Surfactants are molecules with a dual nature: a water-loving (hydrophilic) head and a water-repelling (hydrophobic) tail. This unique structure enables them to reduce surface tension and form micelles – tiny molecular clusters – once a specific concentration, known as the Critical Micelle Concentration (CMC), is reached.
The CMC is a key threshold for optimizing surfactant usage in applications like cleaning, drug delivery, and oil recovery. Below the CMC, surfactants exist as single molecules, while above it, they self-assemble into micelles, boosting performance without unnecessary waste. Factors like molecular structure, ionic strength, and temperature significantly influence the CMC.
Understanding these principles helps industries – from pharmaceuticals to petroleum – improve formulations and efficiency. Whether you’re creating detergents, stabilizing emulsions, or developing drug delivery systems, knowing how surfactants behave at and above their CMC is essential for achieving desired results.
Surfactant Structure and Amphiphilic Properties
Molecular Structure of Surfactants
Surfactants are composed of two distinct parts: a hydrophilic (water-attracting) head and a hydrophobic (water-repelling) tail. This dual nature allows them to arrange themselves at interfaces, making them highly effective in various industrial roles.
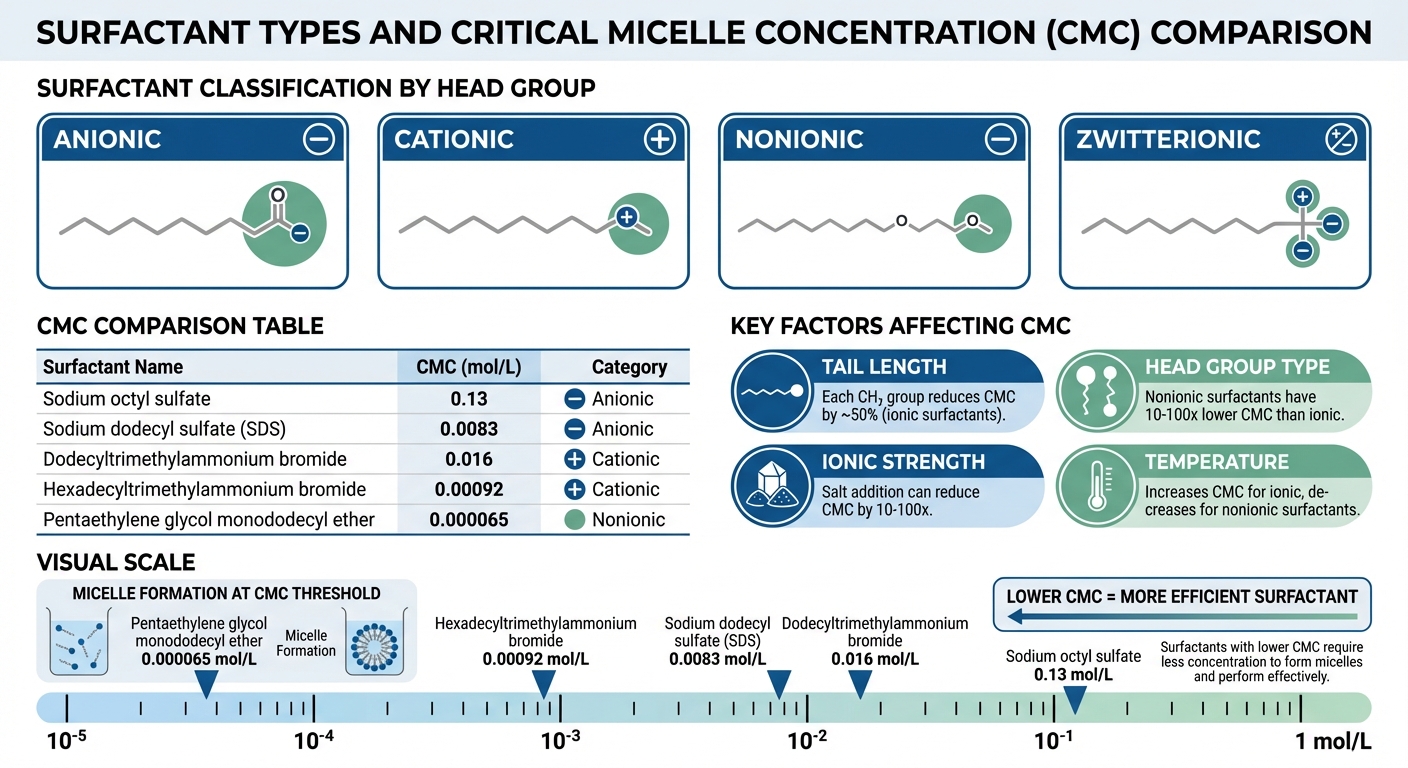
The hydrophilic head can carry different types of charges. For example, it can be anionic (negative, like in Sodium Lauryl Sulfate), cationic (positive, such as Benzalkonium Chloride), non-ionic (uncharged, like Triton X-100), or zwitterionic (containing both positive and negative charges, like Cocamidopropyl Betaine) [1][2]. The hydrophobic tail is typically a hydrocarbon chain, though specialized applications may require fluorocarbon or siloxane chains [1][2]. This amphiphilic design is what enables surfactants to organize into micelles and assemble at interfaces once the critical micelle concentration (CMC) is reached [3].
