Attenuated Total Reflectance Fourier Transform Infrared Spectroscopy (ATR-FTIR) is a method used to analyze how molecules attach to nanoparticle surfaces in liquid environments. Unlike traditional methods that require drying samples, ATR-FTIR enables real-time analysis without altering the sample’s natural state. This technique is critical for studying adsorption processes that are key to fields like catalysis, drug development, and environmental cleanup.
ATR-FTIR works by using infrared light and a crystal to create an "evanescent wave", which interacts with the sample’s surface. Molecules absorb specific wavelengths of light, producing a spectrum that reveals their chemical bonds and interactions. By immobilizing nanoparticles on the crystal, researchers can monitor adsorption dynamics and molecular changes with high sensitivity.
Studies have shown that ATR-FTIR can differentiate between types of molecular binding (e.g., inner-sphere and outer-sphere complexes) and track reactions in real time. For example, it has been used to observe protein adsorption on zinc oxide films, ligand interactions with titanium dioxide, and CO2 reduction on copper-based nanoparticles. These insights are essential for improving industrial processes, designing pharmaceuticals, and developing advanced materials.
ATR-FTIR’s ability to provide precise, surface-specific information makes it a valuable tool for understanding nanoparticle behavior in real-world applications.
How ATR-FTIR Analyzes Surface Adsorption
Evanescent Wave and Surface Sensitivity
ATR-FTIR owes its effectiveness to the evanescent wave, a unique phenomenon that occurs during total internal reflection of infrared light within the crystal. Unlike regular light, this wave penetrates the sample only slightly – typically between 0.5 and 5 μm – decaying exponentially as it moves away from the crystal surface. This makes ATR-FTIR particularly adept at analyzing molecules adsorbed on surfaces, rather than those in the bulk solution.
The depth of penetration varies based on factors like the infrared light’s wavelength and the type of crystal used. For instance, a silicon ATR crystal in the mid-infrared range reaches about 1 μm into the sample, while a zinc selenide (ZnSe) crystal, paired with a sample refractive index of 1.5 at a wavelength of 10 μm, achieves a penetration depth of roughly 2.0 μm [2]. Wolfgang G. Hinze from the University of Konstanz highlights this precision:
"At each reflection point, the intensity of the IR beam decreases exponentially with the distance to the surface, thus processes in close proximity to the ATR crystal surface can be studied with higher sensitivity." [6]
Longer wavelengths result in deeper penetration, influencing the effective path length across the spectrum [2]. This near-surface sensitivity makes ATR-FTIR an ideal tool for studying immobilized nanoparticles on crystal surfaces.
Nanoparticle Immobilization on ATR Crystals
To examine surface adsorption, nanoparticles are immobilized as a thin, uniform layer on the ATR crystal. This setup provides a stable platform for studying the liquid–solid interface during solution exchanges [1]. By ensuring that observed spectral changes directly correspond to adsorption events, this technique underscores ATR-FTIR’s value in nanoparticle surface analysis.
Imali A. Mudunkotuwa and her team at the University of Iowa describe this approach:
"ATR-FTIR spectroscopy facilitates monitoring these dynamics by interrogating a layer of immobilized nanoparticles coated on the ATR element while changing the overlying aqueous phase." [1]
Other methods include depositing thin metal films, such as gold, onto the crystal surface to investigate molecular adsorption. Alternatively, species like lipid vesicles can spontaneously adsorb from a bulk solution, enabling real-time kinetic studies. The choice of crystal material also plays a key role; durable options like diamond and silicon are often preferred for their resistance to abrasion and harsh conditions [4].
Once nanoparticles are securely immobilized, ATR-FTIR can then measure molecular interactions on these surfaces with precision.
Detecting Molecular Adsorption and Dynamics
After immobilization, ATR-FTIR tracks how molecules interact with the nanoparticle surface by detecting changes in specific infrared frequencies associated with chemical bonds. These shifts in vibrational bands reveal details about adsorption modes, ligand conformations, and displacement reactions in real time [1].
