclusions apply to your case [9].
When reviewing the TSCA Inventory, pay attention to regulatory flags such as SNURs, as these may impose additional requirements even for chemicals already listed [8].
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
sbb-itb-aa4586a
Filing TSCA Certification with CBP
Once you’ve completed your pre-import compliance steps, the next critical task is electronically filing your TSCA certification with U.S. Customs and Border Protection (CBP). This step is required for all chemical imports and must be completed through the appropriate channels before your shipment can clear customs. Typically, this responsibility falls to the Importer of Record (IOR) or an authorized customs broker, who handles the submission as part of the entry documentation process [5].
Using the ACE System
TSCA certifications must be submitted electronically via the Automated Commercial Environment (ACE) system, using the Automated Broker Interface (ABI) [5]. ACE serves as the main platform for transmitting necessary documentation to both CBP and the Environmental Protection Agency (EPA). Licensed customs brokers are often relied upon to manage these technical submissions and help avoid any delays at the border.
For detailed specifications on electronic submissions, refer to the CAMIR (Customs Automated Manifest Interface Requirements) and CATAIR (Customs and Trade Automated Interface Requirements) guidelines [11][12]. These requirements differ depending on the mode of transportation – whether your shipment arrives by air, ocean, truck, or rail – since each has unique message identifiers. Be sure to have all required supporting documents readily available [5].
Certification Statement Requirements
Your TSCA certification must include the appropriate regulatory statement, depending on whether the shipment includes TSCA-regulated chemicals or substances excluded from TSCA. Use the exact language specified by regulations:
- For TSCA-regulated chemicals, submit a positive certification:
"I certify that all chemical substances in this shipment comply with all applicable rules or orders under TSCA and that I am not offering a chemical substance for entry in violation of TSCA or any applicable rule or order thereunder" [3]. - For substances excluded from TSCA – such as pesticides, food additives, drugs, cosmetics, or nuclear materials – submit a negative certification:
"I certify that all chemicals in this shipment are not subject to TSCA" [3].
Accuracy is crucial here. Double-check the regulatory status of your shipment to ensure the correct certification is filed. The Importer of Record bears responsibility for the accuracy of the certification and compliance with all TSCA regulations [5]. Mistakes or violations can lead to severe consequences, including civil penalties of up to $50,000 per day per violation, and even criminal penalties, such as fines and imprisonment for intentional violations [3]. This final step ties together your earlier compliance measures, completing the TSCA import process.
This material is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
Post-Import Compliance and Record Retention
Once you’ve completed the initial filing and pre-import preparations, maintaining compliance becomes an ongoing effort. This involves disciplined recordkeeping, staying updated on regulatory changes, and being audit-ready. Filing your TSCA certification is just the start of this process. To ensure compliance with the Toxic Substances Control Act (TSCA), you must keep meticulous records, monitor for updates, and prepare for potential audits. Under TSCA Section 11, the EPA has the authority to inspect facilities where chemical substances are stored or handled, and they may access or copy any required records [14]. Failing to follow proper post-import practices can result in civil penalties of up to $25,000 per day [13].
Document Retention Periods
Proper organization and retention of records are critical for demonstrating compliance. Generally, records should be kept for 3 years, though certain documents, like TSCA Chemical Substance Inventory reports under 40 CFR 710, must be retained for 5 years [13].
Your recordkeeping system should include all materials needed to verify or reconstruct any data submitted in TSCA reports. This includes primary materials used to calculate production volumes or assess exposure data, as well as customer notifications about their reporting responsibilities, complete with signed return receipts. For importers of Per- and Polyfluoroalkyl Substances (PFAS), it’s essential to maintain detailed records documenting chemical identity, molecular structure, annual production volumes (dating back to January 1, 2011), and any related health and safety studies [13][15].
Monitoring Regulatory Updates
TSCA regulations are constantly evolving, so staying informed is key to compliance. With more than 83,000 chemical substances listed on the TSCA Inventory, the EPA frequently updates rules that impact import requirements [1]. To stay up to date, regularly consult the Federal Register and the Electronic Code of Federal Regulations (ECFR) [13][17].
Subscribing to EPA news releases can help you stay informed, especially on topics like the PFAS Strategic Roadmap and proposed changes to reporting requirements [16]. It’s also a good practice to routinely review the TSCA Chemical Substance Inventory to check for updates on imported substances or changes to their status [1]. Pay close attention to "sunset dates" for chemicals subject to TSCA Section 4 testing requirements, as these dates determine when notification obligations for import and export expire [9]. Additionally, leverage tools like the EPA’s Central Data Exchange (CDX) and the Chemical Information Submission System (CISS) to access updated electronic reporting forms and guidance [13][9]. These steps will help ensure your documentation is accurate and ready for any audits.