"A surfactant… is a substance that, when present at low concentration in a system, has the property of adsorbing onto the surfaces or interfaces of the system and of altering to a marked degree the surface or interfacial free energies."
– Milton J. Rosen, Author of Surfactants and Interfacial Phenomena [1][2]
These structural features are central to their functionality across many industries.
Industrial Uses of Surfactants
The unique molecular design of surfactants directly influences their diverse applications across industries.
In pharmaceuticals, surfactants play a critical role in improving the bioavailability of poorly water-soluble drugs by encapsulating them in micelles. Pulmonary surfactants, such as Survanta (beractant), are used to treat respiratory distress syndrome by enhancing lung compliance [1][2].
In sectors like food, cosmetics, and agrochemicals, surfactants act as emulsifiers and wetting agents, ensuring product stability and uniform distribution [1][2]. In the petroleum industry, surfactants such as alkyl aryl sulfonates are used in Enhanced Oil Recovery (EOR) to reduce interfacial tension and improve oil extraction efficiency [3].
The global production of surfactants is estimated at approximately 15 million tons annually, with linear alkylbenzene sulfonates (LAS) accounting for around 1.7 million tons [1][2]. Their importance is evident in applications ranging from household cleaning products, which trap dirt and oils in micelles, to environmental emergencies. For instance, during the April 2010 Deepwater Horizon oil spill, dispersants like Corexit (containing compounds such as Dioctyl Sodium Sulfosuccinate and Tween‑80) were used to break oil into smaller droplets, aiding microbial degradation [1][2].
These examples highlight how the structure of surfactants translates into practical solutions for challenges across various industries.
For specialty surfactants and technical-grade chemicals, Allan Chemical Corporation (https://allanchems.com) offers over 40 years of expertise to meet industry needs.
This content is provided for informational purposes only. Always consult official guidelines and qualified professionals before making sourcing or formulation decisions.
sbb-itb-aa4586a
CMC in Surface Chemistry | Critical Micelle Concentration | Determination & Significance in Pharmacy
The Hydrophobic Effect in Self-Assembly
The hydrophobic effect is a key force that drives surfactants to organize themselves in water. When nonpolar groups, like the tails of surfactant molecules, are introduced into water, they naturally cluster together to reduce their interaction with water molecules [8]. This behavior stems from how water molecules rearrange themselves around nonpolar groups.
"The hydrophobic effect is the observed tendency of nonpolar substances to aggregate in an aqueous solution and to be excluded by water." – Wikipedia [8]
At the molecular level, water molecules form ordered, cage-like structures around the hydrophobic tails of surfactants. This arrangement significantly limits the movement of water molecules. For instance, the reorientational correlation time of water increases from 2 to 4–8 picoseconds near large nonpolar molecules [8]. This restricted movement causes a notable loss of entropy, making the dissolved state less thermodynamically favorable.
When surfactants aggregate into structures like micelles, their hydrophobic tails group together in a core, shielding them from water. This process releases the water molecules that were previously trapped in ordered structures, allowing them to return to the bulk solution. The result? A significant increase in the system’s overall entropy [9]. Essentially, micellization is a dehydration process that frees up water molecules [10][11]. This molecular rearrangement is the foundation for the precise, geometry-driven self-assembly of surfactants.
How Surfactants Form Organized Structures
Surfactants spontaneously assemble into specific structures based on their molecular geometry. This behavior is governed by the packing parameter ($N_s$), which considers the volume of the hydrophobic tail, the area of the head group, and the tail length [4]. For example:
- Cone-shaped surfactants ($N_s$ ≤ 1/3) form spherical micelles.
- Wedge-shaped surfactants (1/3 < $N_s$ ≤ 1/2) create cylindrical micelles.
