Plastic waste recycling is evolving beyond traditional methods. Depolymerization – a chemical recycling process – breaks plastics into their original monomers, offering higher-quality recycled material suitable for demanding uses like food packaging and medical devices. This article examines five key depolymerization methods:
- Photodriven Depolymerization: Uses light and additives like carbon quantum dots to break polymer chains at lower temperatures.
- Enzymatic Depolymerization: Employs enzymes to selectively degrade plastics under mild conditions.
- Thermochemical Depolymerization (Electrified Heating): Applies precise, pulsed heat to vaporize plastics without catalysts.
- Chemical Depolymerization Routes: Includes glycolysis, methanolysis, and hydrolysis to recover high-purity monomers.
- Quantum Dot-Assisted Depolymerization: Leverages carbon quantum dots for selective, light-driven recycling with minimal by-products.
Each method has unique benefits and challenges, from handling mixed waste to achieving high monomer yields. Below is a quick comparison of these techniques.
Quick Comparison
| Method | Operating Temperature | Monomer Yield | Key Strength | Main Limitation |
|---|---|---|---|---|
| Photodriven | 248–356°F | Up to 82% | Low energy use | Limited scalability |
| Enzymatic | 86–158°F | Variable | High selectivity | Slower, sensitive to contaminants |
| Thermochemical (Electrified) | ~1,112°F (pulsed) | ~43% | No catalysts needed | High energy demand |
| Chemical (Solvolysis) | 302–842°F | Up to 99.79% | High-purity monomers | Solvent handling required |
| Quantum Dot-Assisted | Ambient bulk | 43–95% | Minimal oxidized by-products | Light penetration challenges |
Depolymerization shows promise for improving recycling rates, but scalability and cost remain hurdles. Each method’s suitability depends on plastic type, contamination levels, and desired monomer purity.
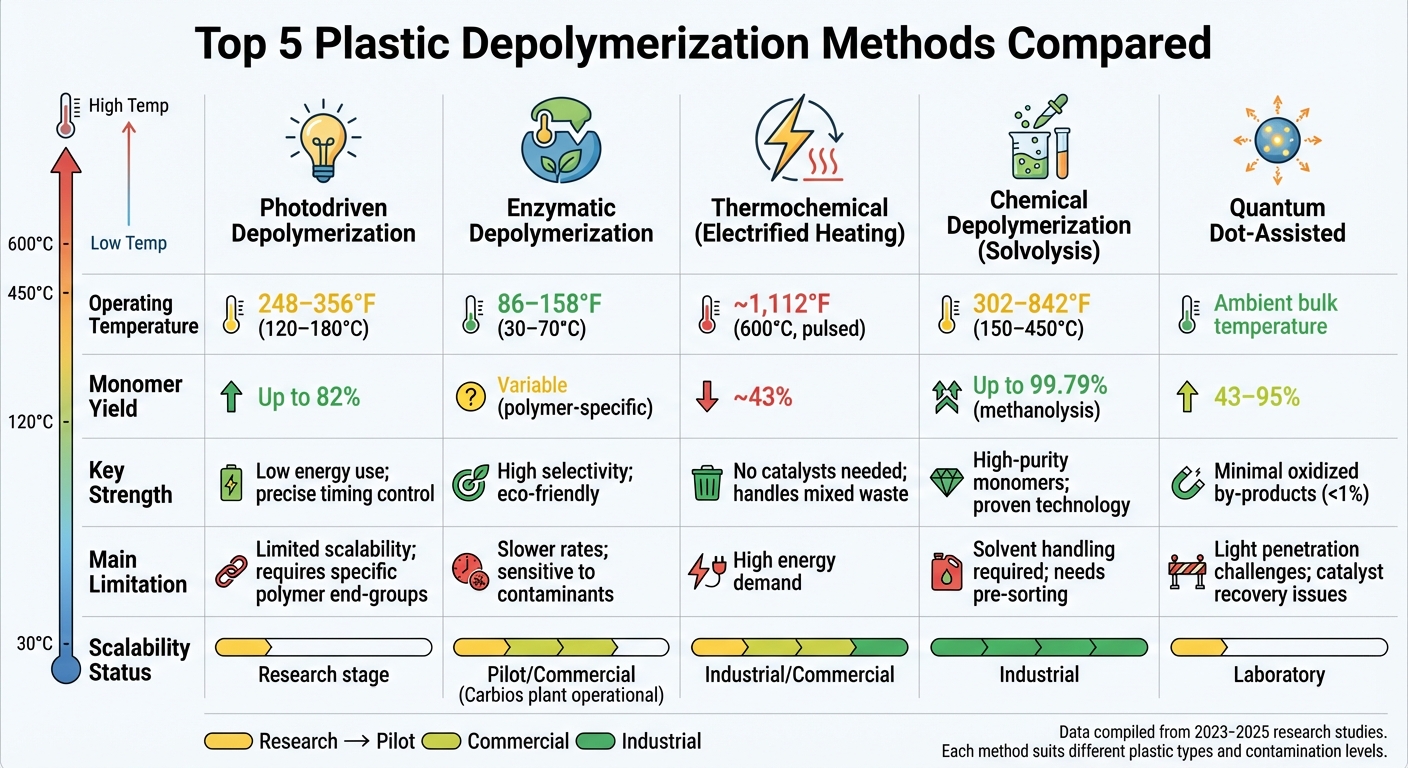
Comparison of 5 Plastic Depolymerization Methods: Temperature, Yield, and Scalability
1. Photodriven Depolymerization
Mechanism
Photodriven depolymerization uses light to generate radicals that "unzip" polymer chains. This process can occur through direct activation methods like photoiniferter or PET-RAFT, or via hydrogen atom transfer (HAT) mechanisms [3][5].
