Preservatives are essential in pharmaceuticals to prevent microbial growth and maintain product stability. They safeguard medicines, especially water-based and multi-dose products, from contamination during production, storage, and use. Without them, products risk degradation, reduced potency, and serious health hazards.
Key preservatives include:
- Parabens (e.g., Methylparaben, Propylparaben): Effective across a pH range of 4–9; commonly used in syrups, creams, and injectables.
- Benzalkonium Chloride: Ideal for ophthalmic solutions and nasal sprays; works well in pH 4–9.
- Benzyl Alcohol: Used in injectables; effective below pH 5.5.
Preservatives protect against risks such as microbial contamination from raw materials, water systems, and repeated product use. Proper selection involves considering the product’s pH, water activity, and microbial risk. Testing, including Antimicrobial Effectiveness Testing (AET), ensures safety and compliance with regulatory standards.
For reliable results, sourcing high-purity, compendial-grade preservatives from trusted suppliers is critical. This ensures consistency, safety, and adherence to regulations.
Why Preservatives Are Necessary
Where Microbial Contamination Comes From
Contamination can occur at any stage of a product’s lifecycle. Raw materials are often the first source of microbial threats, especially those derived from natural origins, which tend to carry higher microbial loads. Purified water systems are another major concern. Water, essential as a solvent, can become a breeding ground for Gram-negative bacteria such as Pseudomonas, which can colonize storage tanks and distribution pipes.
The manufacturing process itself introduces risks, including airborne particles, equipment surfaces, and transfer lines. If cleaning and sanitization are inadequate, biofilms can form on processing equipment, creating persistent contamination. Even packaging components can harbor microorganisms if not sterilized properly before use. Risks don’t stop at the factory door. Multi-dose containers – like eye drops, nasal sprays, and oral syrups – are particularly vulnerable, as repeated openings during patient use increase the chance of contamination through droppers, syringes, or exposure to the surrounding environment.
What Contamination Can Cause
Recognizing these contamination sources underscores the need for strict control measures. When microorganisms infiltrate a pharmaceutical product, the consequences can range from minor quality issues to severe health risks. Physical signs of spoilage, such as turbidity, color changes, unpleasant odors, or gas formation, indicate product failure. Beyond these visible effects, microbial enzymes can break down active ingredients and excipients, leading to a loss of potency. Additionally, bacteria and fungi can degrade preservatives, emulsifiers, and thickening agents, destabilizing formulations like emulsions and suspensions.
The most serious concerns involve patient safety. Contaminated products can cause local infections at the site of application, such as eye infections from ophthalmic drops, skin infections from creams, or respiratory infections from nasal sprays. If microbes or their toxins enter the bloodstream through injectable products, they can cause systemic infections, potentially leading to sepsis, hospitalization, or even death. Even when bacteria are killed, Gram-negative species leave behind endotoxins that trigger pyrogen formation, which can cause fever and severe inflammation. Over the years, contamination incidents have led to recalls, serious infections, and fatalities, highlighting the critical need for effective antimicrobial measures.
For vulnerable populations, such as immunocompromised patients, robust antimicrobial protection is particularly crucial.
Natural Protection vs. Added Preservatives
Some pharmaceutical products naturally resist microbial growth due to their composition. For example, low water activity in dry powders, tablets, high-sugar syrups, or hydrophobic ointments creates an environment unsuitable for microbial survival. Similarly, extreme pH levels or high concentrations of solvents like ethanol or propylene glycol can provide natural antimicrobial effects. When these factors are sufficient to inhibit microbial growth, products may meet safety requirements without added preservatives.
However, natural protection has its limits. Formulations that include water, are stored for long periods, or are repeatedly used from the same container generally require added preservatives. Changes such as dilution, pH shifts, or temperature fluctuations during storage or use can create conditions favorable to microbial growth. For multi-dose products like ophthalmic solutions, nasal sprays, oral liquids, and topical formulations, U.S. regulations often mandate preservatives unless manufacturers can demonstrate equivalent safety through alternative designs or controls. Added preservatives provide a measurable antimicrobial barrier that complements good manufacturing practices and can be regularly monitored through quality testing. This dual-layer approach ensures a higher level of safety, tailored to the specific risks of each product.
