HPLC pressure problems often stem from solvent-related factors, such as viscosity, impurities, or improper preparation. Viscosity impacts the force required to push solvents through the system, with higher viscosity leading to increased pressure. For example, Methanol generates more pressure than Acetonitrile under the same conditions. Impurities like air bubbles, particulates, or microbial growth can cause blockages, erratic pressure, or system instability. Proper solvent preparation, including filtration and degassing, is essential to prevent these issues.
Key takeaways:
- Viscosity Matters: Acetonitrile generates ~60% of the pressure of Methanol.
- Air and Contaminants: Air bubbles disrupt pressure; particulates clog columns.
- Preventative Steps: Use HPLC-grade solvents, filter through 0.45 μm filters, and degas properly.
- Maintenance: Regularly clean and replace filters, seals, and check valves to maintain system performance.
Understanding how solvents influence pressure and addressing common issues ensures reliable HPLC performance and reduces downtime.
PRESSURE PROBLEMS IN HPLC : A Step-By-Step Guide ׀ Top 5 Tips For Preventing HPLC Pressure Issues
How Solvents Affect HPLC Pressure
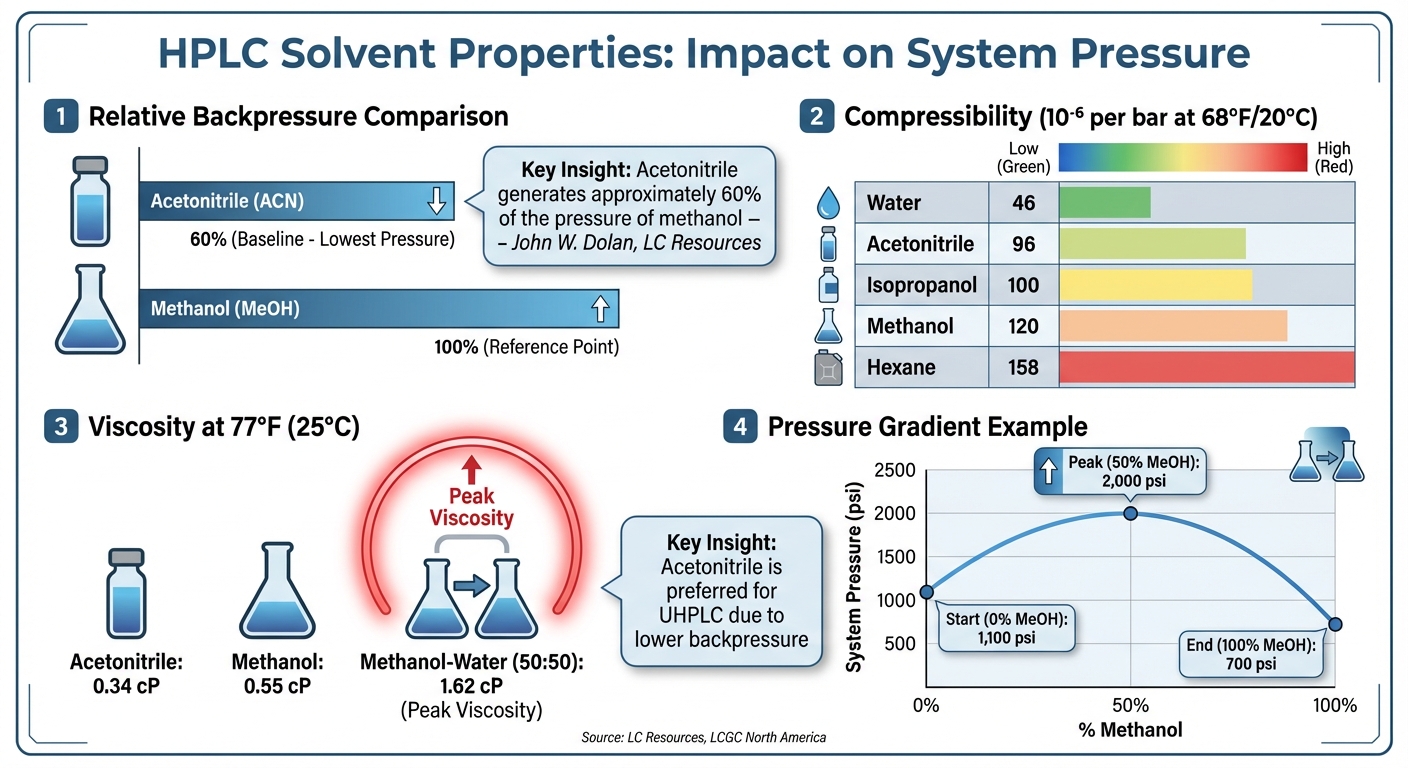
HPLC Solvent Properties Comparison: Viscosity, Compressibility, and Backpressure
Solvents play a major role in determining the pressure within an HPLC system. The key factor here is viscosity – thicker solvents require the pump to exert more force to push the mobile phase through the tightly packed particles in the column. Another factor to consider is compressibility, which becomes especially important at the elevated pressures typical in modern chromatography. Different solvents exhibit varying degrees of volume change under high pressure, influencing the system’s overall performance.