Photothermal effects also play a role when additives such as carbon quantum dots (CQDs) are introduced. These additives absorb photons and create localized "hot spots", pushing polymer chains to their ceiling temperature (Tc) – the point where depolymerization becomes favorable. Meanwhile, the bulk material remains cooler, minimizing unwanted side reactions [3]. Additionally, aromatic solvents like dichlorobenzene can undergo bond homolysis under light exposure, which facilitates the breaking of polymer main chains [5][6].
These mechanisms form the foundation for the improved efficiency metrics highlighted below.
Efficiency Metrics
Photodriven depolymerization methods significantly outperform traditional recycling techniques. For example, conventional PMMA recycling requires temperatures between 350–400°F (662–752°F), while photodriven approaches operate at much lower temperatures, typically 120–180°C (248–356°F) [5]. CQD-assisted processes also produce fewer oxidized by-products – less than 1% compared to 16% with conventional heating methods [3]. Additionally, chlorine radical-based depolymerization driven by visible light achieves monomer recovery rates exceeding 98% for polymethacrylates [6].
In early 2025, researchers successfully recycled consumer Perspex waste, including violet and clear laser-cut pieces, back into methyl methacrylate (MMA) monomers. Using UV irradiation in dichlorobenzene at 175°C for five hours, they achieved over 95% degradation into small molecules and more than 65% monomer conversion. The recovered monomers were then repolymerized into virgin-quality PMMA [5]. Later that year, a study published in Nature Communications demonstrated the use of the organocatalyst PTH-3CN for plastic upcycling. Remarkably, this method required catalyst loadings as low as 500 ppm, a significant reduction compared to earlier photocatalytic techniques [4].
Scalability Considerations
Although laboratory results are encouraging, scaling photodriven methods for industrial use presents challenges. One major issue is the reduced penetration of light through dense or opaque polymer mixtures, a limitation explained by the Beer–Lambert Law [3]. Real-world plastic waste often contains dyes and pigments that block UV and visible light, further lowering conversion efficiency [5]. Additionally, many effective photodriven processes rely on chlorinated aromatic solvents like dichlorobenzene, which pose environmental and safety risks at industrial scales [5][6].