- Cylinder-shaped surfactants ($N_s$ > 1/2) assemble into bilayers or vesicles [4].
A typical spherical micelle has a radius of 1–3 nanometers, roughly matching the length of an extended surfactant molecule [9]. This self-assembly happens naturally once the surfactant concentration exceeds the Critical Micelle Concentration (CMC). For instance, sodium dodecyl sulfate (SDS) has a CMC of 8.3 x 10⁻³ mol/L in water at 77°F (25°C) [3][4]. Below the CMC, surfactants exist as individual molecules, but above it, they rapidly form organized aggregates. These structures play a pivotal role in influencing the performance of formulations across industries.
Importance in Product Formulations
Understanding the hydrophobic effect is essential for creating stable and effective formulations. In drug delivery, micelles can encapsulate poorly water-soluble drugs at concentrations far above the CMC [11]. Similarly, block copolymers utilize hydrophobic forces to form nanostructures that protect and deliver pharmaceutical compounds [4][6].
In the petroleum sector, maintaining surfactant concentrations slightly above the CMC is critical for Enhanced Oil Recovery (EOR) [3]. For cleaning products, the hydrophobic effect helps surfactants adjust contact angles and emulsify oily soils [3]. However, in systems with large interfaces, surfactant levels dropping below the CMC can destabilize formulations [3].
Allan Chemical Corporation (https://allanchems.com) supports these applications by offering sourcing expertise for specialty surfactants and technical-grade chemicals, drawing on over 40 years of industry experience.
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Micelle Formation and Critical Micelle Concentration (CMC)
CMC Values and Properties of Different Surfactant Types
What Micelles Are
Micelles are colloidal aggregates formed by surfactant molecules, where the hydrophilic heads face outward and hydrophobic tails cluster inward. This arrangement, driven by the hydrophobic effect, exists in dynamic balance with free monomers in solution [13]. In water, these structures adopt a "normal-phase" configuration.
"Particle of colloidal dimensions that exists in equilibrium with the molecules or ions in solution from which it is formed." – IUPAC [13]
A single micelle typically includes 30–100 surfactant molecules, referred to as the aggregation number [5][2]. While spherical micelles are most common, they can also take on ellipsoidal, cylindrical, or bilayer shapes based on molecular structure and environmental conditions [13]. The hydrophobic effect drives this self-assembly by releasing structured water molecules surrounding the surfactant tails, which increases the system’s entropy [13][2]. To form micelles, the surfactant concentration must exceed the CMC and the temperature must be above the Krafft point [5][13]. Let’s take a closer look at the critical micelle concentration.
Defining Critical Micelle Concentration (CMC)
The CMC is the lowest concentration of surfactant needed for micelles to form [2][3]. Below this threshold, surfactant molecules remain as individual monomers, reducing surface tension as they adsorb at interfaces. Once the CMC is reached, the interface becomes saturated, and additional surfactant molecules aggregate into micelles rather than further reducing surface tension [3][7].
"There is a relatively small range of concentrations separating the limit below which virtually no micelle are detected and the limit above which virtually all additional surfactant molecules form micelles." – IUPAC Gold Book [14]
For sodium dodecyl sulfate (SDS), a commonly used anionic surfactant, the CMC in water at 77°F (25°C) is approximately 8.2 x 10⁻³ mol/L [2][3]. This value is critical for determining the minimum surfactant concentration needed to achieve specific interfacial properties and to enhance the solubilization of hydrophobic substances [2]. Applications such as Enhanced Oil Recovery (EOR) often utilize surfactants with extremely low CMCs, ranging from 10⁻⁵ to 10⁻⁴ mol/L [7].
Variables That Affect CMC
Several factors influence the CMC, with the hydrophobic tail length being one of the most significant. For ionic surfactants, each additional methylene (CH₂) group in the tail reduces the CMC by roughly half [2][4]. This occurs because longer tails strengthen hydrophobic interactions, making micelle formation more favorable.
The head group type also has a major impact. Nonionic surfactants generally exhibit much lower CMC values than ionic surfactants – sometimes by factors of 10 to 100 – because they lack the electrostatic repulsion between charged head groups that can hinder aggregation [2][4].