For instance, a 2017 study by Wolfgang G. Hinze, Mohammad A. Fallah, and Karin Hauser at the University of Konstanz explored protein adsorption on zinc oxide (ZnO) films using a flow-through system. The researchers synthesized ZnO films on silicon ATR substrates and introduced Bovine Serum Albumin (BSA) solutions at concentrations ranging from 0.25 to 1 mg/mL, flowing at 0.48 mL/min. Over 120 minutes, they observed a two-step adsorption process by analyzing the amide I and amide II bands (around 1650 cm⁻¹ and 1550 cm⁻¹). When the system was flushed with pure water for another 120 minutes, BSA on ZnO-coated surfaces retained 92% of its signal, compared to just 65% on plain silicon. This showed that ZnO provided stronger binding sites [6].
ATR-FTIR also distinguishes between different types of surface complexes. Inner-sphere complexes, where molecules form direct chemical bonds with the nanoparticle surface, produce distinct spectral signatures compared to outer-sphere complexes, which are separated by a water layer [8]. These real-time measurements highlight ATR-FTIR’s versatility in studying surface interactions, making it invaluable for understanding catalytic processes, designing drug delivery systems, and predicting nanoparticle behavior in environmental or biological contexts.
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
ATR FTIR Basics | Attenuated Total Reflectance | Principles of Infrared Spectroscopy
Recent Studies on Nanoparticle Surface Adsorption Using ATR-FTIR
Recent advancements in ATR-FTIR spectroscopy have provided valuable insights into how molecules interact with nanoparticle surfaces, revealing distinct adsorption behaviors and mechanisms.
Ligand Adsorption on TiO2 Nanoparticles
A team at the University of Iowa, led by Vicki H. Grassian, used ATR-FTIR to study how TiO2 nanoparticles interact with biological media. Their 2014 research, published in Analyst, highlighted how the surface composition of these nanoparticles changes based on the surrounding medium – an observation critical to understanding nanotoxicity [1]. Through spectral analysis, they identified adsorption modes, ligand displacement reactions, and conformational changes [1].
Further research at UC San Diego examined how coatings of bovine serum albumin (BSA) and fulvic acid (FA) influence TiO2 reactivity. Published in Environmental Science: Nano in 2020, the study demonstrated that uncoated TiO2 promotes the photooxidation of sodium benzoate into hydroxyl benzoate. However, a BSA coating significantly suppressed this reaction by scavenging reactive oxygen species (ROS). FA coatings also reduced ROS generation, though less effectively than BSA. These findings highlighted the role of inner- and outer-sphere mechanisms in modifying surface reactivity based on the medium [9][3].
An IUPAC Technical Report from 2019 provided further insights into adsorption mechanisms. Oxalate ions were found to form inner-sphere bidentate bonds with TiO2, altering their infrared spectra. In contrast, perchlorate ions exhibited outer-sphere adsorption, with spectra nearly identical to their free solution form. As the report notes:
"the IR spectrum of an outer-sphere adsorbed species is almost identical to that of its free solution form, while the IR spectrum of an inner-sphere adsorbed species is generally altered from that in solution by the molecular perturbation of covalent bonding" [3]
Additionally, nucleotides, the building blocks of DNA, were observed to bind preferentially to TiO2 via their phosphate groups, rather than through ribose or nitrogenous bases [10]. These findings on TiO2 provide a foundation for exploring similar behaviors in other nanoparticle systems.
Adsorption Intermediates in CO2 Reduction on CuO Nanoparticles
ATR-FTIR has also been instrumental in studying the redox behavior of copper-based nanoparticles, particularly Cu/CuxO core–shell structures. M. K. Schröter and colleagues used the technique to monitor oxidation states during CO adsorption [11]. Their findings revealed the formation of oxide shells (CuxO) and their reversible reduction back to metallic Cu⁰ upon CO exposure, even in ligand-stabilized particles. The team observed:
"small molecules penetrate the ligand shell of the nanoparticles and reversibly adsorb at the surface without affecting the particle morphology and size distribution" [11]
By combining experimental IR data with Density Functional Theory (DFT) calculations, the researchers identified different coordination modes, such as monodentate versus bidentate binding, on mineral surfaces [10]. This approach has been widely cited, including in studies on CO2 hydrogenation [11]. ATR-FTIR also enabled real-time tracking of reaction kinetics and detection of dynamic intermediates – such as formate and bicarbonate – critical for understanding catalytic processes [1].