Flow reactor designs offer a potential solution for these challenges. In May 2025, researchers demonstrated the upcycling of post-consumer Styrofoam into benzoic acid on a 10-gram scale using a flow reactor. This process required just 17.5 mg of the organocatalyst PTH-3CN and operated under visible light and ambient air conditions. Such advancements suggest a promising path for scaling the upcycling of mixed plastic waste [4]. Transitioning from UV to visible light–driven catalysts not only reduces energy consumption but also improves safety in large-scale operations [4][6].
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
sbb-itb-aa4586a
2. Enzymatic Depolymerization
Mechanism
Enzymatic depolymerization uses enzymes to break down plastic polymers at the molecular level. These biological catalysts work through two main pathways, depending on the type of plastic. In the hydrolytic pathway, enzymes like PETase, cutinases, and lipases target ester bonds in polyesters such as PET, polyurethane (PU), and polylactic acid (PLA). They achieve this by introducing water molecules to cleave the polymers into monomers [7][10]. On the other hand, oxidoreductases, including laccases and peroxidases, tackle more durable hydrocarbon-based plastics like polyethylene (PE) and polypropylene (PP). They do so by oxidizing the strong carbon–carbon bonds, creating reactive sites for further breakdown [7].
Some advanced enzymes take this a step further by unzipping semi-crystalline polyesters from their chain ends, a process known as chain-end-mediated processivity [8]. These enzymes often have specialized binding pockets. For instance, PETase is highly specific to PET’s aromatic terephthalate units. Additionally, some enzymes exhibit "trans-selectivity", meaning they prefer certain polymer chain conformations that result from pre-treatment processes like microwave heating [7][10].
These enzyme-driven reactions are notable for their ability to achieve high conversion rates under relatively mild conditions.
Efficiency Metrics
Unlike traditional recycling methods that require extreme heat, enzymatic processes operate at much lower temperatures. For example, pyrolysis demands temperatures around 500°C (932°F), whereas enzymatic reactions typically occur between 30–70°C (86–158°F) and can go up to 90°C (194°F) in some cases [13][12]. A noteworthy example from February 2024 demonstrated that Candida antarctica lipase B, stabilized in ionic liquids, fully depolymerized post-consumer PLA in just 24 hours at 90°C (194°F) [12]. Similarly, a 2021 study published in Nature found that embedding BC-lipase into polycaprolactone (PCL) and PLA enabled nearly complete conversion – up to 98% – into small molecules within days in standard compost conditions [8].
Scalability Considerations
While laboratory results are encouraging, scaling enzymatic depolymerization for industrial use remains a challenge. Enzymes primarily target the amorphous regions of polymers, making high-crystallinity materials like semi-crystalline PET bottles (≥30% crystallinity) harder to degrade [10]. Moreover, natural enzymes often lack the stability needed to perform under industrial conditions [10][11].
Cost is another significant factor. Traditional enzymatic processes can be expensive due to the need for pH control and the energy required for monomer recovery [14]. However, researchers at the National Renewable Energy Laboratory have made strides in addressing these issues. By using extrusion and quenching to amorphize substrates, they achieved near-complete conversion in just 50 hours. They also replaced standard buffers with ammonium hydroxide and adopted fed-batch processing, which reduced acid and base usage by over 99% and cut energy consumption for ethylene glycol recovery by 65%. These optimizations lowered annual operating costs by 74%, bringing the price of recycled PET down to $1.51 per kg compared to $1.87 per kg for virgin PET [14].
Innovative engineering solutions are gradually making enzymatic depolymerization more viable for large-scale applications.
Current Applications
In 2024, the French company Carbios opened the first industrial-scale PET biorecycling plant. By May 2025, its demonstration facility in France was processing two tons of PET waste daily, achieving monomer recovery with over 99% purity under mild conditions [9][7]. Advances in enzyme technology have further improved efficiency. For example, machine learning has been used to develop FAST-PETase, an enzyme capable of fully depolymerizing various post-consumer PET products at 50°C (122°F) within a week [11]. Additionally, directed evolution techniques have led to mutant enzymes with up to 30 times the activity of their natural counterparts [7].