Below is a table showcasing typical CMC values for various surfactants:
| Surfactant | CMC (mol/L) | Category |
|---|---|---|
| Sodium octyl sulfate | 0.13 | Anionic |
| Sodium dodecyl sulfate (SDS) | 0.0083 | Anionic |
| Dodecyltrimethylammonium bromide | 0.016 | Cationic |
| Hexadecyltrimethylammonium bromide | 0.00092 | Cationic |
| Pentaethylene glycol monododecyl ether | 0.000065 | Nonionic |
Ionic strength is another considerable factor. Adding salts like sodium chloride reduces electrostatic repulsion between charged head groups, which can lower the CMC by factors of 10 to 100. Counterions can neutralize up to 92% of an ionic micelle’s charge [13]. Temperature also plays a role: for ionic surfactants, the CMC increases with temperature above the Krafft point, while for nonionic surfactants, it decreases as temperature rises due to dehydration of the hydrophilic head groups [2][4]. Additionally, molecular architecture matters – Gemini surfactants, which feature two linked heads and tails, have CMC values one to two orders of magnitude lower than their single-chain counterparts [2].
This information is intended for educational purposes. Always consult official guidelines and experts for sourcing or formulation decisions.
Thermodynamics and Factors Affecting Micellization
Thermodynamic Principles of Micelle Formation
Micelle formation hinges on the Gibbs free energy (∆Gₘᵢc) of the process, which must be negative for spontaneous self-assembly to occur. For non-ionic surfactants, this relationship is often expressed as ∆Gₘᵢc ≈ RT ln(CMC), while for ionic surfactants, it is modified to ∆Gₘᵢc ≈ (1 + β)RT ln(CMC), where β represents the degree of counter-ion binding [4]. For example, Sodium Dodecyl Sulfate (SDS) has a ∆G°ₘᵢc of about –22.0 kJ/mol, while Cetyl Trimethylammonium Bromide (CTAB) shows a more negative ∆G°ₘᵢc of approximately –30.5 kJ/mol, indicating a stronger thermodynamic drive for micelle formation [4].
The process is primarily driven by entropy. As the hydrophobic tails of surfactants aggregate, they release ordered water molecules, leading to an increase in system entropy [4]. Professor Laurence S. Romsted from Rutgers University explains:
"Aggregation is driven by the hydrophobic effect, the minimization of surfactant tail-water contact by transfer of the tail of the surfactant unimer from water to the aggregate core with the concomitant release of water of hydration, and an increase in the entropy of the system" [12].
However, micelle formation is not without resistance. Repulsive interactions among head groups – due to electrostatic, hydration, or steric effects – counteract the process. The dressed micelle model breaks down the Gibbs free energy into contributions from favorable hydrophobic interactions, unfavorable head group repulsions, and interfacial energy at the micelle surface [4].
These principles provide the foundation for understanding how external factors influence micellization.
Environmental Conditions and Their Effects
Temperature plays a distinct role in the micellization of ionic and non-ionic surfactants. For ionic surfactants, the critical micelle concentration (CMC) typically shows a non-monotonic pattern, with a minimum near the Krafft temperature – the point below which micelle formation does not occur [4]. In contrast, non-ionic surfactants generally exhibit a decrease in CMC as temperature rises. This occurs because the dehydration of polyoxyethylene head groups at higher temperatures promotes aggregate formation [2]. At sufficiently high temperatures, non-ionic surfactants may reach their cloud point, where micelles aggregate into a separate phase [4].
Ionic strength also significantly impacts micellization in ionic surfactants. Adding salts like Sodium Chloride reduces electrostatic repulsion between charged head groups, effectively lowering the CMC [4]. Similarly, pH influences the ionization state of surfactants with ionizable head groups, affecting both the CMC and micelle stability [2][16].
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Methods for Measuring Self-Assembly
Experimental techniques play a crucial role in validating theoretical models of surfactant self-assembly. These methods not only confirm thermodynamic predictions but also offer practical insights for formulation development.
Surface Tension Measurements
Measuring surface tension is one of the simplest ways to determine the critical micelle concentration (CMC). When surfactant molecules are added to water, they migrate to the air-liquid interface, reducing surface tension. This reduction continues until the interface becomes saturated with surfactant monomers. At this saturation point, any additional surfactant molecules begin forming micelles in the bulk solution. The CMC, therefore, marks the transition where surface tension levels off, forming a plateau as the surface monomer concentration stabilizes [3][19].
To pinpoint the CMC, researchers plot surface tension against the logarithm of surfactant concentration. The CMC is identified as the point where the downward slope of the curve intersects the horizontal plateau. For example, Sodium Dodecyl Sulfate (SDS) has a CMC of approximately 8×10⁻³ mol/L [3][20]. Automated tensiometers equipped with dosing systems enhance accuracy by reducing operator bias [20]. Additionally, mathematical model fitting is increasingly used to identify the CMC more precisely, minimizing subjective interpretation [3].
