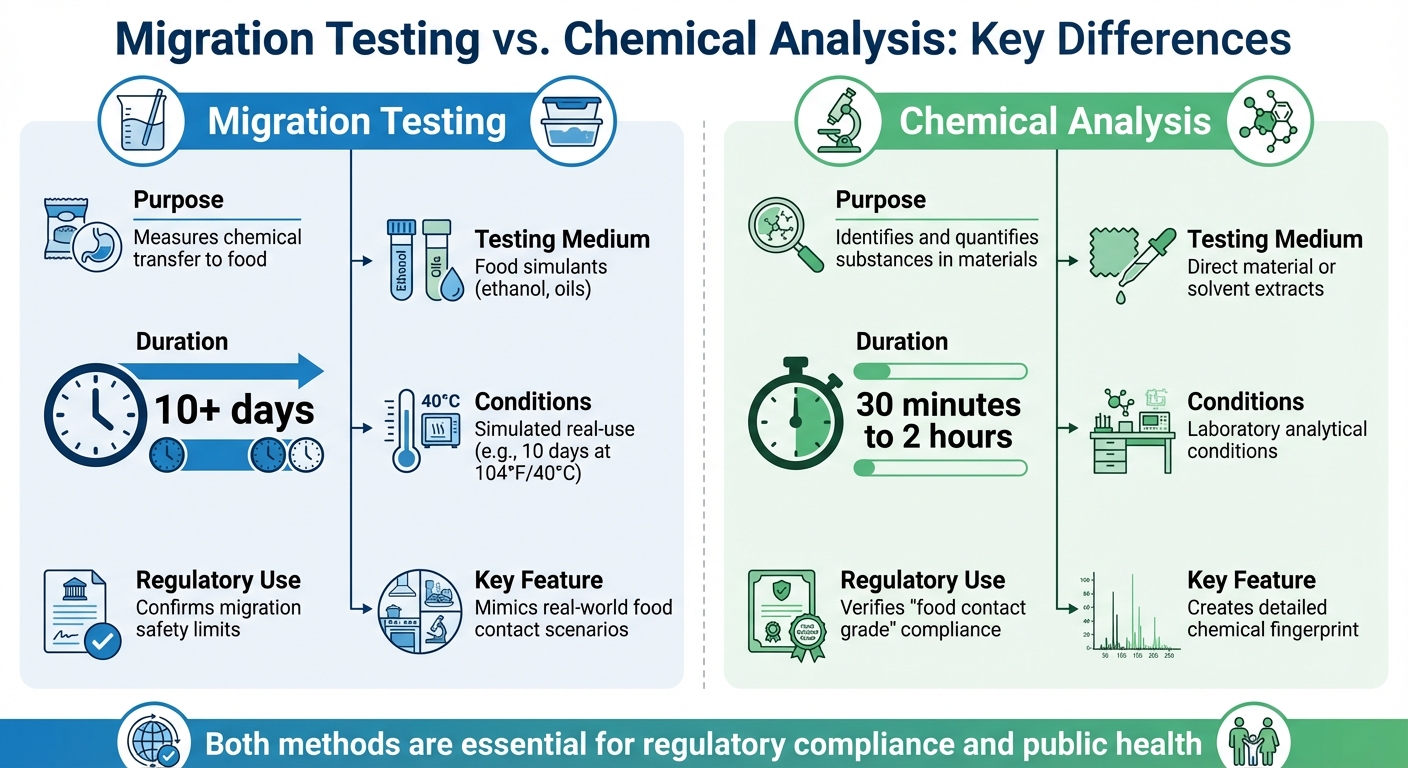
Migration testing and chemical analysis are two distinct methods used to ensure food packaging safety. Migration testing evaluates how much of a substance transfers from packaging into food under simulated real-use conditions, helping to estimate consumer exposure. In contrast, chemical analysis identifies and measures the substances present in the packaging material itself, focusing on its composition and purity.
Key Points:
- Migration Testing:
- Simulates real-world food contact scenarios.