Organic Molecules on Hematite Nanoparticles
Studies on hematite nanoparticles have offered additional insights into molecular interactions. For instance, research on nucleotide adsorption showed that deoxyadenosine monophosphate (dAMP), dGMP, dCMP, and dTMP all bind preferentially through their phosphate groups [10]. The binding modes differ depending on the mineral surface – hematite tends to favor monodentate coordination, while goethite supports bidentate bridging [10]. ATR-FTIR also detected surface-induced deprotonation, such as the loss of the 1,710 cm⁻¹ N1H⁺ feature in dAMP upon adsorption to hematite, a change not observed with dCMP [10].
In another study, organoarsenical interactions with hematite were compared. Dimethylarsinate (DMA) exhibited a mix of outer-sphere and monodentate adsorption, while inorganic arsenate (iAs) formed more stable bidentate binuclear complexes. iAs adsorbed 2.5 times faster and desorbed four times slower than DMA, reflecting its stronger binding affinity. The activation energy for iAs bidentate complexes was also three times higher than that for outer-sphere DMA [12].
Quantitative measurements using ATR-FTIR have further determined maximum surface concentrations for molecular layers. For example, the maximum surface concentration was reported as 7.17 × 10⁻¹³ mol/cm² for BSA and 6.1 × 10⁻¹³ mol/cm² for anti-PSA antibodies [7]. These findings demonstrate the precision of ATR-FTIR in quantifying adsorption phenomena.
sbb-itb-aa4586a
Comparing ATR-FTIR Results Across Nanoparticle Studies
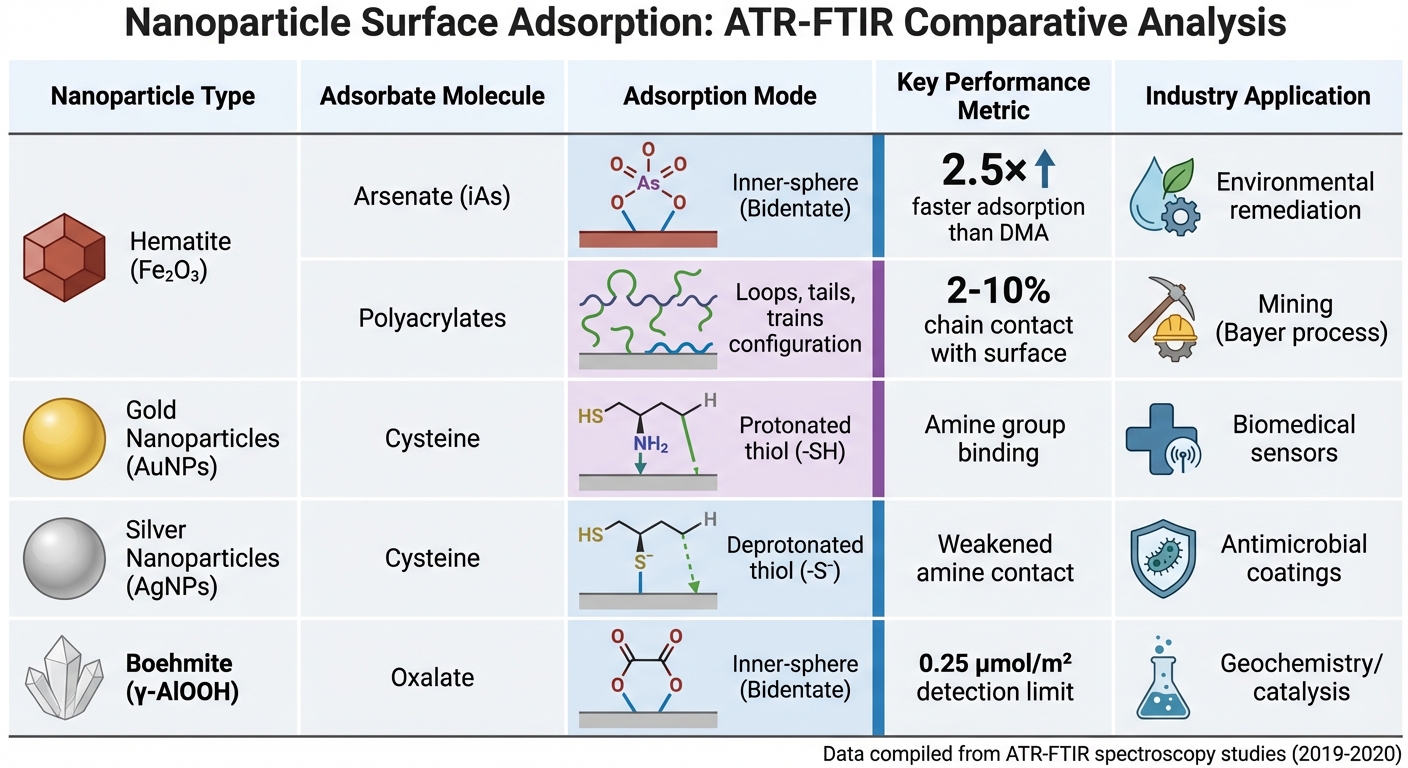
ATR-FTIR Nanoparticle Adsorption Comparison: Key Metrics Across Materials
Key Parameters and Trends
Building on earlier discussions of adsorption mechanisms, recent research highlights how ATR-FTIR enhances our understanding of nanoparticle surface interactions. For instance, studies have shown that inorganic arsenate (iAs) forms bidentate binuclear complexes on hematite, adsorbing 2.5 times faster and desorbing four times slower compared to dimethylarsinate (DMA), which primarily binds via outer-sphere interactions [12].