When running gradient methods, the viscosity of solvent mixtures doesn’t behave predictably. For instance, a 50:50 mix of methanol and water is more viscous than either solvent on its own, often causing a noticeable pressure spike mid-gradient. Temperature adjustments, such as using a column oven, can help reduce viscosity and alleviate backpressure. It’s also worth noting that doubling the flow rate results in a doubling of pressure, making solvent properties even more critical during gradient runs.
Solvent Properties That Affect Backpressure
Solvents differ significantly in the pressure they generate under identical conditions. For example, acetonitrile produces only about 60% of the backpressure that methanol does at the same flow rate, which makes it a popular choice for ultra-high-performance liquid chromatography (UHPLC), where pressure limits are a concern. John W. Dolan, Vice President of LC Resources, explains:
"Acetonitrile generates approximately 60% of the pressure of methanol, which is one reason it is favored for UHPLC mobile phases." [4]
Compressibility adds another layer of complexity to solvent behavior. Water compresses at approximately 46 × 10⁻⁶ per bar at 68°F (20°C), while methanol compresses nearly three times more at 120 × 10⁻⁶ per bar [6]. Acetonitrile and isopropanol fall in between, with compressibility values of 96 and 100, respectively [6]. These differences can create challenges for pumps trying to maintain consistent flow, especially if the system uses a default compressibility setting of 100. Using solvents with significantly different compressibility values can lead to baseline ripples and flow inconsistencies.
| Solvent | Compressibility (10⁻⁶ per bar at 68°F) |
|---|---|
| Water | 46 |
| Acetonitrile | 96 |
| Isopropanol | 100 |
| Methanol | 120 |
| Hexane | 158 |
Blending water with organic solvents like methanol or acetonitrile also impacts viscosity. Methanol-water mixtures reach their highest viscosity at 50% methanol (1.62 cP at 77°F), while acetonitrile-water mixtures peak at just 10% acetonitrile before steadily decreasing [4]. This explains why a gradient method transitioning from 0% to 100% methanol might start at 1,100 psi, rise to 2,000 psi at the midpoint, and drop to 700 psi at pure methanol [4]. Comparatively, pure acetonitrile at 77°F (25°C) has a viscosity of only 0.34 cP, much lower than methanol’s 0.55 cP, which further explains its lower backpressure [4].
Normal vs. Problem Pressure Changes
Understanding the difference between expected pressure changes and genuine issues can save significant troubleshooting time. In isocratic methods, pressure should remain stable, with only minor variations of about 1%. Any larger fluctuations often signal a problem [3]. In contrast, gradient methods naturally show pressure changes that reflect shifts in solvent viscosity throughout the run. Running a blank gradient (without sample injection) provides a baseline pressure curve for comparison [7].
Certain pressure changes indicate specific problems:
- Sudden spikes usually point to physical blockages, such as particulates clogging the inlet frit or precipitation from incompatible solvents.
- Erratic fluctuations resembling a sinusoidal pattern often result from air bubbles in the pump head or faulty check valves [7] [3].
- Gradual pressure increases over multiple injections often stem from chemical contamination or fines (broken packing particles) accumulating on the column inlet [2] [3].
- A complete pressure drop to zero is typically caused by a system leak or total pump failure [4] [1].