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
Preservation of Pharmaceuticals by Anti-microbial agents
Main Types of Preservatives and How They Work
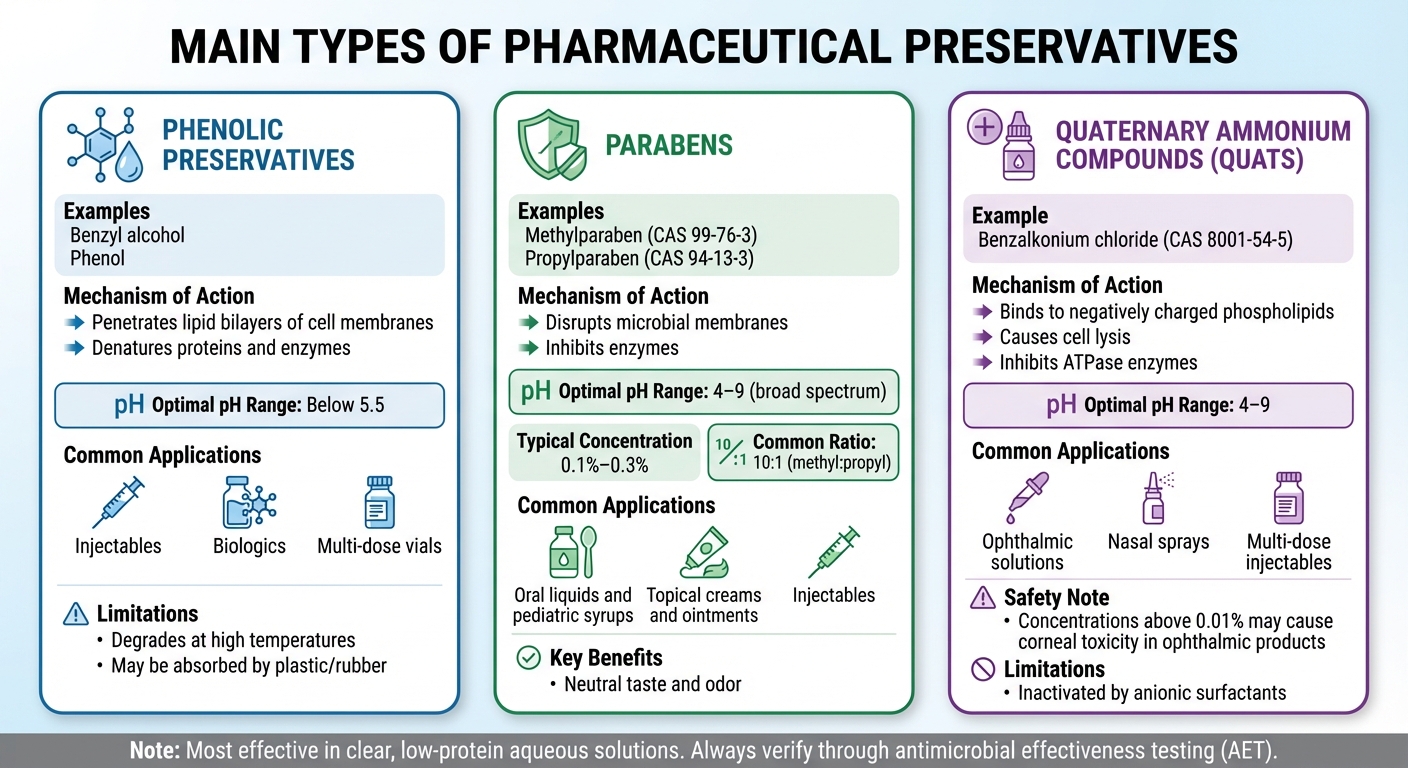
Main Types of Pharmaceutical Preservatives: Mechanisms and Applications
Pharmaceutical preservatives are categorized based on how they combat microbial cells, each with unique mechanisms. Knowing how these preservatives function is crucial for formulators to choose the right antimicrobial approach for their products. The most common types include phenolic compounds, parabens, and quaternary ammonium compounds. Here’s a closer look at how each works.
