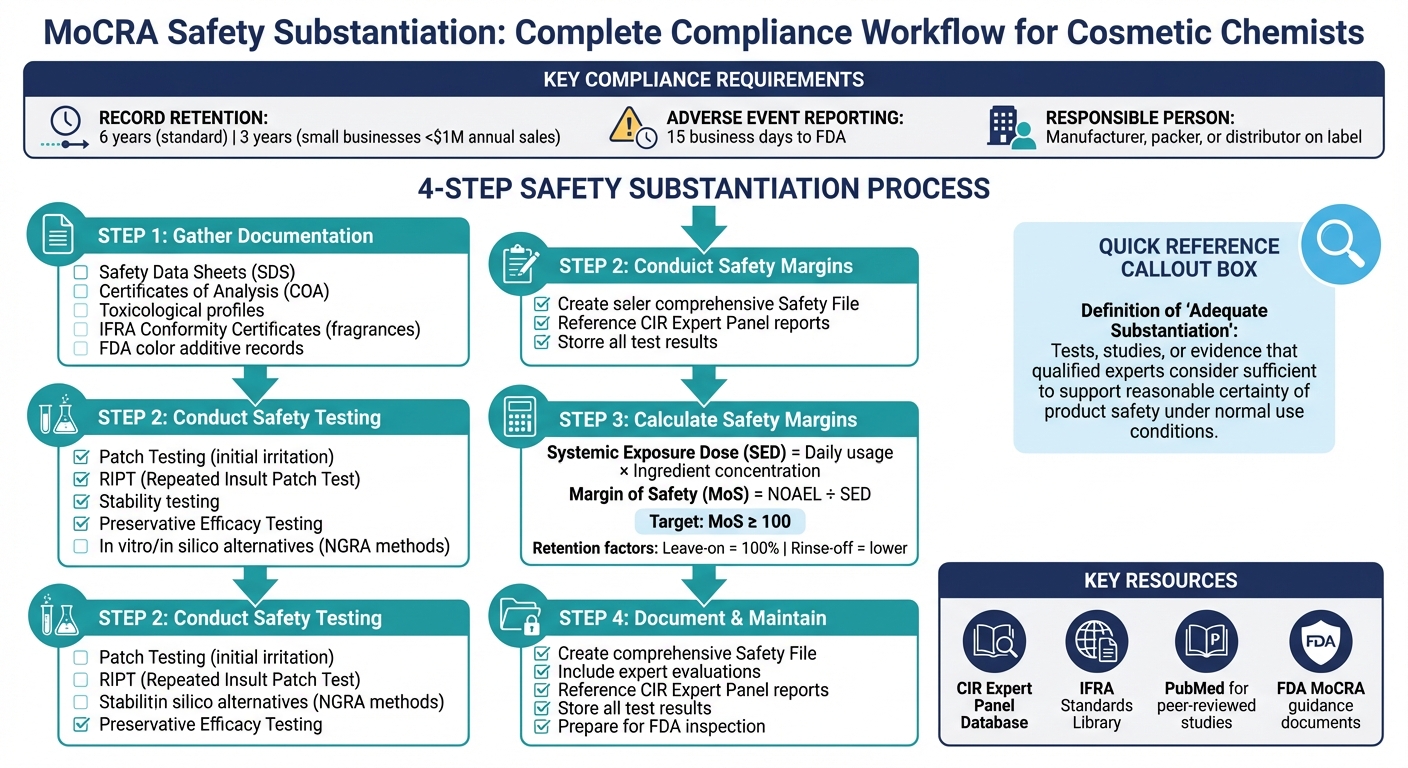
The Modernization of Cosmetics Regulation Act (MoCRA), signed into law on December 29, 2022, is the most comprehensive update to U.S. cosmetics regulations since 1938. It mandates that cosmetic products must be scientifically proven safe before reaching consumers. Key requirements include maintaining detailed safety records, ensuring proper labeling, and reporting adverse events to the FDA within 15 business days. Manufacturers, distributors, and brand owners (collectively known as the "Responsible Person") are legally accountable for compliance.