Establishing reference points is key to identifying abnormal pressure readings. Begin by measuring your "system reference pressure" by running the mobile phase through all components except the column. Then, record your "method reference pressure" with a new column installed [4] [5]. If pressure rises by 10–20% above the method reference, it’s time to investigate before the issue escalates.
Common Solvent Problems That Cause Pressure Fluctuations
Even the most advanced HPLC systems can experience pressure fluctuations if solvents aren’t prepared correctly. Issues like air bubbles, contamination, and incompatible solvent mixtures each leave distinctive pressure patterns, making it easier to identify the underlying problem.
Air Bubbles and Dissolved Gases
Air trapped in the HPLC system is a common culprit behind unstable pressure. This often happens when mixing aqueous and organic solvents. The combination reduces the overall solubility of air, creating a supersaturated solution. John W. Dolan, Vice President of LC Resources, explains this phenomenon:
"When aqueous and organic solvents are mixed… the solubility of air in the mixture is less than that of individual components… generating an unstable condition in which air will bubble out, or outgas, from the solution." [8]
When air reaches the pump head, it disrupts the system’s ability to maintain consistent pressure. Pumps are designed to compress liquids, not gases, leading to erratic pressure cycles or even a complete loss of prime. If these bubbles travel to the detector, they can scatter light, causing noise spikes or off-scale signals in UV and fluorescence systems [8].
To manage dissolved air, helium sparging is the most effective method, removing about 80% of air. Vacuum degassing removes 60–70%, while sonication is less effective, reducing air content by only 20–25% [8]. Additionally, placing solvent reservoirs below pump level or using restricted inlet tubing can lead to cavitation – vapor bubbles that form during the inlet stroke – further destabilizing pressure [9].
Air isn’t the only issue; particulate contamination and microbial growth can also wreak havoc on pressure stability.
Particulates, Microbial Growth, and Precipitation
Over time, contaminants in the solvent can gradually increase pressure. Particulates from unfiltered solvents tend to accumulate on inlet frits or column heads, creating blockages that restrict flow. Microbial growth is another serious issue, especially in systems using 100% aqueous solutions. Bacteria and algae can colonize inlet filters and degasser membranes, starving the pump of solvent [8] [9]. If contamination becomes severe, flushing the system with 30% phosphoric acid followed by water can help clear the pathways [8].
Precipitation of buffer salts occurs when the organic solvent ratio gets too high, forming crystals that damage pistons and seals. This can lead to visible leaks and pressure drift [9]. John W. Dolan, Editor of LCGC North America, highlights:
"A gradual increase in pressure over time is a normal symptom of column aging, and excessive pressure is often the first indicator that something is wrong with the system." [4]
To prevent these issues, adding 10–20% organic solvent (like methanol or acetonitrile) to aqueous mobile phases can inhibit microbial growth. Filtering all solvents through 0.5 µm filters protects seals and check valves, while avoiding the storage of buffers or water in degasser tubing when the system is idle helps prevent pore blockages [8] [9].
Solvent Mixing and Compatibility Problems
Improper solvent mixing introduces another set of challenges that can disrupt pressure stability. For instance, molecules that are soluble in the sample solvent may precipitate when they encounter the mobile phase, clogging frits and increasing pressure [3] [4]. While gradient transitions naturally cause some pressure variation due to changes in solvent viscosity, steep gradients or incorrect solvent ratios can lead to significant pressure spikes mid-run.
Buffer salts require extra care to ensure compatibility with organic solvent ratios throughout the gradient. If not managed properly, salts can crystallize, causing blockages and pressure fluctuations. Modern HPLC pumps are engineered to maintain pressure variations within about 1%, so any deviation beyond this range signals a problem that needs immediate attention [3].
sbb-itb-aa4586a
How to Diagnose Solvent-Related Pressure Problems
When pressure readings deviate from the expected range, a systematic approach is essential to determine if solvents are the root cause. Dwight R. Stoll, Professor of Chemistry at Gustavus Adolphus College, emphasizes the importance of recognizing anomalies:
"Recognizing that there is a problem usually amounts to recognizing that what is happening with the instrument is different from what is expected to happen" [3].
To troubleshoot effectively, compare the current pressure to your baseline and investigate potential causes step by step.