Phenolic Preservatives
Phenolic compounds, such as benzyl alcohol (used in injectables and biologics) and phenol (found in multi-dose vials), work through a two-pronged attack. They penetrate the lipid bilayers of microbial cell membranes, causing leakage of cellular contents, while also denaturing vital proteins and enzymes that microbes rely on to survive. This dual action makes them effective against bacteria and fungi in water-based formulations.[2][7]
However, phenolic preservatives come with some limitations. Benzyl alcohol is most effective at pH levels below 5.5 and can degrade when exposed to high temperatures during manufacturing or storage.[2][4] Additionally, phenolic compounds may be absorbed by plastic or rubber, reducing their effectiveness over time.
Parabens
Parabens, which are esters of p-hydroxybenzoic acid, provide broad-spectrum antimicrobial protection by disrupting microbial membranes and inhibiting enzymes. Methylparaben (CAS 99-76-3) is particularly effective against bacteria, while propylparaben (CAS 94-13-3) targets fungi more effectively.[2][4][3] These are often used together in a 10:1 ratio, with a total concentration of 0.1%–0.3%, to ensure broader activity.[3]
Parabens work well across a wide pH range (4 to 9), making them suitable for oral liquids (e.g., pediatric syrups), topical creams, ointments, and injectables.[2][4] Their neutral taste and odor are advantageous for oral formulations. However, their effectiveness can decrease in high-protein formulations, and there is ongoing regulatory scrutiny over potential endocrine disruption at higher concentrations.[2][4][5]
Quaternary Ammonium Compounds
Quaternary ammonium compounds, commonly known as "quats", are cationic surfactants with strong antimicrobial activity. Benzalkonium chloride (CAS 8001-54-5) is a widely used example in pharmaceuticals, frequently found in ophthalmic solutions, nasal sprays, and multi-dose injectables.[2][4] These compounds bind to negatively charged phospholipids in microbial membranes, leading to cell lysis. They also inhibit critical enzymes like ATPase, which microbes need for energy production and growth.[2][4][7]
Benzalkonium chloride is effective across a pH range of 4 to 9 and works particularly well against gram-positive bacteria and enveloped viruses. However, it can be inactivated by anionic surfactants or high organic content. In ophthalmic products, concentrations above 0.01% may cause corneal toxicity.[2][4][6] These preservatives are most effective in clear, low-protein aqueous solutions, underscoring the need for precise formulation to meet safety standards.
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
How to Select and Optimize Preservative Systems
Choosing the right preservative system for pharmaceutical products involves more than just picking an antimicrobial agent off a list. It requires a careful, step-by-step approach that balances microbial protection, formulation chemistry, patient safety, and regulatory compliance. Below, we’ll walk through the key considerations for selecting and fine-tuning preservative systems for U.S. pharmaceutical products.
Identify Product and Microbial Risk Profiles
Start by defining the product’s dosage form – whether it’s an oral liquid, topical cream, injectable, ophthalmic solution, or nasal spray – and its route of administration. These details set the stage for understanding regulatory requirements and narrowing down preservative options. For example, ophthalmic and parenteral products must meet stricter limits compared to oral liquids [4][7]. Multi-dose containers, particularly those frequently opened by patients (like eye drops or topical creams), demand stronger preservation than single-dose or sealed systems [4][7].
Next, assess the formulation’s water activity and nutrient content. Water-rich formulations like solutions, suspensions, creams, and gels pose a higher microbial risk compared to low-water systems like oily solutions or certain ointments [4]. Consider potential contamination sources, including raw materials (e.g., purified water, natural excipients), the manufacturing environment, packaging processes, and patient usage (such as tip contamination in eye drops). Identifying these risks ensures compliance with U.S. regulatory standards and enhances patient safety. Additionally, determine which microorganisms are most likely to pose a threat, such as gram-positive and gram-negative bacteria (e.g., Staphylococcus aureus, Pseudomonas aeruginosa), yeasts, and molds [4][6]. This microbial risk profile will guide your choice of antimicrobial spectrum and preservative strength.