- Uses food simulants (e.g., ethanol, oils) to measure chemical transfer.
- Typically takes 10+ days under controlled conditions.
- Focuses on consumer safety by assessing dietary exposure.
- Chemical Analysis:
- Examines the material’s chemical profile directly.
- Identifies substances like monomers, impurities, and degradation products.
- Shorter test duration (30 minutes to 2 hours).
- Ensures material purity and compliance with regulatory standards.
Quick Comparison:
| Factor | Migration Testing | Chemical Analysis |
|---|---|---|
| Purpose | Measures chemical transfer to food | Identifies and quantifies substances in materials |
| Testing Medium | Food simulants (e.g., ethanol, oil) | Direct material or solvent extracts |
| Duration | 10+ days | 30 minutes to 2 hours |
| Focus | Consumer exposure | Material composition and purity |
| Regulatory Use | Confirms migration safety limits | Verifies "food contact grade" compliance |
Both methods are essential for regulatory compliance and public health, ensuring that food packaging materials are safe for use.
Migration Testing vs Chemical Analysis: Side-by-Side Comparison
Overall migration | Αnalysis in one minute
What is Migration Testing?
Migration testing plays a critical role in ensuring the safety of food contact materials by measuring the transfer of chemicals from packaging into food. This testing, required by regulations, assesses whether materials are safe for food contact under conditions that mimic real-life use [1]. By providing data on chemical migration, it helps manufacturers and regulators evaluate consumer exposure to packaging components through diet. The following sections explain the purpose, testing methods, and regulatory guidelines for migration testing.
Purpose of Migration Testing
The main goal of migration testing is to verify that food contact materials meet safety and compliance standards. This involves measuring two types of migration: total migration (all non-volatile substances) and specific migration (individual chemicals) [1]. Agencies like the FDA mandate these tests to support toxicological risk assessments, ensuring that any substances transferring into food remain within safe limits.
- Overall migration evaluates the total amount of non-volatile substances migrating into food.
- Specific migration focuses on individual chemicals or groups, such as BPA or phthalates, which may pose specific health risks [1].
This dual approach ensures a thorough evaluation of potential risks from packaging materials.
Testing Conditions and Simulants
Since testing with actual food can be complex and costly, migration experiments use food simulants – solvents that mimic the behavior of different food types [1][8]. For instance:
- Aqueous and acidic foods (e.g., water, fruit juice): Tested with 10% ethanol.
- Fatty foods (e.g., butter, oils): Tested with vegetable oil or synthetic triglycerides like Miglyol 812 [2].
Testing conditions are designed to replicate the most extreme scenarios the packaging might encounter, such as high temperatures or extended storage times. For example, a standard accelerated test of 10 days at 104°F (40°C) simulates 6–12 months of storage at 68°F (20°C) [2]. To ensure consistency, the ratio of simulant volume to packaging surface area is standardized at 10 mL per square inch [2]. These tests are conducted under strict regulatory protocols to ensure reliable results.
Regulations for Migration Testing
In the United States, migration testing is governed by FDA Title 21 CFR and the Food Contact Notification program [1][9]. The FDA requires the use of food simulants that accurately represent the migration behavior seen with actual food [2]. In the European Union, standards such as DIN EN 1186 (for overall migration from plastics) and DIN EN 13130 (for specific migration limits) set the framework for compliance [1]. These regulations ensure that food packaging meets consistent safety standards, regardless of where products are manufactured or sold.
Disclaimer: This content is for informational purposes only. Always consult official regulations and qualified professionals when making sourcing or formulation decisions.
What is Chemical Analysis?
Chemical analysis is the process of identifying and measuring substances in food contact materials. It looks at the composition of packaging materials, including the primary food contact substance (FCS), residual starting materials like monomers and solvents, catalysts, and any impurities or degradation products [2]. This method creates a detailed chemical fingerprint of the material, offering a complete chemical profile that complements migration testing and supports thorough safety evaluations.