Check Solvent Quality and Preparation
Start by visually inspecting your solvents. Look for cloudiness or residues in the reservoirs, which may indicate salt precipitation or microbial growth. Aqueous mobile phases stored for over two weeks, particularly at mid-range pH levels, are prone to bacterial growth that can clog frits and columns [5]. If the mobile phase appears compromised, discard it and prepare a fresh batch.
Keep in mind that different solvents naturally produce varying levels of backpressure due to their unique properties. During gradient runs, mixtures of solvents can lead to pressure changes distinct from those seen with pure solvents [4].
Test HPLC Components One by One
If the solvents pass inspection, the next step is to systematically test each component of the HPLC system to identify any blockages. According to Restek Corporation:
"High back pressure in LC instruments is usually caused by foreign material blocking the flow of mobile phase" [5].
Begin at the detector and work backward toward the pump, testing each component individually to locate any obstructions [5] [2].
To check the pump inlet flow, disconnect the tubing and ensure that siphoning delivers approximately 10× the normal flow rate [4]. If the flow is restricted, the solvent reservoir filter may be partially clogged and should be replaced. This is a common cause of high-pressure issues [3]. Additionally, modern pumps should maintain pressure variations within 1%; fluctuations beyond this range could indicate air bubbles or a failing check valve [3].
By following this methodical approach, you can isolate the problematic component and address the issue effectively.
Read Pressure Patterns
Pressure patterns can offer valuable clues about solvent-related issues. John W. Dolan, Vice President of LC Resources, notes:
"Pressure readings that bounce around are usually the result of a faulty check valve or air in the pump" [4].
If the pressure fluctuates rhythmically with the pump stroke, air bubbles may be trapped in the pump head. To resolve this, open the purge valve and flush 5–10 mL of mobile phase at a high flow rate to remove the trapped air [4].
A gradual increase in pressure often signals column aging or the buildup of particulates [4] [2]. On the other hand, sudden pressure spikes may indicate salt precipitation caused by mixing incompatible solvents [2] [1]. A sharp drop in pressure could point to a leak, trapped air bubbles, or a restricted solvent inlet filter [3] [1]. Ronald E. Majors, Columnist for LCGC North America, advises:
"The best way to deal with particulates is to remove them before they reach the inlet frit" [2].
By carefully analyzing these pressure patterns, you can narrow down the cause of the problem and take the appropriate corrective actions.
Disclaimer: This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
How to Prevent HPLC Pressure Problems
Avoiding pressure issues in High-Performance Liquid Chromatography (HPLC) starts with thoughtful solvent selection and diligent system upkeep. By following key practices for solvent preparation and regular maintenance, you can prevent many of the common disruptions that pressure problems cause in analytical workflows.
Use High-Quality Solvents and Prepare Them Correctly
Using HPLC-grade solvents is essential for maintaining pressure stability. These solvents are prefiltered by manufacturers to minimize particulates, unlike lower-grade alternatives that may introduce contaminants into your system [2]. If you’re preparing your own buffers or solvent blends, always filter them through 0.45‑μm or 0.20‑μm filters to remove dust, salt crystals, and other particles that could clog the system’s inlet frit [2].
Degassing your mobile phases is another critical step. Bubbles can interfere with your results, so take advantage of modern systems equipped with in-line degassers. For an added layer of protection, consider vacuum filtration as part of your preparation routine [2][3].
For reversed-phase HPLC applications, acetonitrile is often a better option when pressure management is a concern [2]. However, always verify solvent compatibility before mixing. Check for cloudiness in high-organic mixtures, as salt precipitation from incompatible solvents can lead to blockages [2].
Once your solvents are prepared, proper storage and system maintenance are key to preventing pressure-related challenges.
Storage and Maintenance Guidelines
Proper storage practices can help prevent contamination that could disrupt system performance. Aqueous mobile phases, in particular, are prone to bacterial growth. To avoid this, prepare fresh solutions regularly and keep solvent reservoirs sealed. Using amber or opaque bottles can also protect light-sensitive solvents from degradation [3][5].
Routine maintenance is equally important. Replace pump seals every 6 to 12 months to prevent polymeric debris from clogging frits [4]. Keep an eye on in-line filters, replacing them whenever the pressure drop exceeds 10 bar [3]. As Dwight R. Stoll, a Professor of Chemistry at Gustavus Adolphus College, explains:
"In my laboratory, partially plugged filters account for 95% of our high-pressure problems" [3].