The influence of pH on adsorption is another critical factor. For example, BSA adsorption on hematite peaks near pH 5, aligning closely with the protein’s isoelectric point. Research from the Parker CRC for Integrated Hydrometallurgy Solutions revealed that only 2–10% of polyacrylate chains interact with hematite surfaces, adopting a "loops, tails, and trains" configuration [15].
Differences in metal-specific adsorption are also noteworthy. Work from AGH University of Science and Technology demonstrated that cysteine deprotonates its thiol group (-S⁻) when adsorbed on silver nanoparticles (AgNPs), while it remains protonated (-SH) on gold nanoparticles (AuNPs) [13].
| Nanoparticle | Adsorbate | Adsorption Mode | Key Metric | Industry Application |
|---|---|---|---|---|
| Hematite (Fe₂O₃) | Arsenate (iAs) | Inner-sphere (Bidentate) | 2.5× faster than DMA | Environmental remediation [12] |
| Hematite (Fe₂O₃) | Polyacrylates | Loops, tails, trains | 2–10% chain contact | Mining (Bayer process) [15] |
| Gold (AuNPs) | Cysteine | Protonated thiol | Amine group binding | Biomedical sensors [13] |
| Silver (AgNPs) | Cysteine | Deprotonated thiol | Weakened amine contact | Antimicrobial coatings [13] |
| Boehmite (γ-AlOOH) | Oxalate | Inner-sphere (Bidentate) | 0.25 μmol/m² detection limit | Geochemistry/catalysis [17] |
Experimental parameters also play a role in ATR-FTIR results. For example, the choice of ATR crystal material – such as zinc selenide (ZnSe) or germanium (Ge) – affects penetration depth and band intensities due to their differing refractive indices. These factors highlight the importance of standardizing conditions when comparing adsorption behaviors across various nanoparticle systems.
Applications Across Industries
The insights gained from these adsorption studies have direct implications for a range of industries. In mining and hydrometallurgy, understanding polyacrylate adsorption on hematite is critical for improving the Bayer process, where flocculants help separate red mud waste from valuable alumina [15].
Environmental remediation efforts benefit from identifying adsorption modes. For example, the stronger and more persistent binding of inorganic arsenate to iron oxide surfaces can inform the design of more effective arsenic-removal filters for water treatment facilities [12].
In the pharmaceutical and biomedical fields, protein adsorption studies guide the development of biocompatible materials. Research has shown that BSA loses its α-helix structure – dropping from around 47% in solution to 30–35% when adsorbed on hematite – providing valuable insights for predicting protein behavior on implant surfaces [14]. Similarly, the enhanced adsorption of BSA on positively charged nanoceria informs the design of cerium oxide nanoparticles for drug delivery applications [16].
ATR-FTIR also supports advancements in chemical manufacturing and catalysis. By enabling real-time tracking of reaction kinetics and intermediates, it aligns with Quality by Design (QbD) principles, helping to identify Critical Process Parameters during processes like polymerization and hydrogenation [18]. Additionally, surface-enhanced infrared absorption (SEIRA) techniques, which use rough metal surfaces, amplify signals by factors of 10¹ to 10³, making it possible to detect trace concentrations critical for process control [13]. These examples illustrate how ATR-FTIR insights contribute to better nanoparticle surface adsorption analysis, driving advancements across industries.