While enzymatic depolymerization has made significant progress, challenges remain in degrading non-hydrolyzable plastics with carbon–carbon backbones, such as PE, PP, and polystyrene (PS) [9][10].
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
3. Thermochemical Depolymerization (Electrified Heating)
Mechanism
Electrified thermochemical depolymerization breaks down plastics by using precise temperature control without relying on catalysts. The STH (spatiotemporal heating) method employs a bilayer porous carbon felt, where the top layer is heated through Joule heating [15]. This setup delivers two important features that enable selective depolymerization:
- Spatial temperature gradient: Heat conducts from the top layer into the plastic, causing it to melt, wick into the porous structure via capillary action, and then vaporize [15].
- Temporal heating profile: Electrical current pulses create short, high-peak temperatures of about 1,112°F (600°C) followed by rapid cooling [16].
"This selective depolymerization process is realized by two features: [15] a spatial temperature gradient and [16] a temporal heating profile."
– Qi Dong, Aditya Dilip Lele, et al. [15]
These heating pulses last approximately 0.11 seconds – just enough time to break polymer bonds while avoiding unwanted side reactions like dehydrogenation, aromatization, or char formation. This controlled, far-from-equilibrium state eliminates the need for catalysts [23,24]. Such precision in heating dynamics directly influences the efficiency metrics discussed below.
Efficiency Metrics
Electrified spatiotemporal heating shows promising results in laboratory settings. For example, polypropylene (PP) achieves a monomer recovery yield of about 36%, while poly(ethylene terephthalate) (PET) reaches approximately 43% [15]. In April 2023, researchers from the University of Maryland and Princeton University demonstrated a larger-scale STH system. Using a 34 mm inner diameter quartz tube, they processed up to 1.0 g of PP in 35 minutes in batch mode. This setup used a 2.3 mm thick carbon felt heater layer and copper foil electrodes to generate the required Joule heating [15].
Another related method, solvothermal liquefaction, combines electrified heating with an organic solvent like acetone. Operating at 662°F (350°C) for 90 minutes, this technique achieved a 75.43% conversion rate for polyethylene cable waste. The resulting oil products had a heating value of 43.83 MJ/kg, comparable to gasoline’s 43.4 MJ/kg. The solvent enhances mass and thermal energy transfer, reducing the thermal cracking temperature [17].
| Feature | Electrified STH | Conventional Pyrolysis | Solvothermal Liquefaction |
|---|---|---|---|
| Temperature | ~1,112°F (600°C), pulsed | 842–1,472°F (450–800°C), continuous | ~662°F (350°C) |
| Heating Method | Pulsed Joule heating | Continuous external heat | Solvent-mediated heating |
| Catalyst | None required | Often required | Can use ionic liquids |
| Primary Benefit | High selectivity; avoids side reactions | Well-established technology | Lower temperatures; handles wet/unsorted waste |
While these laboratory results are encouraging, scaling these methods to industrial levels introduces new challenges.
Scalability Considerations
Transitioning from laboratory to industrial applications is no small feat. One significant challenge is shifting from batch reactions to continuous processing. Prototype designs for continuous STH systems have been proposed using commercially available components, hinting at possible industrial use [15].
Maintaining selectivity on a larger scale demands precise control of the pulsed electrical currents. The system must consistently deliver high peak temperatures for 0.11 seconds every 1.10 seconds to prevent unwanted side reactions [15]. Additionally, as reactor sizes increase, thermal management becomes more complex. Sustaining a stable spatial temperature gradient is critical to ensure continuous melting, wicking, and vaporization of plastics.
The bilayer carbon felt structure scales relatively well, with demonstrations progressing from 10.5 mm to 34 mm diameter systems [15]. However, the processing volumes are still modest compared to traditional recycling facilities. Currently, mechanical recycling reintegrates only about 2% of end-of-life packaging plastics into manufacturing [2]. Advanced thermochemical methods like this one offer the potential to improve recycling rates significantly.