Match Mechanisms to Formulation Conditions
The formulation’s conditions, particularly its pH, play a significant role in preservative selection and effectiveness. Weak acids like sorbic acid and sodium benzoate work best in acidic environments (pH 3.0–6.5), making them suitable for oral syrups and acidic topical products [2][4]. In contrast, parabens and quaternary ammonium compounds, such as benzalkonium chloride, remain active over a broader pH range (approximately 4 to 9), making them ideal for emulsions and neutral formulations [2][4].
Other formulation components also impact preservative performance. For instance, anionic surfactants or charged polymers can interact with cationic preservatives like benzalkonium chloride, reducing their free active concentration [4]. Similarly, nonionic surfactants, oils, and emulsifiers may sequester preservatives away from the aqueous phase – where microbes tend to thrive [4][7]. Adjusting pH, buffers, and excipient levels can help address these challenges, but it’s critical to verify preservative effectiveness through antimicrobial testing. Suppliers like Allan Chemical Corporation offer compendial-grade preservatives (USP, NF, FCC, ACS) along with documentation and technical support to simplify compatibility assessments and regulatory submissions. If single-preservative adjustments fall short, combining multiple agents may be necessary.
Combine Preservatives for Better Results
When a single preservative doesn’t provide adequate protection, combining agents with complementary mechanisms can be a game-changer. This approach not only enhances antimicrobial performance but also allows for lower concentrations of each preservative, reducing the risk of irritation or sensitization [4][3].
For instance, pairing methylparaben with propylparaben expands the antimicrobial spectrum [2][3]. Similarly, combinations like benzyl benzoate and benzyl alcohol or boric acid and benzalkonium chloride can extend activity across broader pH ranges [4]. These strategies help meet strict preservative-efficacy requirements while maintaining patient safety. Always validate these combinations through antimicrobial effectiveness testing, such as USP <51>, using the final container system. Packaging and formulation matrices can significantly influence preservative performance, so testing ensures the product remains protected throughout its shelf life.
This content is for informational purposes only. Always consult regulatory guidelines and qualified professionals when making formulation or sourcing decisions.
sbb-itb-aa4586a
Testing Performance, Stability, and Safety
Testing is the backbone of ensuring that your preservative system performs as intended. After selecting and fine-tuning a preservative strategy, rigorous evaluations are necessary to confirm its effectiveness, stability, and safety throughout the product’s shelf life. These tests fall into three key categories: antimicrobial effectiveness testing, stability and compatibility studies, and safety assessments. Together, they provide critical data for regulatory submissions and, most importantly, ensure consumer safety in the U.S. market.
Antimicrobial Effectiveness Testing
Antimicrobial effectiveness testing (AET), also known as preservative efficacy testing, is the standard method for verifying that your preservative system can effectively control microbial growth. In the U.S., this testing adheres to pharmacopeial protocols like USP <51>, which specify the organisms to test, conditions to use, and acceptance criteria based on the product type.
The process involves introducing known microbial strains – such as S. aureus, P. aeruginosa, E. coli, C. albicans, and A. brasiliensis – into the final packaged product. Microbial counts are then measured at intervals (days 7, 14, and 28). The requirements differ by product type and route of administration. For example, multi-dose ophthalmic and injectable products must meet stricter standards than oral liquids or topical creams. Typically, bacterial counts need to drop by at least 3 log units within 14 days, with no rebound. Testing is conducted at room temperature (20–25 °C) to reflect typical storage conditions. Importantly, AET must be performed both at product release and at the end of its shelf life, using the lowest expected preservative concentration, to confirm consistent protection over time [3]. Once antimicrobial efficacy is validated, the next step is to assess the preservative’s stability and compatibility.