Disclaimer: This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Conclusion and Future Directions
Key Takeaways
ATR-FTIR has proven to be a powerful tool for studying nanoparticle surface interactions directly in their natural environments. Recent research highlights its ability to characterize the liquid–solid interface in real time, offering insights into adsorption behaviors, ligand conformational changes, and ligand displacement reactions as they occur [1].
In the biopharmaceutical field, ATR-FTIR is used to monitor critical parameters such as protein concentration, aggregation, and chemical modifications. Impressively, it can detect surfactants down to 0.3 ng and trace species at levels as low as 100 ppm [19][20][5]. These established applications pave the way for exciting advancements in the technique’s capabilities.
Future Opportunities
The future of ATR-FTIR is poised to bring even greater precision and versatility to nano-scale analysis. Emerging innovations, such as Nano-FTIR (which integrates Atomic Force Microscopy with infrared spectroscopy) and SR-FTIR (leveraging synchrotron radiation), are pushing the boundaries of resolution and sensitivity. These advancements enable detailed chemical mapping at the level of individual molecules or bacteria – a milestone demonstrated in 2020 by L. Mester and colleagues at the Catalysis Research Centre [21].
In addition, advancements in detector technology are enhancing the precision of ATR-FTIR measurements. High-resolution detector arrays now allow for the simultaneous capture of thousands of spectra, which facilitates high-throughput imaging of protein distribution and stability [20]. When combined with analytical tools like Principal Component Analysis (PCA) and Partial Least Squares (PLS), these systems can unravel the complexities of mixtures and reaction dynamics. As noted by Bernadette Byrne and Sergei G. Kazarian:
"In-line ATR-FTIR and NIR spectroscopy can be incorporated into a feedback loop to reduce the presence of contaminants or aggregates, therefore reducing the time and monetary costs of production" [20].
Looking ahead, integrating ATR-FTIR with hybrid sensor systems – such as those combining Raman spectroscopy, Laser Backscattering, and Focused Beam Reflectance Measurement – offers exciting possibilities. These combined approaches could provide a more detailed and comprehensive understanding of multi-phase systems, benefiting fields like catalysis, nanomedicine, and advanced materials development. Such innovations are expected to play a critical role in advancing industrial chemical applications [4]. As these technologies evolve, ATR-FTIR is set to remain a key technique in the next wave of nanomaterial research.
Disclaimer: This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
FAQs
How does ATR-FTIR identify differences between inner-sphere and outer-sphere complexes on nanoparticle surfaces?
ATR-FTIR is a valuable technique for identifying inner-sphere and outer-sphere complexes by examining how the adsorbed ligand vibrates. Inner-sphere complexes, where the ligand forms a direct chemical bond with the nanoparticle surface, typically display noticeable changes in the frequencies and intensities of functional group peaks.
On the other hand, outer-sphere complexes rely on electrostatic interactions or water-mediated adsorption. These interactions generally cause little to no frequency shifts and often preserve water-related vibrational bands. This ability to differentiate between the two types of complexes makes ATR-FTIR an essential tool for investigating molecular-level surface adsorption processes.
What makes ATR-FTIR effective for studying nanoparticle surface adsorption in real time?
ATR-FTIR is an excellent method for examining how nanoparticles interact with surfaces, particularly because it allows non-destructive, real-time monitoring at the liquid-solid interface. This capability makes it possible to observe critical processes as they happen, including adsorption behavior, molecular interactions, structural changes, and ligand exchanges.
By offering a close-up view at the molecular level, ATR-FTIR provides researchers with a deeper understanding of nanoparticle behavior in various environments. This makes it a key technique for advancing studies in surface chemistry and materials science.
How does the material of the ATR crystal influence the analysis of nanoparticle surface adsorption?
The material of the ATR crystal is crucial when studying surface adsorption on nanoparticles. To achieve the best results, the crystal’s refractive index should be slightly higher than that of the sample, ensuring the optimal depth of the evanescent wave.
High-refractive-index crystals, such as diamond, are prized for their exceptional chemical durability and broad spectral range. However, they produce a shallower evanescent wave, which can reduce sensitivity to surface interactions. In contrast, lower-index crystals allow the wave to penetrate deeper, making them more effective for detecting surface-adsorbed species.
The choice of crystal material largely depends on the sample’s chemical properties and the specific adsorption phenomena under investigation. For instance, diamond is a great option for demanding applications, while other materials might be better for capturing more delicate surface interactions.





Comments are closed