While tensiometry is widely used, spectroscopic methods offer greater sensitivity, especially for surfactants with very low CMCs.
Spectroscopic Techniques
Fluorescence spectroscopy is particularly effective for detecting micelle formation in surfactants with low CMC values, where other techniques may fall short [18]. This method relies on fluorescent probes like pyrene or Nile Red, which exhibit changes in their emission properties when they transition from the polar aqueous phase to the hydrophobic micelle core. As Wu et al. explain:
"Fluorescence spectroscopy, based on the differences in fluorescence intensity and wavelength of the fluorescent probe in the solvent phase and micellar phase, can sensitively detect the CMC of surfactants" [17][21].
The pyrene I/III ratio, which compares the intensity of its first and third emission peaks, is commonly used to monitor micelle formation. A sharp change in this ratio indicates the probe’s movement into the micelle interior [11].
High-resolution ultrasound spectroscopy (HR-US) provides another innovative approach. By measuring changes in sound speed and attenuation, this technique captures detailed information about the compressibility and hydration of surfactant molecules as they transition from unimers (individual molecules) to micelles [11].
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
Conclusion and Practical Applications
Key Points
The Critical Micelle Concentration (CMC) is the threshold concentration at which surfactants achieve their full interfacial properties, such as reducing surface tension and enhancing solubilization. Working near this concentration helps optimize processes like cleaning, emulsification, and drug delivery while minimizing waste and costs. The structure of the surfactant significantly influences the CMC. For example, adding a single methylene (CH₂) group to the hydrophobic tail of ionic surfactants typically halves the CMC, while for nonionic surfactants, every two CH₂ groups can lower it by a factor of 10. Additionally, external conditions – such as temperature, pH, salinity, and electrolyte presence – can cause notable shifts in the CMC, making constant monitoring essential for industrial applications [2][15]. These principles provide a foundation for improving processes across various industries.
Industry Applications
Understanding and controlling the CMC offers practical advantages in a variety of fields. By fine-tuning surfactant behavior, manufacturers can improve product performance and efficiency across sectors.
In pharmaceuticals, micelles formed above the CMC act as nanocarriers for hydrophobic drugs, protecting them and improving their absorption by the body. This enhances bioavailability, a critical factor in drug development [2]. The global surfactant market, valued at $43,655 million in 2017, is expected to grow to $66,408 million by 2025, with a compound annual growth rate (CAGR) of 5.4% [11]. For cosmetics and personal care, CMC knowledge helps formulators create stable emulsions for creams and lotions while managing toxicity, which is often linked to concentrations near or above the CMC for nonionic surfactants [11]. In enhanced oil recovery (EOR), surfactants with extremely low CMCs – between 10⁻⁵ and 10⁻⁴ mol/L – are used to reduce interfacial tension between oil and water, maximizing oil extraction efficiency [7].
For sourcing surfactants and specialty chemicals tailored to these needs, Allan Chemical Corporation offers technical- and compendial-grade materials (USP, FCC, ACS, NF). With over 40 years of experience in regulated industries, they provide reliable just-in-time delivery and maintain strong supplier partnerships to ensure consistent availability for pharmaceutical, cosmetic, and food manufacturing applications.
This content is for informational purposes only. Always consult relevant regulations and qualified professionals when making sourcing or formulation decisions.
FAQs
How do I choose the right surfactant concentration relative to the CMC?
To determine the ideal surfactant concentration, target a level at or slightly above the critical micelle concentration (CMC). Below this threshold, surfactant molecules exist as individual monomers, whereas micelles begin to form once the CMC is surpassed. Operating just above the CMC promotes efficient micelle formation, which is essential for tasks like solubilization and emulsification, while avoiding unnecessary surfactant use. Precise CMC measurement is crucial for achieving the best results.
Why does adding salt change the CMC so much for ionic surfactants?
When salt is added, it decreases the electrostatic repulsion between the charged head groups of ionic surfactants. This reduction lowers the critical micelle concentration (CMC), which means micelles can form more easily. Consequently, the self-assembly process becomes more effective.
What’s the easiest way to measure CMC in a formulation?
The easiest method to determine the critical micelle concentration (CMC) is by measuring surface tension. This technique identifies the point where micelles begin to form, indicated by a noticeable change in surface tension. For surfactants with the right characteristics, spectroscopic methods – such as fluorescence polarization or UV-Vis spectroscopy – can provide fast and sensitive results. However, surface tension measurements remain the go-to approach for routine CMC analysis due to their simplicity.





Comments are closed