What does this mean for chemists?
You’ll now need to provide robust safety documentation, including toxicological testing, ingredient safety data, and cumulative exposure assessments. This ensures that products are safe under normal conditions of use. If you’re a small business with annual sales under $1 million, some exemptions apply, but safety substantiation is still required.
Key Compliance Steps:
- Collect Safety Data Sheets (SDS), Certificates of Analysis (COA), and toxicological profiles for ingredients.
- Conduct required tests like irritation, sensitization, and stability testing.
- Keep safety records for 6 years (3 years for small businesses).
- Report serious adverse events promptly to the FDA.
This regulatory shift emphasizes science-backed safety to protect consumers and hold companies accountable. Chemists must now integrate these practices into product development and documentation processes.
MoCRA Compliance Workflow for Cosmetic Chemists
Practical Steps to MoCRA Compliance: The Quick & Easy Way
sbb-itb-aa4586a
MoCRA Safety Substantiation Requirements
MoCRA’s safety substantiation rules emphasize the importance of scientifically validated claims, ensuring that products meet safety standards before reaching the market. This section outlines what constitutes adequate evidence and the responsibilities for compliance.
What Constitutes Adequate Scientific Evidence?
Under MoCRA, “adequate substantiation of safety” is defined as scientifically valid tests, studies, analyses, or other evidence that experts – such as toxicologists – deem sufficient to establish a "reasonable certainty" of a product’s safety [6][8]. This definition allows chemists some flexibility in their approach, but the evidence must withstand scientific scrutiny.
"Adequate substantiation of safety is defined as: tests or studies, research, analyses, or other evidence or information that is considered, among experts qualified by scientific training and experience to evaluate the safety of cosmetic products and their ingredients, sufficient to support a reasonable certainty that the product is safe." – Section 608 of MoCRA [8]
Key elements of safety substantiation include:
- Toxicological testing: This covers irritation, sensitization (e.g., Patch Testing or Repeat Insult Patch Test/RIPT), and toxicity assessments [5][8].
- Ingredient safety data: Safety Data Sheets (SDS), Certificates of Analysis (COA), and toxicological profiles from suppliers are essential [6].
- Published research: Reports from the Cosmetic Ingredient Review (CIR) Expert Panel and peer-reviewed studies accessed through databases like PubMed can serve as valuable references [5][6][3].
- Product-specific testing: Tests like microbiological challenge testing (Preservative Efficacy Testing), stability testing, and physicochemical evaluations are critical for certain formulations [5][8].
- Alternative methods: When traditional testing isn’t practical, in vitro cell culture testing or computer modeling can provide predictive safety data [5].
- Regulatory compliance records: Documentation like IFRA Conformity Certificates for fragrances and FDA-approved color additive records ensures adherence to legal standards [6].
Before investing in costly new trials, consult the CIR Expert Panel database for existing evaluations that may support your safety claims. Additionally, always request IFRA certificates for fragrances and COAs for color additives to confirm compliance with FDA requirements [6].
Once the necessary data is gathered, it becomes the responsibility of the designated Responsible Person to document and maintain these records.
Responsible Person Obligations
MoCRA assigns clear accountability to the Responsible Person (RP), defined as the manufacturer, packer, or distributor whose name appears on the product label. This includes brand owners, contract manufacturers, and private-label sellers [6][9][1]. The RP is legally obligated to ensure all safety substantiation requirements are met.
"The Responsible Person for a cosmetic product must ensure and maintain records supporting that there is adequate substantiation of safety of such cosmetic products." – Section 608 of MoCRA [8]
The RP’s responsibilities extend beyond initial testing. They must:
- Maintain all safety substantiation records.
- Report serious adverse events to the FDA within 15 business days.
- Provide documentation for FDA review upon request [8].
A product is deemed "safe" if it does not harm users under normal or prescribed conditions of use. Minor, temporary reactions like slight skin irritation do not automatically classify a product as unsafe [3][8].
If you are the RP, establish a Standard Operating Procedure (SOP) for handling adverse event reports to ensure compliance with the 15-day reporting requirement. Include a domestic address, phone number, or website on your product label to allow consumers to report adverse events directly [9][8].