Stability and Compatibility Studies
Preservatives can degrade, interact with other formulation components, or even be absorbed by packaging materials over time, which is why stability and compatibility studies are so critical. These studies are typically conducted under conditions that simulate both normal and accelerated aging, such as 25 °C/60% relative humidity for long-term testing and 40 °C/75% RH for accelerated testing.
Throughout these studies, preservative levels are monitored using analytical tools like HPLC, alongside other parameters such as pH, appearance, and microbial quality. For instance, preservatives like sorbic acid and its salts are particularly sensitive to environmental factors such as heat and alkalinity, which can hasten their degradation and reduce their antimicrobial activity. Additionally, packaging materials must be carefully evaluated to ensure they don’t absorb or otherwise compromise the preservative [4].
Safety and Tolerability Considerations
Even the most effective preservative system must prioritize patient safety. Regulatory guidance, toxicology data, and clinical experience establish safety thresholds for preservatives, which are reflected in resources like the FDA Inactive Ingredient Database. Testing with compendial-grade preservatives (e.g., USP, NF, or FCC grades) ensures compliance with strict quality standards, covering impurities, residual solvents, heavy metals, and microbial loads. Suppliers like Allan Chemical Corporation provide compendial-grade preservatives along with Certificates of Analysis, which streamline regulatory submissions and ensure batch-to-batch consistency.
Special attention is needed for vulnerable populations, such as infants, the elderly, and those with sensitivities. For example, benzyl alcohol has been linked to "gasping syndrome" in premature infants, leading to strict usage limits. Similarly, certain quaternary ammonium compounds and parabens are known to cause irritation or sensitization, prompting the use of lower concentrations or preservative-free options in sensitive applications like ophthalmic, pediatric, or injectable products. U.S. risk–benefit assessments consider factors like dose, frequency, and exposure route. In some cases, specific labeling – such as "discard after opening" instructions – helps minimize risks.
When combining preservatives to expand antimicrobial coverage, safety evaluations become more complex. Formulators in the U.S. typically start with the minimum effective concentration, validate combinations through AET and stability studies, and choose preservatives with a proven track record of clinical safety. Early collaboration with specialty suppliers provides access to crucial technical data – such as stability and impurity profiles – that can simplify the design of testing programs and support regulatory compliance [5].
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
Sourcing Preservatives from Specialty Suppliers
Once you’ve validated your preservative system, the next critical step is sourcing high-quality preservatives. The origin of these preservatives plays a key role in regulatory compliance, batch uniformity, and patient safety. After confirming efficacy and stability, obtaining quality preservatives becomes essential to ensure your product remains consistent and safe. Specialty chemical suppliers serve as a vital link between formulation testing and the final product, offering approved grades, comprehensive documentation, and expert guidance.
Why Compendial-Grade Preservatives Matter
Compendial-grade preservatives – those adhering to standards set by the USP (United States Pharmacopeia), NF (National Formulary), or FCC (Food Chemicals Codex) – are a must for pharmaceutical applications. These standards ensure the purity, potency, and consistency required to meet FDA guidelines for inactive ingredients. For instance, USP-grade parabens like methylparaben (CAS 99-76-3) and propylparaben (CAS 94-13-3) are validated for their broad-spectrum antimicrobial properties in oral liquids and topical formulations, typically used at concentrations of 0.1%–0.3% [2][4]. Using preservatives that don’t meet these stringent standards can lead to inconsistent performance, instability, or even recalls due to microbial contamination [2][4].
Compendial standards also ensure traceability and regulatory compliance throughout the supply chain. Suppliers provide essential documentation, such as Certificates of Analysis (CoAs), to support audit readiness and compliance – especially crucial for multi-dose products like injectables and ophthalmics, where preservative performance undergoes intense scrutiny.