This method’s strengths and limitations will be further compared to other depolymerization techniques in subsequent sections.
This content is for informational purposes only. Please consult official regulations and qualified professionals before making sourcing or formulation decisions.
4. Chemical Depolymerization Routes (Methanolysis, Glycolysis, Hydrolysis)
Mechanism
Chemical depolymerization uses solvents to break down condensation polymers like PET, polyamides, polycarbonates, and polyurethanes [1]. For PET, the process targets ester bonds through three main methods: glycolysis, methanolysis, and hydrolysis.
Glycolysis involves a transesterification reaction where PET reacts with ethylene glycol, breaking ester linkages and forming bis(2-hydroxyethyl) terephthalate (BHET) [19]. Methanolysis uses methanol at elevated temperatures and pressures – often as superheated vapor – to produce dimethyl terephthalate (DMT) and ethylene glycol (EG) [19][21]. Hydrolysis breaks ester bonds using water under neutral (high-pressure steam), alkaline (sodium hydroxide), or acidic (sulfuric acid) conditions, yielding terephthalic acid (TPA) and EG [19][20].
Among these, glycolysis is the least energy-intensive, while hydrolysis requires the most energy due to the added complexity of salt handling and purification [18][21]. Hydrolysis is autocatalytic, meaning the reaction speeds up as carboxyl groups accumulate [20].
Efficiency Metrics
Different methods produce varying yields depending on conditions and catalysts. With optimized zinc acetate catalysis, methanolysis achieves DMT yields of 98%, while supercritical methanolysis at 572°F (298°C) can reach up to 99.79% [19]. Alkaline hydrolysis typically produces TPA yields above 90%, whereas neutral hydrolysis yields range from 76% to 84% [19]. Glycolysis benefits significantly from homogeneous catalysis, which enhances reaction rates compared to heterogeneous methods [22].
"We reasonably expect a PET depolymerization process to be able to convert PET into monomers with an energy demand between 6,000 and 10,000 kJ/kg of PET."
– Adam McNeeley and Y. A. Liu, Virginia Polytechnic Institute and State University [18]
| Method | Primary Product | Complexity | Energy Intensity | Best Use Case |
|---|---|---|---|---|
| Glycolysis | BHET | Low | Lowest | Clean PET waste; simple purification [18][21] |
| Methanolysis | DMT | Moderate | Moderate | Challenging purification; high-quality monomer needs [18][21] |
| Hydrolysis | TPA | High | Highest | Heterogeneous PET waste; direct TPA recovery [20][21] |
These metrics help determine the suitability of each method for specific applications.
Scalability Considerations
Glycolysis is the most scalable option for clean PET streams due to its lower energy requirements and straightforward process design [18]. Methanolysis is becoming a preferred choice for contaminated waste, as DMT can be purified through evaporation and distillation, though the reaction itself tends to be slower [18][21]. Hydrolysis, while effective for mixed or contaminated waste, faces scalability challenges due to the high costs of purifying TPA from post-reaction mixtures. Additional precipitation steps are often required, which generate inorganic salt waste [19].
"Hydrolysis effectively processes heterogeneous PET waste streams, including copolyesters, offering the direct recovery of terephthalic acid (TPA), the primary monomer in the PET industry."
– Polymer Degradation and Stability [20]
Current Applications
Chemical depolymerization is particularly useful for handling low-purity or contaminated plastic waste, where mechanical recycling falls short [18][2]. As of February 2024, the gr3n alkaline hydrolysis process serves as a leading example of industrial-scale implementation. This process combines PET with a caustic solution and an EG co-solvent, using a multi-effect evaporation system to concentrate salts and an electrolysis unit to regenerate sodium chloride into sodium hydroxide and hydrochloric acid for reuse [21].
One promising area is fiber-to-fiber recycling in synthetic textiles. Chemical depolymerization can tackle dyes and mixed-material contaminants that mechanical methods cannot process effectively [18].