Regular system flushing can also help prevent buildup. When switching mobile phases, thoroughly purge the system to avoid mixing incompatible solvents. Recording a baseline system pressure using a standard column and mobile phase can provide a reference point for detecting abnormal pressure readings in the future [4].
These practices, combined with sourcing high-quality solvents, can significantly improve system reliability.
How Allan Chemical Corporation Supports Your Solvent Needs
Allan Chemical Corporation specializes in supplying high-purity solvents and technical-grade materials tailored for industries like pharmaceuticals and analytical laboratories. With over 40 years of expertise, they provide HPLC-grade chemicals and compendial-grade solutions (USP, FCC, ACS, NF) that meet strict quality standards. This helps prevent clogs and ensures consistent backpressure, keeping your system running smoothly. Their efficient delivery process ensures you’ll have access to fresh, high-quality solvents whenever needed, reducing downtime and supporting reliable analytical outcomes.
Disclaimer: This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Conclusion
Keeping HPLC pressure stable requires attention to solvent management and routine system maintenance. The foundation lies in three essential practices: selecting appropriate solvents, preparing them correctly, and performing regular upkeep on your system. Solvent viscosity plays a significant role in backpressure – acetonitrile, for example, produces about 60% of the pressure generated by methanol under comparable conditions [4]. This insight helps guide your choice of mobile phase for optimal pressure control.
Particulate contamination is the primary culprit behind pressure spikes and column blockages. To combat this, filter mobile phases using 0.45‑μm or 0.20‑μm filters to remove dust, salt crystals, and debris [2]. Additionally, thorough degassing minimizes air bubbles, ensuring smoother pump operation and consistent performance [10].
Routine maintenance is essential for preventing pressure-related problems. As Adam Dickie, Ph.D., emphasizes:
"Maintaining optimal pressure is crucial for achieving efficient separations and extending the lifespan of the HPLC system’s components" [10].
Documenting baseline pressure for each method helps you identify anomalies early on [4].
By using high-quality solvents, filtering and degassing consistently, and monitoring pressure trends, you can avoid most solvent-related issues. These practices not only ensure reliable results but also keep your HPLC system running efficiently over time.
Disclaimer: This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
FAQs
How does the viscosity of solvents affect HPLC system pressure?
The thickness, or viscosity, of a solvent plays a key role in determining the pressure within an HPLC system. Solvents with higher viscosity create greater resistance to flow, which raises backpressure. Over time, this can restrict flow rates, put stress on the system, and even lead to potential damage to the column.
To reduce these risks, choose solvents with viscosity levels suited to your method. Regular maintenance of the system is equally important to ensure smooth operation. Additionally, making gradual transitions between solvents with varying viscosities can help avoid sudden pressure increases.
What are the signs that solvents are causing pressure issues in HPLC systems?
Solvent-related pressure problems in HPLC systems can manifest in various ways, including fluctuating pump pressure, unstable or noisy baselines, and inconsistent retention times. Other signs might include peak broadening, the appearance of double or ghost peaks, or pronounced baseline noise.
These challenges often stem from issues like poorly prepared solvents, contamination, or the use of solvents that aren’t compatible with the system. To resolve these problems, it’s crucial to assess solvent quality, ensure proper filtration, and confirm that the solvents are suitable for the system in use.
How can I stop air bubbles from affecting my HPLC system?
Air bubbles in an HPLC system can wreak havoc by disrupting flow rates, lowering column efficiency, and generating noisy detector signals. To avoid these issues, it’s essential to degas your mobile-phase solvents. Use methods like vacuum degassing or helium sparging to remove dissolved gases effectively. Additionally, filter your solvents through a 0.2 µm membrane filter to remove particulates that could trap air and cause blockages.
Make sure all fittings and tubing are tightly secured to prevent leaks. After switching solvents, take the time to purge the pump and lines at a low flow rate. This helps flush out any trapped air. It’s also a good idea to maintain a steady solvent temperature of around 68°F (±2°F) to reduce the chance of out-gassing. For extra protection, consider adding a guard column or pre-column filter, which can catch any stray bubbles before they reach the analytical column. By following these practices, you can keep your HPLC system running efficiently and avoid air-related disruptions.





Comments are closed