Purpose of Chemical Analysis
While migration testing simulates how materials interact with food, chemical analysis focuses on the source material itself. It provides the full chemical profile of food contact materials, which is crucial for regulatory and safety assessments. The main objective is to characterize the food contact substance by identifying its chemical name, CAS Registry Number, and molecular structure [2]. For polymers, this includes measuring molecular weights such as weight average (Mw) and number average (Mn), as well as physical properties like melting point and solubility [2]. Beyond the primary substance, chemical analysis also quantifies major impurities – such as residual monomers, solvents, catalysts, by-products, and degradation products – that could migrate into food [2]. This data is essential for toxicological risk assessments, as it helps estimate consumer dietary exposure and ensures it stays within safe limits [3].
Analytical Techniques Used
Chemical analysis relies on highly sensitive instruments capable of detecting substances at parts per billion (ppb) or parts per trillion (ppt) levels [11]. Gas chromatography-mass spectrometry (GC-MS/MS) is commonly used to detect volatile and semi-volatile organic compounds like residual solvents and phthalates. Liquid chromatography-mass spectrometry (LC-MS/MS) is ideal for analyzing non-volatile substances, such as large oligomers and additives like bisphenol A (BPA). For unknown contaminants and non-intentionally added substances (NIAS), advanced methods like UHPLC-Q-Orbitrap-MS and UFLC-MS/MS are increasingly used to identify complex compounds [8][12]. Additional techniques, including infrared (IR), ultraviolet (UV), and nuclear magnetic resonance (NMR) spectroscopy, help with structural characterization and identity verification [2].
Regulations for Chemical Analysis
Validated analytical methods form the backbone of the regulatory framework for food contact materials. In the U.S., 21 CFR Parts 174–179 outline standards for chemical analysis [10]. According to Section 409 of the Federal Food, Drug, and Cosmetic Act (FD&C Act), any substance intended for use in food contact materials must be proven safe for its intended purpose [2][7]. Chemical analysis plays a critical role in Food Contact Notifications (FCN) and Food Additive Petitions (FAP) submitted to the FDA [2][3].
Under the Threshold of Regulation (TOR) in 21 CFR 170.39, substances with dietary concentrations at or below 0.5 ppb may be exempt from full food additive petitions [2][10]. Analytical methods must comply with the FDA’s validation guidelines, which set performance standards for accuracy, precision, and detection limits (LOD and LOQ) [6]. Additionally, safety studies and chemical analyses submitted to the FDA must adhere to Good Laboratory Practice (GLP) regulations under 21 CFR 58 to ensure the reliability of the data [7].
Disclaimer: This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
sbb-itb-aa4586a
Key Differences Between Migration Testing and Chemical Analysis
Let’s break down the main distinctions between migration testing and chemical analysis, two approaches that play unique roles in assessing the safety of food contact materials. Migration testing mimics real-life scenarios, measuring how much of a substance transfers from packaging into food under specific conditions of time and temperature. On the other hand, chemical analysis focuses on identifying and quantifying the substances present in the material itself, creating a detailed chemical profile without simulating food contact. As Keller and Heckman LLP explains:
"Compliance testing can differ significantly from ‘migration testing,’ which is done prior to clearance to determine whether a food contact substance migrates to the food itself" [4].
Comparison Table: Migration Testing vs. Chemical Analysis
| Factor | Migration Testing | Chemical Analysis |
|---|---|---|
| Primary Purpose | Evaluate consumer exposure and safety [1][2] | Identify substances, impurities, and purity [1][2] |
| Testing Medium | Food simulants (e.g., 10% ethanol, oils) [1][2] | Direct material or solvent extracts [1][2] |
| Duration | Long-term (typically 10+ days) [4] | Short-term (30 minutes to 2 hours) [4] |
| Conditions | Simulated real-use (e.g., 10 days at 104°F) [2][4] | Laboratory analytical conditions [1][2] |
| Analytical Focus | Amount of migrants in food/simulant [1] | Chemical structure, molecular weight, and purity [2] |
| Regulatory Use | Confirm compliance with migration limits [1] | Verify "food contact grade" and material identity [1][4] |
This table highlights the distinct purposes and methodologies of each approach. Now, let’s explore how these differences translate into their practical applications.