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
5. Quantum Dot-Assisted Photodriven Depolymerization
Mechanism
Quantum dot-assisted depolymerization leverages carbon quantum dots (CQDs) as photothermal agents. When exposed to LED light, these CQDs convert light energy into localized heat, creating intense thermal gradients that break apart polymer chains. This method allows for precise control over the process – both spatially and temporally – by adjusting the light’s intensity or wavelength. Importantly, it only targets polymers that incorporate CQDs, reducing the formation of reactive intermediates and oxidized byproducts [23][3].
"This is the first instance for depolymerization where we’re using things that take the light energy in and release it as heat instead of releasing it as equivalent chemical energy."
– Clotilde Tagnon, Graduate Student, Stache Lab [23]
Efficiency Metrics
In 2024, researchers at the Stache Lab at Princeton University, led by Assistant Professor Erin Stache and lead author Liat Kugelmass, demonstrated the first use of CQDs for photothermal chemical recycling. Their work successfully depolymerized plastics such as PMMA, PS, PET, and PLA, achieving monomer yields ranging from 43% to 95% [3][23]. A standout result was observed with poly(α-methylstyrene) (PAMS), where the CQD method produced less than 1% oxidized byproducts, compared to 16% with traditional bulk heating at 290°C. Unlike industrial pyrolysis, which heats entire reactors to temperatures between 572–932°F (300–500°C), this method uses light to heat only the nanoparticle sites. This localized heating keeps the bulk material near ambient temperature while still generating the high localized temperatures required for depolymerization. Additionally, the CQDs are designed with low fluorescence to optimize heat release [23].
Scalability Considerations
While lab results are promising, scaling this technology to industrial levels presents challenges. One significant issue is limited light penetration in bulk materials, which complicates the process. Other hurdles include the synthesis of CQDs, effective separation methods, and precise dosage control [23][24][5]. A potential solution involves embedding CQDs into plastics during their initial manufacturing, enabling targeted depolymerization at the end of the product’s life cycle. Carbon-based nanomaterials also offer a cost advantage over more expensive alternatives like plasmonic metals, making this approach more feasible for industrial applications. Furthermore, the dramatic reduction in oxidized byproducts – from 16% with conventional methods to under 1% – could reduce the need for extensive post-process purification [23][3].
"One of the biggest challenges for the future of Stache’s process is scalability, or, bringing the method to the level of chemical recycling as practiced today in massive industrial reactors."
– Erin Stache, Assistant Professor of Chemistry, Princeton University [23]
This CQD-based method represents an exciting addition to the toolkit of depolymerization strategies, pushing forward advancements in chemical recycling.
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
What Is Depolymerization In Plastic Recycling? – Earth Science Answers
Comparison of Depolymerization Methods
This section examines the trade-offs between different depolymerization methods, focusing on their operating conditions, efficiency, and scalability. Each method offers distinct advantages and limitations, which are summarized in the table below to help guide their application in specific scenarios.
| Method | Operating Temperature | Monomer Yield | Primary Advantages | Main Limitations | Scalability Status |
|---|---|---|---|---|---|
| Photodriven | 194–248°F (90–120°C) [3] | Up to 82% [3] | Low energy use; precise timing control | Requires specific polymer end-groups (RAFT/ATRP) | Research stage |
| Enzymatic | Ambient to 158°F (70°C) [2] | Variable (polymer-specific) | High selectivity; eco-friendly; low energy | Slow reaction rates; sensitive to contaminants | Pilot/Commercial |
| Thermochemical | 662–1,472°F (350–800°C) [2] | Variable (often <60% for monomers) | Handles mixed and contaminated waste | Energy-intensive; often produces fuels instead of pure monomers | Industrial/Commercial [2] |
| Chemical (Solvolysis) | 302–842°F (150–450°C) [2][3] | Up to 99.79% (methanolysis) [19] | High-purity monomers; proven for PET and polyurethanes | Requires solvents and pre-sorting | Industrial [25] |
| Quantum Dot-Assisted | Ambient bulk temperature [3] | 43–95% [3] | Extremely selective; minimal oxidized by-products (<1%) | Catalyst recovery challenges; limited light penetration | Laboratory |
Light-driven methods, including photodriven and quantum dot-assisted techniques, stand out for their ability to minimize energy consumption by avoiding the need to heat entire reactors [3]. However, thermochemical processes require substantial heat input, making them energy-intensive [2]. Chemical depolymerization occupies a middle ground, balancing energy use and monomer purity [2][3].