Required Documentation and Record-Keeping
To comply with MoCRA, maintain detailed records that demonstrate the safety of your products. These include:
- Ingredient safety data: SDS, COA, CIR reports, and IFRA certificates for every ingredient.
- Toxicological test results: Documentation of irritation, sensitization, and toxicity tests [5][8].
- Clinical and laboratory reports: Results from preservative challenge testing and stability testing to confirm safety during storage and use.
- Expert evaluations: Opinions from qualified toxicologists supporting a "reasonable certainty" of safety.
- External references: CIR Expert Panel reports and evidence of compliance with FDA regulations.
Records must be retained for 6 years, though small businesses with annual U.S. sales under $1,000,000 can keep records for 3 years [8][1][9]. The FDA has the authority to access and copy these records if there is reason to believe a product poses a health risk.
To streamline compliance, create a digital dossier containing all supplier documentation, test results, and expert evaluations. Use automated alerts to track updates and expiration dates [5]. Organizing these records effectively ensures readiness for FDA inspections.
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
How to Conduct Safety Assessments
To conduct a thorough safety assessment, you’ll need to analyze ingredients, calculate exposure levels, and evaluate toxicity risks. Every step must be documented to comply with legal safety standards. Start by identifying each ingredient using its INCI name and CAS number. Then, move on to exposure modeling and risk calculations to establish the product’s overall safety profile.
Ingredient Safety Testing Methods
Begin by reviewing your ingredient list and gathering Safety Data Sheets (SDS) and Certificates of Analysis (COA). These documents help verify ingredient purity and detect impurities, guiding the choice of appropriate safety tests.
Patch Testing is a standard method to assess initial skin irritation. This involves a single application under controlled conditions. For products designed for prolonged use, such as leave-on formulations, Repeated Insult Patch Testing (RIPT) is recommended to evaluate potential reactions from repeated exposure.
The industry is increasingly adopting New Generation Risk Assessment (NGRA) methods, which focus on ethical, non-animal testing alternatives. These include:
- In vitro testing, which uses lab-grown cell cultures to assess toxicity.
- In silico testing, which employs computer models to predict safety based on chemical properties.
Both approaches are scientifically validated and widely accepted.
To quantify safety, calculate the Margin of Safety (MoS). This is the ratio of the No-Observed-Adverse-Effect Level (NOAEL) to the exposure dose. A MoS of 100 or greater is generally considered a safe threshold for systemic exposure [4]. This calculation provides a clear safety margin between typical use and potential harm.
Once individual ingredient safety is confirmed, the next step is to assess cumulative exposure.
Cumulative Exposure Assessments
The Modernization of Cosmetics Regulation Act (MoCRA) mandates consideration of "cumulative or other relevant exposure" when evaluating product safety [4]. This acknowledges that consumers often use multiple products daily – typically 6 to 12 – leading to repeated exposure to the same ingredients from different sources [1].
To calculate the Systemic Exposure Dose (SED), multiply the daily product usage by the concentration of the ingredient in your formulation. For example, if a product contains 5% of an active ingredient, the systemic dose would reflect this percentage alongside the quantity applied daily. Retention factors are key here: leave-on products assume a 100% retention rate, while rinse-off products like cleansers have much lower retention since most of the product is washed away [4].
"In determining… whether a cosmetic product is safe, the Secretary may consider, as appropriate and available, the cumulative or other relevant exposure to the cosmetic product, including any ingredient thereof."
– Modernization of Cosmetics Regulation Act of 2022 (21 USC 364d) [4]
Pay special attention to sensitive populations, such as pregnant women, who may require stricter exposure limits due to potential systemic effects. When formulating products for these groups, adjust your safety calculations accordingly and document the reasoning behind your approach.
Using Existing Safety Data
Once exposure levels are determined, existing safety databases can save time and resources. Before conducting new tests, consult trusted sources like:
- The Cosmetic Ingredient Review (CIR) Expert Panel, which provides detailed monographs summarizing toxicological data and safety conclusions for thousands of ingredients.