Real-Use Conditions vs. Substance Identification
The core distinction lies in what each method measures. Migration testing simulates the actual conditions a food package might encounter – whether it’s frozen storage, room temperature, or microwave heating. For example, accelerated conditions like 10 days at 104°F are used to replicate long-term storage. This method helps determine how much of a substance could migrate into food, providing a clear picture of dietary exposure.
Chemical analysis, in contrast, examines the material itself before any food contact occurs. It identifies residual components such as monomers, catalysts, and degradation products to ensure the raw materials meet regulatory standards. This is particularly important because a substance might exist in the packaging but never transfer into food at levels that pose a safety concern. Migration testing ensures exposure levels remain safe, while chemical analysis confirms the material’s identity and purity [1][4].
Disclaimer: This content is for informational purposes only. Always consult official regulations and qualified professionals when making sourcing or formulation decisions.
When to Use Migration Testing vs. Chemical Analysis
Factors That Determine Testing Method Selection
Choosing between migration testing and chemical analysis depends on several factors, including regulatory requirements and the specific properties of the materials involved. Regulatory guidelines, such as those from the FDA and international standards, play a major role in determining the testing method. For example, overall migration limits and end-use tests often dictate the approach [1][5]. The intended application of the material also matters – a high-temperature environment (like retort pouches or microwaveable containers) typically demands migration testing, while packaging for frozen foods may only require chemical analysis to confirm material purity [2].
The material’s composition is another critical consideration. Paper migration is largely influenced by solubility, whereas migration in plastics is governed by diffusion. For polymers, testing is usually performed on the lowest molecular weight version of the material to simulate a worst-case migration scenario [2]. Additionally, dietary exposure thresholds guide whether further testing is necessary [7].
The nature of the substance being tested also impacts the decision. Migration testing is appropriate for substances with well-defined toxicological limits. On the other hand, chemical analysis, particularly non-targeted screening, is essential for identifying unknown contaminants or Non-Intentionally Added Substances (NIAS) [1][13]. Often, manufacturers begin by calculating a worst-case scenario assuming 100% migration. If the estimated exposure is below 0.5 ppb, further testing may not be required [2][7].
Examples of Testing Applications
In August 2019, FDA researchers explored accelerated migration testing for epoxy can coatings. Their findings showed that cyclo-di-BADGE migration into 50% ethanol under accelerated conditions (140°F for 10 days) resulted in 314 µg/dm². This closely matched the 325 µg/dm² observed after 490 days at 104°F. This study demonstrated that a 10-day test at 140°F could effectively simulate 1.5 years of shelf-life storage, with an acceleration factor of 49 [8].
Chemical analysis is particularly useful for quality control of materials already regulated. For instance, "wet-end" paper additives – those not strongly bound to paper fibers – are evaluated through specification testing. The FDA assumes that 98% of these substances remain in the process water, with only 2% transferring to the finished product [5]. Similarly, biocides, which are inherently toxic, require safety testing at reduced exposure levels [7].
These examples highlight how the choice between migration testing and chemical analysis is guided by practical considerations and regulatory requirements.
Disclaimer: This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Conclusion: Choosing the Right Approach for Food Contact Safety
Key Takeaways
When it comes to ensuring food contact safety, migration testing and chemical analysis each play a distinct role. Migration tests, which typically span over 10 days, use tools like GC, LC, and MS to measure how much of a substance transfers into food under specific conditions. These tests are crucial for pre-market approval and assessing dietary exposure. On the other hand, chemical analyses, which usually take 30 minutes to 48 hours, focus on quality control by identifying and quantifying substances in materials that have already passed regulatory clearance[4].
The choice between these methods depends on factors such as regulatory requirements, the status of the substance, and dietary exposure levels. For substances already listed in a Food Additive Regulation or Food Contact Notification (FCN), compliance tests ensure they meet established specifications. For new substances, migration testing provides the critical data needed to determine if the material could act as a food additive under FDA oversight. Testing needs are often guided by the Cumulative Estimated Daily Intake (CEDI); substances with exposure below 0.5 ppb may not need further studies, while those above 1 ppm generally require a full Food Additive Petition[7].