Preparing for Audits
Maintaining accessible and well-organized records is essential for audit readiness. Use a centralized digital system to store all records related to controlled substances, and establish a routine review process to ensure compliance. Make sure all relevant documentation, regardless of its origin, is easy to retrieve [13].
Using electronic reporting tools like the EPA’s CDX and CISS can provide a secure digital trail for all filings [15]. Develop a formal process to evaluate information under the "known to or reasonably ascertainable" standard, which requires organizations to account for all data they possess or should reasonably know [13]. For high-risk substances, such as Polychlorinated Biphenyls (PCBs), additional logs may be required [14]. Furthermore, keep records of significant adverse health reactions – 30 years for employee-related incidents and 5 years for non-employee incidents involving health or environmental impacts [14].
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
Final Compliance Checklist
To ensure your shipment is fully compliant with TSCA regulations, review this checklist before proceeding:
- Verify TSCA Inventory Status
Confirm whether the chemical is listed on the TSCA Inventory. If it’s not, it’s classified as a new chemical and requires submitting a Premanufacture Notice (PMN) at least 90 days before shipment [3]. - Select the Correct Certification
Choose from the following certifications based on your shipment:- Positive Certification: "I certify that all chemical substances in this shipment comply with all applicable rules or orders under TSCA and that I am not offering a chemical substance for entry in violation of TSCA or any applicable rule or order thereunder." [3]
- Negative Certification: "I certify that all chemicals in this shipment are not subject to TSCA." [3]
- Confirm Absence of Prohibited Substances
Make sure the shipment does not include substances banned under Section 6, such as certain PCBs or products containing asbestos. - Evaluate Import Volume Against CDR Thresholds
Determine if your import volume triggers Chemical Data Reporting (CDR) requirements, which typically apply at 25,000 pounds per site annually – or 2,500 pounds for chemicals under specific TSCA actions [3]. - File Documentation and Maintain Records
Submit all required paperwork through the Automated Commercial Environment (ACE) system. Keep certifications, Safety Data Sheets, and related records for at least three years [13] [18]. - Seek Expert Assistance
For complex compliance challenges, consult Allan Chemical Corporation. Their expertise can help navigate the intricacies of TSCA requirements.
Non-compliance can result in severe penalties, so thorough preparation is essential.
Note: This checklist is for informational purposes only. Always consult official regulations and qualified experts before making sourcing or formulation decisions.
FAQs
What should I do if a chemical is not listed on the TSCA Inventory?
If a chemical isn’t included on the TSCA Inventory, it cannot be legally imported into the United States unless it qualifies for an EPA-approved exemption. Attempting to bring in unlisted chemicals without the correct exemption will lead to the shipment being denied entry.
To prevent delays or penalties, double-check that all chemicals you intend to import are either listed on the TSCA Inventory or meet the requirements for a valid exemption. Accurate documentation and strict compliance are key to ensuring a hassle-free import process.
How can I tell if my shipment needs a positive or negative TSCA certification?
To figure out the right TSCA certification for your shipment, start by listing all the chemicals, mixtures, or articles it includes. Be sure to note their CAS Registry Numbers, commercial names, and intended uses. Then, check if each substance is on the TSCA Inventory or qualifies for an exclusion, such as chemicals used only in research or specific articles. If the shipment includes substances regulated by TSCA, you’ll need a positive certification. On the other hand, if none of the substances fall under TSCA, a negative certification is required.
For shipments that include exempt items like tobacco or certain articles, certification might not be necessary. However, it’s essential to clearly document the exemption. If you’re uncertain about a chemical’s TSCA status, reach out to the EPA TSCA Assistance Office for clarification before submitting your certification. Taking these steps ensures you’ll determine the correct certification for your shipment without complications.
What happens if you fail to comply with TSCA import regulations?
Failure to follow TSCA import regulations can lead to strict actions by the EPA. These actions might include hefty fines, shipment denials or seizures, and even civil or criminal penalties in more severe cases.
To prevent these issues, it’s crucial to verify that all imported chemicals comply with TSCA certification rules and to keep thorough documentation to meet regulatory standards.





Comments are closed