- The International Fragrance Association (IFRA) Standards Library, which outlines maximum safe use levels for fragrance components and essential oils. It also offers Conformity Certificates to confirm compliance [6].
Additionally, peer-reviewed studies available through databases like PubMed and Google Scholar can provide valuable insights. If direct data for a specific ingredient is unavailable, consider read-across data from chemically similar substances with well-documented safety profiles [4].
Ensure all data is validated by qualified experts. Cross-check ingredient identifiers (INCI and CAS) with safety data to maintain consistency. Review COAs to confirm that your ingredients meet the same purity standards as those in the safety literature. Finally, compile all documentation into a comprehensive safety dossier. This should include ingredient safety data, IFRA certificates, color additive approvals, and any correspondence verifying ingredient composition [6].
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
Common Compliance Challenges and Solutions
Navigating MoCRA’s safety substantiation requirements presents several hurdles. Key challenges include sourcing compliant ingredients, staying updated on regulatory changes, and managing documentation within tight development timelines. Tackling these issues requires a well-thought-out, proactive approach.
Sourcing Compliant Ingredients
One major compliance challenge is obtaining reliable safety data from suppliers. Many global suppliers lack U.S.-specific documentation, leaving significant gaps that complicate safety validation [5][11]. Additionally, products developed before MoCRA often don’t meet the "adequate scientific evidence" standard now required, prompting the need for retrospective testing and new risk assessments [5]. Federal compliance also doesn’t guarantee state-level approval, as over 39 states have introduced their own regulations targeting hazardous cosmetic ingredients [10].
To address these issues, chemists should request detailed documentation from suppliers at the very start of a project. Collaborating with experienced specialty chemical providers – such as Allan Chemical Corporation, which offers thorough technical documentation – can simplify this process. These practices ensure a smoother adaptation to evolving regulatory demands.
Keeping Up with Regulatory Changes
The regulatory environment is shifting rapidly, with new guidance and deadlines emerging regularly. For example, the FDA’s report on PFAS, expected in December 2025, underscores how quickly requirements can change [1]. Chemists must also keep track of rulemaking timelines for Good Manufacturing Practices (GMP), fragrance allergen labeling, and talc testing [2].
To stay ahead, consider using digital tools to track FDA updates and maintain standardized documentation for quick adaptation [1]. Participating in professional training sessions and webinars can clarify complex topics, such as substantiation requirements and the 15-business-day reporting window for serious adverse events [1]. Consistent and organized record-keeping remains a cornerstone of compliance in this dynamic landscape.
Meeting Development Deadlines
Tight timelines add another layer of complexity to compliance efforts. Small companies, in particular, often lack in-house toxicology expertise. Rising costs of compliance and materials further strain resources [10]. Reformulating products to replace non-compliant ingredients can also impact texture, performance, or shelf life, leading to additional development cycles [10].
A tiered evaluation strategy can help save both time and money. Start with an in-depth literature review using resources like the Cosmetic Ingredient Review (CIR) Expert Panel reports, PubMed, and Google Scholar before committing to lab testing [4]. If an ingredient is well-documented, additional testing may not be necessary. Instead, rely on "read-across" data from chemically similar substances to support the safety of less-researched ingredients [4]. Calculating the Margin of Safety (MoS) early – aiming for a benchmark ratio of 100 or more – can confirm ingredient safety before finalizing formulations [4]. For more complex toxicological assessments or novel ingredients, partnering with accredited third-party labs can ease the scientific workload without delaying internal development [5].
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
Key Takeaways for Chemists
MoCRA Safety Substantiation Summary
The safety of cosmetic products is a top priority under MoCRA regulations. Every product must be proven safe before hitting the market. Under MoCRA, a "safe" product is one that doesn’t harm users under normal or expected conditions of use, even if minor, temporary reactions might occur [4][3]. The entity named on the product label, referred to as the Responsible Person, must maintain detailed safety substantiation records. The FDA has the authority to review these records during inspections and can issue mandatory recalls if a product is found unsafe [6][1]. As one expert put it: "Safety substantiation isn’t about red tape – it’s about knowing and having proof that your products are as safe as you believe they are" [6].