Starting with worst-case scenarios can streamline the process. Manufacturers often assume 100% migration when calculating dietary exposure. If this estimate results in a concentration below 0.5 ppb, additional lab testing might not be necessary[2][7]. These standard assumptions help validate exposure estimates and optimize resources[2].
By applying these principles, industry stakeholders can ensure a reliable chemical supply to support effective testing protocols.
Supporting Industry Needs with Trusted Chemical Sourcing
Implementing these testing strategies requires dependable access to high-quality chemicals, and Allan Chemical Corporation is well-positioned to meet these needs. With over 40 years of experience in regulated industries, Allan Chemical provides analytical-grade and compendial-grade solutions (USP, FCC, ACS, NF) tailored to food safety testing. These materials are essential for both migration studies and chemical analysis, helping laboratories and manufacturers maintain compliance with FDA and international standards.
Allan Chemical ensures just-in-time delivery and competitive pricing, enabling efficient testing schedules without compromising on quality. Whether you need simulants for migration testing, reference materials for analysis, or technical guidance for developing methods, Allan Chemical’s expertise and ISO 9001-certified systems provide reliable access to the tools necessary for food contact safety evaluations. Their commitment to bridging regulatory requirements with practical testing solutions reinforces the food and beverage industry’s dedication to consumer safety.
Disclaimer: This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
FAQs
What are the key regulatory guidelines for migration testing and chemical analysis of food-contact materials?
In the United States, FDA Food Additive Regulations (21 C.F.R. § 170–190) establish the rules for migration testing and chemical analysis of food-contact materials. These regulations are designed to ensure that any substance potentially migrating into food is either authorized, exempt, or awaiting approval. Safety evaluations consider the combined status of all components involved.
When it comes to migration testing, the FDA focuses on two key areas: overall migration (measuring total non-volatile substances) and specific migration (analyzing individual compounds). Standards such as DIN EN 1186 are often used for plastics testing. Although the U.S. does not specify an overall migration limit, testing must follow rigorous protocols, frequently drawing on international standards to maintain consistency.
For chemical analysis, methods must meet the FDA Foods Program Validation Guidelines, which define criteria like accuracy, precision, and detection limits. These requirements ensure that analytical procedures are scientifically sound and dependable. Allan Chemical Corporation plays a crucial role by supplying technical-grade reagents and standards that manufacturers rely on to comply with these stringent regulations.
What are the differences between migration testing and chemical analysis in ensuring food packaging safety?
Migration testing and chemical analysis work hand in hand to ensure the safety of food packaging materials. Migration testing focuses on measuring how much of a substance moves from packaging into food, ensuring compliance with safety limits set by regulatory bodies like the FDA and EU. This step is crucial for identifying potential risks and safeguarding food from harmful chemical exposure.
Meanwhile, chemical analysis dives deeper by pinpointing and quantifying the specific substances that migrate. These can include monomers, oligomers, or even unintended chemicals. This detailed data helps evaluate risks, uncover unexpected compounds, and determine safe exposure levels for each substance.
By combining these approaches, food-contact materials are held to rigorous safety standards, ensuring public health is protected and product quality remains intact. Allan Chemical Corporation supports these efforts by supplying top-tier chemicals and standards tailored for migration testing and precise chemical analysis in regulated industries.
When should manufacturers use migration testing instead of chemical analysis?
Manufacturers should opt for migration testing when they need to determine how much of a substance moves from a food-contact material into food under realistic conditions. This testing plays a crucial role in confirming compliance with migration limits – whether specific or overall – and ensuring the material’s safety in its intended use.
In contrast, chemical analysis is better suited for identifying and measuring the substances present in a material, rather than evaluating their potential to transfer. Migration testing offers a more practical approach to assessing consumer safety, making it the go-to method when regulatory or safety concerns are tied to real-world applications.





Comments are closed