Scalability varies significantly across methods. Thermochemical and chemical solvolysis processes have already been implemented at the industrial level, whereas enzymatic depolymerization is in the transition phase from pilot to commercial scale [2][25]. In contrast, photodriven and quantum dot-assisted methods remain in early research stages, facing challenges like limited light penetration and difficulties in catalyst recovery [3]. Interestingly, quantum dot-assisted depolymerization shows potential for lowering purification costs, as it produces less than 1% oxidized by-products compared to 16% generated by conventional bulk heating at 554°F (290°C) [3].
Selecting the right method depends on factors such as the type of plastic, contamination levels, and the desired purity of monomers. For instance, thermochemical methods are better suited for mixed waste, while chemical and enzymatic approaches are ideal for high-value polymers requiring greater purity.
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Conclusion
Each depolymerization method comes with its own set of strengths and limitations for industrial use. Photodriven depolymerization works under mild temperatures but requires polymers with specific end-groups, such as RAFT or ATRP, which limits its scalability for now. Enzymatic depolymerization is highly selective and operates under gentle conditions, especially for PET, but faces challenges like slow reaction rates and sensitivity to contaminants. Thermochemical processes handle mixed and contaminated waste effectively but require significant energy and often produce unselective product streams. Chemical solvolysis methods – like methanolysis, glycolysis, and hydrolysis – recover high-purity monomers but demand careful solvent handling and pre-sorting. Meanwhile, quantum dot-assisted techniques, though hindered by issues like catalyst recovery and light penetration, offer potential by reducing oxidized by-products compared to traditional methods.
Looking ahead, the future of chemical recycling depends heavily on ongoing technological advancements. Electrified and light-driven methods are emerging as promising solutions to reduce the carbon footprint of chemical recycling by replacing energy-intensive furnace heating with more precise, localized energy inputs. For instance, in 2024, solar thermal catalysis using carbon nanotubes achieved 100% PET conversion and a 51% BHET yield at just 150°C (302°F) – a process that was nearly three times more efficient than traditional thermal methods[3]. Additionally, developing polymers with reactivatable end-groups or weaker backbone bonds could pave the way for lower-temperature depolymerization, potentially reshaping the economic and environmental viability of recycling[3].
Despite these advancements, global recycling rates remain alarmingly low. Only 9% of plastic waste is currently recycled, with the majority either downcycled into lower-quality products or discarded altogether[26][28]. Achieving a true circular plastics economy will require more than just innovative technologies – it will demand substantial investment and a commitment to meeting circularity goals[27]. Scaling these breakthroughs to industrial levels is a critical hurdle, and as highlighted, the trade-offs of each method emphasize the need for customized approaches to industrial plastic recycling.
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
FAQs
Which plastics work best with each depolymerization method?
The type of plastic determines the best method for breaking it down. For example, thermoplastics like PET (Polyethylene Terephthalate) are often broken apart using chemical or enzymatic processes. On the other hand, polyolefins like polyethylene are tougher to handle and usually require catalytic or thermal methods to break them down effectively.
Can depolymerization handle dirty or mixed plastic waste?
Depolymerization is capable of handling dirty or mixed plastic waste, but its performance can drop when faced with contamination, additives, dyes, or the diverse makeup of plastics. These elements can complicate the process and lead to less consistent results.
How close are these methods to large-scale use in the U.S.?
As research and development efforts progress, these methods are moving closer to widespread adoption across the U.S. However, hurdles such as energy efficiency and cost-effectiveness still need to be tackled. Ongoing advancements aim to resolve these challenges, paving the way for broader industrial use.





Comments are closed