Compliance Best Practices
To meet safety substantiation requirements, chemists should adopt clear, systematic approaches to compliance. Begin every formulation with a thorough ingredient review, referencing CIR Expert Panel reports and IFRA Standards before moving to lab testing [4][6]. Use each ingredient’s INCI name and CAS number to simplify global compliance processes [4]. For safety evaluations, calculate the Margin of Safety (MoS) by dividing the Point of Departure (e.g., NOAEL) by the Systemic Exposure Dose (SED). Aim for an MoS of 100 or more [4]. Beyond ingredient safety, test the final formulation for stability, microbial contamination, and shelf life to ensure long-term safety [5][7]. Keep a complete "Safety File" with all supporting documentation [6][7]. Additionally, report any serious adverse events to the FDA within 15 business days [1]. Following these steps helps ensure your formulations meet regulatory requirements while maintaining safety.
Working with Reliable Suppliers
Reliable ingredient data from suppliers is essential for meeting MoCRA’s stringent standards. Collaborating with global partners can be tricky due to varying regulations [5]. To address this, request specific documentation at the start of every project, including Safety Data Sheets (SDS), Certificates of Analysis (COA), and IFRA Conformity Certificates for fragrance oils [6]. For complex ingredients like blends, preservatives, or trademarked items, insist on a full ingredient breakdown to identify every substance [6]. Allan Chemical Corporation supports compliance by providing detailed technical documentation and working with vetted manufacturers. Establishing clear agreements with suppliers ensures timely access to safety data and updates on any ingredient changes [5].
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
FAQs
What types of tests are needed to meet MoCRA’s safety substantiation requirements?
MoCRA doesn’t dictate specific tests for manufacturers to follow. Instead, it emphasizes the need for scientifically valid evidence – this could include tests, studies, research, or analyses – that proves a product is safe for its intended use. This evidence must meet rigorous standards to convince qualified experts and establish a reasonable certainty of safety.
To meet MoCRA’s requirements, manufacturers should rely on dependable methods and follow recognized industry practices. Partnering with skilled professionals or sourcing premium materials from reputable suppliers can make navigating compliance requirements more manageable.
How can small cosmetic businesses comply with MoCRA requirements on a limited budget?
Small cosmetic businesses can navigate MoCRA requirements by prioritizing key safety and labeling responsibilities. While companies with less than $1 million in annual revenue are exempt from full GMP compliance, FDA facility registration, and product listing, they are still required to ensure product safety, disclose allergen information, make truthful claims, and provide a way for consumers to submit complaints. This allows smaller companies to focus their efforts on building a strong safety substantiation file without the need for significant infrastructure investments.
A practical and budget-conscious strategy involves starting with a review of existing toxicology data for ingredients, estimating consumer exposure levels, and only commissioning additional testing if the available data proves insufficient. Collaborating with a MoCRA-compliant contract lab can also help reduce expenses compared to developing in-house testing facilities. With the FDA’s enforcement deadline extended to July 1, 2024, small businesses have extra time to complete their safety documentation and set up straightforward record-keeping systems.
Using pre-qualified, compendial-grade raw materials can simplify compliance even further. Allan Chemical Corporation offers USP/FCC-grade and technical-grade ingredients, complete with detailed safety documentation, making it easier for small businesses to save both time and resources while adhering to regulatory requirements.
What should I do if a serious adverse event is reported for a cosmetic product?
If a serious adverse event occurs, the responsible party must first verify that the incident aligns with the FDA’s definition of a serious adverse event as outlined under MoCRA. Once verified, the event must be reported to the FDA within 15 business days. This report should provide detailed information about the incident and include a copy of the product label. Submissions can be made using the MedWatch Form 3500A or through the FDA’s electronic reporting system.
If additional or significant medical information about the event is received within one year of the initial report, this information must also be sent to the FDA within another 15 business days. Timely reporting not only ensures compliance with MoCRA but also prioritizes consumer safety.





Comments are closed