Advantages of Partnering with Specialty Suppliers
Beyond the importance of compendial-grade preservatives, working with specialty suppliers offers several additional benefits. These suppliers bring sourcing expertise, regulatory support, and reliable logistics to the table. For example, Allan Chemical Corporation, with over 40 years of experience in regulated industries, provides just-in-time delivery, competitive pricing, and adherence to strict quality standards [2]. Their products often exceed compendial requirements, meeting certifications such as ACS, USP, NF, FCC, Kosher, and Halal [1].
Specialty suppliers also offer valuable technical resources, including stability data, compatibility studies, and impurity profiles, which help streamline testing and speed up time to market. When selecting a supplier, prioritize those with a proven track record in the pharmaceutical sector, robust documentation (like CoAs and Safety Data Sheets), and the capability to source specialized preservatives. Reliable partnerships with such suppliers ensure access to high-quality materials, such as USP-grade benzalkonium chloride for ophthalmic solutions or potassium sorbate for sugar-based suspensions, all while meeting the stringent traceability and quality standards required by regulatory bodies [2][4].
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
Conclusion
Preservatives play a critical role in safeguarding pharmaceutical products from microbial contamination, ensuring their integrity across a variety of dosage forms – such as multi-dose injectables, oral liquids, and topical treatments. By disrupting cell membranes or inhibiting enzyme systems, these agents help protect both patients and products. Understanding the mechanisms behind each preservative allows formulators to strike the right balance between antimicrobial effectiveness and patient safety.
Creating an effective preservative system involves more than just choosing the right agent. Factors like microbial risk, pH levels, and ingredient compatibility must align. Combining preservatives can offer broader protection while keeping concentrations to a minimum. Validation through antimicrobial effectiveness testing and stability studies ensures the system remains reliable throughout the product’s shelf life and usage period. These steps are not just best practices – they are regulatory necessities that directly impact product approval and market success.
In addition to formulation, quality sourcing is key to maintaining long-term product integrity. Using compendial-grade preservatives (USP, NF, FCC) ensures the purity and consistency required for regulatory compliance. Working with specialized suppliers can provide the documentation, technical support, and dependable logistics that keep production running smoothly. For example, Allan Chemical Corporation supports manufacturers with pharmaceutical-grade materials, competitive pricing, and just-in-time delivery, all backed by stringent quality systems[2].
By combining rigorous testing protocols with trusted sourcing partnerships, pharmaceutical companies can build preservative systems that extend shelf life, minimize waste, and prioritize patient safety. The strategies outlined here offer a practical roadmap to meet the challenges of today’s regulatory landscape.
This content is for informational purposes only. Always consult official regulations and qualified professionals when making sourcing or formulation decisions.
FAQs
What should you consider when choosing a preservative for a pharmaceutical product?
When choosing a preservative for a pharmaceutical product, the top priority is ensuring antimicrobial effectiveness to keep the product free from contamination. Equally important is the preservative’s compatibility with the formulation, its stability during storage, and its safety for the intended use.
Additional considerations include complying with regulatory standards (like USP, FCC, or NF), maintaining effectiveness across the product’s pH range, and staying stable under different temperature conditions. Selecting the right preservative safeguards the product’s quality and helps ensure patient safety.
How do preservatives help keep pharmaceuticals safe and free from contamination?
Preservatives are essential in pharmaceuticals, serving to stop the growth of harmful microorganisms such as bacteria and fungi. As antimicrobial agents, they safeguard medications from contamination and spoilage, ensuring these products stay safe and effective for patient use.
By preserving the stability and quality of pharmaceutical products, these agents play a key role in protecting patient health while also extending the shelf life of medications when stored correctly.
Why are high-quality, compendial-grade preservatives essential in pharmaceuticals?
Using compendial-grade preservatives plays a key role in pharmaceuticals because they meet strict quality, safety, and efficacy standards outlined by recognized pharmacopeias such as USP, NF, and FCC. These preservatives are essential for preventing microbial contamination, which helps ensure that pharmaceutical products remain stable and safe throughout their shelf life.
By adhering to regulatory standards, compendial-grade preservatives not only support manufacturers in maintaining compliance but also safeguard patients by reducing the risks associated with lower-quality or contaminated ingredients.





Comments are closed